Abstract
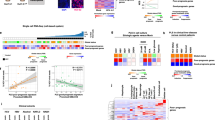
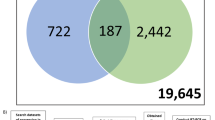
Preventing clinical drug-induced liver injury (DILI) remains a major challenge, because DILI develops via multifactorial mechanisms. Immune and inflammatory reactions are considered important mechanisms of DILI; however, biomarkers from in vitro systems using immune cells have not been comprehensively studied. The aims of this study were (1) to identify promising biomarker genes for predicting DILI in an in vitro coculture model of peripheral blood mononuclear cells (PBMCs) with a human liver cell line, and (2) to evaluate these genes as predictors of DILI using a panel of drugs with different clinical DILI risk. Transcriptome-wide analysis of PBMCs cocultured with HepG2 or differentiated HepaRG cells that were treated with several drugs revealed an appropriate separation of DILI-positive and DILI-negative drugs, from which 12 putative biomarker genes were selected. To evaluate the predictive performance of these genes, PBMCs cocultured with HepG2 cells were exposed to 77 different drugs, and gene expression levels in PBMCs were determined. The MET proto-oncogene receptor tyrosine kinase (MET) showed the highest area under the receiver-operating characteristic curve (AUC) value of 0.81 among the 12 genes with a high sensitivity/specificity (85/66%). However, a stepwise logistic regression model using the 12 identified genes showed the highest AUC value of 0.94 with a high sensitivity/specificity (93/86%). Taken together, we established a coculture system using PBMCs and HepG2 cells and selected biomarkers that can predict DILI risk. The established model would be useful in detecting the DILI potential of compounds, in particular those that involve an immune mechanism.






Similar content being viewed by others
References
Albrecht W, Kappenberg F, Brecklinghaus T et al (2019) Prediction of human drug-induced liver injury (DILI) in relation to oral doses and blood concentrations. Arch Toxicol 93(6):1609–1637. https://doi.org/10.1007/s00204-019-02492-9
Aleo MD, Shah F, Allen S et al (2020) Moving beyond binary predictions of human drug-induced liver injury (DILI) toward contrasting relative risk potential. Chem Res Toxicol 33(1):223–238. https://doi.org/10.1021/acs.chemrestox.9b00262
Andrade RJ, Chalasani N, Bjornsson ES et al (2019) Drug-induced liver injury. Nat Rev Dis Primers 5(1):58. https://doi.org/10.1038/s41572-019-0105-0
Ball P, Mandell L, Niki Y, Tillotson G (1999) Comparative tolerability of the newer fluoroquinolone antibacterials. Drug Saf 21(5):407–421. https://doi.org/10.2165/00002018-199921050-00005
Bell LN, Chalasani N (2009) Epidemiology of idiosyncratic drug-induced liver injury. Semin Liver Dis 29(4):337–347. https://doi.org/10.1055/s-0029-1240002
Björnsson ES (2014) Epidemiology and risk factors for idiosyncratic drug-induced liver injury. Semin Liver Dis 34(2):115–122. https://doi.org/10.1055/s-0034-1375953
CDER (2006) Warnings and precautions, contraindications, and boxed warning sections of labeling for human prescription drug and biological products—content and format. https://www.fda.gov/downloads/Drugs/GuidanceComplianceRegulatoryInformation/Guidances/ucm075096.pdf. Accessed 31 Mar 2020
Chantarangsu S, Mushiroda T, Mahasirimongkol S et al (2009) HLA-B*3505 allele is a strong predictor for nevirapine-induced skin adverse drug reactions in HIV-infected Thai patients. Pharmacogenet Genomics 19(2):139–146. https://doi.org/10.1097/FPC.0b013e32831d0faf
Chen M, Vijay V, Shi Q, Liu Z, Fang H, Tong W (2011) FDA-approved drug labeling for the study of drug-induced liver injury. Drug Discov Today 16(15–16):697–703. https://doi.org/10.1016/j.drudis.2011.05.007
Chen M, Suzuki A, Thakkar S, Yu K, Hu C, Tong W (2016) DILIrank: the largest reference drug list ranked by the risk for developing drug-induced liver injury in humans. Drug Discov Today 21(4):648–653. https://doi.org/10.1016/j.drudis.2016.02.015
Daly AK, Donaldson PT, Bhatnagar P et al (2009) HLA-B*5701 genotype is a major determinant of drug-induced liver injury due to flucloxacillin. Nat Genet 41(7):816–819. https://doi.org/10.1038/ng.379
de Abajo FJ, Montero D, Madurga M, Garcia Rodriguez LA (2004) Acute and clinically relevant drug-induced liver injury: a population based case-control study. Br J Clin Pharmacol 58(1):71–80. https://doi.org/10.1111/j.1365-2125.2004.02133.x
Deng X, Stachlewitz RF, Liguori MJ et al (2006) Modest inflammation enhances diclofenac hepatotoxicity in rats: role of neutrophils and bacterial translocation. J Pharmacol Exp Ther 319(3):1191–1199. https://doi.org/10.1124/jpet.106.110247
