Abstract
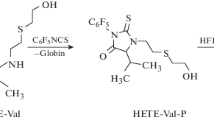
As part of a program to develop methods for the verification of alleged exposure to sulphur mustard, we synthesized and characterized three amino acid adducts presumably formed by alkylation of haemoglobin: 4-(2-hydroxyethylthioethyl)-l-aspartate, 5-(2-hydroxyethylthioethyl)-l-glutamate and N1- and N3-(2-hydroxyethylthioethyl)-l-histidine. Suitable derivatization methods for GC/MS analysis were developed for these adducts as well as for the cysteine and the N-terminal valine adduct. Incubation of human blood with [35S]sulphur mustard in vitro followed by acidic hydrolysis of isolated globin and derivatization with Fmoc-Cl afforded three major radioactive peaks upon HPLC analysis, one of which coeluted with the synthetic Fmoc derivative of N1/N3-(2-hydroxyethylthioethyl)-l-histidine. After pronase digestion of globin the adducts of histidine, glutamic acid, aspartic acid, cysteine and N-terminal valine could be tentatively identified and quantitated. Final identification was obtained from GC/MS analysis. The most abundant adduct, N1/N3-(2-hydroxyethylthioethyl)-l-histidine, could not be sensitively analysed by GC/MS. A convenient LC-tandem MS procedure was developed for this compound, enabling the detection of exposure of human blood to 10␣μM sulphur mustard in vitro.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 29 May 1996 / Accepted: 20 August 1996
Rights and permissions
About this article
Cite this article
Noort, D., Hulst, A., Trap, H. et al. Synthesis and mass spectrometric identification of the major amino acid adducts formed between sulphur mustard and haemoglobin in human blood. Arch Toxicol 71, 171–178 (1997). https://doi.org/10.1007/s002040050372
Issue Date:
DOI: https://doi.org/10.1007/s002040050372