Dragovic S, Vermeulen NP, Gerets HH et al (2016) Evidence-based selection of training compounds for use in the mechanism-based integrated prediction of drug-induced liver injury in man. Arch Toxicol 90(12):2979–3003. https://doi.org/10.1007/s00204-016-1845-1
Edling Y, Sivertsson LK, Butura A, Ingelman-Sundberg M, Ek M (2009) Increased sensitivity for troglitazone-induced cytotoxicity using a human in vitro co-culture model. Toxicol Vitro 23(7):1387–1395. https://doi.org/10.1016/j.tiv.2009.07.026
Fontana RJ (2014) Pathogenesis of idiosyncratic drug-induced liver injury and clinical perspectives. Gastroenterology 146(4):914–928. https://doi.org/10.1053/j.gastro.2013.12.032
Funk C, Roth A (2017) Current limitations and future opportunities for prediction of DILI from in vitro. Arch Toxicol 91(1):131–142. https://doi.org/10.1007/s00204-016-1874-9
Gómez-Lechón MJ, Tolosa L, Donato MT (2016) Metabolic activation and drug-induced liver injury: in vitro approaches for the safety risk assessment of new drugs. J Appl Toxicol 36(6):752–768. https://doi.org/10.1002/jat.3277
Granitzny A, Knebel J, Muller M et al (2017) Evaluation of a human in vitro hepatocyte-NPC co-culture model for the prediction of idiosyncratic drug-induced liver injury: a pilot study. Toxicol Rep 4:89–103. https://doi.org/10.1016/j.toxrep.2017.02.001
Higuchi S, Kobayashi M, Yano A et al (2012) Involvement of Th2 cytokines in the mouse model of flutamide-induced acute liver injury. J Appl Toxicol 32(10):815–822. https://doi.org/10.1002/jat.1706
Holt MP, Ju C (2006) Mechanisms of drug-induced liver injury. AAPS J 8(1):E48–54. https://doi.org/10.1208/aapsj080106
Kaplowitz N (2005) Idiosyncratic drug hepatotoxicity. Nat Rev Drug Discov 4(6):489–499. https://doi.org/10.1038/nrd1750
Kato R, Uetrecht J (2017) Supernatant from hepatocyte cultures with drugs that cause idiosyncratic liver injury activates macrophage inflammasomes. Chem Res Toxicol 30(6):1327–1332. https://doi.org/10.1021/acs.chemrestox.7b00065
Kegel V, Pfeiffer E, Burkhardt B et al (2015) Subtoxic concentrations of hepatotoxic drugs lead to Kupffer cell activation in a human in vitro liver model: an approach to study DILI. Mediators Inflamm 2015:640631. https://doi.org/10.1155/2015/640631
Kim DE, Jang MJ, Kim YR et al (2017) Prediction of drug-induced immune-mediated hepatotoxicity using hepatocyte-like cells derived from human embryonic stem cells. Toxicology 387:1–9. https://doi.org/10.1016/j.tox.2017.06.005
Kleiner DE (2017) Drug-induced liver injury: the hepatic pathologist’s approach. Gastroenterol Clin North Am 46(2):273–296. https://doi.org/10.1016/j.gtc.2017.01.004
Komarowska I, Coe D, Wang G et al (2015) Hepatocyte growth factor receptor c-Met instructs T cell cardiotropism and promotes T cell migration to the heart via autocrine chemokine release. Immunity 42(6):1087–1099. https://doi.org/10.1016/j.immuni.2015.05.014
Kono H, Rock KL (2008) How dying cells alert the immune system to danger. Nat Rev Immunol 8(4):279–289. https://doi.org/10.1038/nri2215
Lecoeur S, Andre C, Beaune PH (1996) Tienilic acid-induced autoimmune hepatitis: anti-liver and-kidney microsomal type 2 autoantibodies recognize a three-site conformational epitope on cytochrome P4502C9. Mol Pharmacol 50(2):326–333
Liguori MJ, Ditewig AC, Maddox JF et al (2010) Comparison of TNFα to lipopolysaccharide as an inflammagen to characterize the idiosyncratic hepatotoxicity potential of drugs: Trovafloxacin as an example. Int J Mol Sci 11(11):4697–4714. https://doi.org/10.3390/ijms11114697
Mak A, Kato R, Weston K, Hayes A, Uetrecht J (2018) An impaired immune tolerance animal model distinguishes the potential of troglitazone/pioglitazone and tolcapone/entacapone to cause IDILI. Toxicol Sci 161(2):412–420. https://doi.org/10.1093/toxsci/kfx219
Martinez VG, Sacedon R, Hidalgo L et al (2015) The BMP pathway participates in human naive CD4+ T cell activation and homeostasis. PLoS ONE 10(6):e0131453. https://doi.org/10.1371/journal.pone.0131453
Miyazono K, Kamiya Y, Morikawa M (2010) Bone morphogenetic protein receptors and signal transduction. J Biochem 147(1):35–51. https://doi.org/10.1093/jb/mvp148
Molnarfi N, Benkhoucha M, Bjarnadottir K, Juillard C, Lalive PH (2012) Interferon-β induces hepatocyte growth factor in monocytes of multiple sclerosis patients. PLoS ONE 7(11):e49882. https://doi.org/10.1371/journal.pone.0049882
O'Brien PJ, Irwin W, Diaz D et al (2006) High concordance of drug-induced human hepatotoxicity with in vitro cytotoxicity measured in a novel cell-based model using high content screening. Arch Toxicol 80(9):580–604. https://doi.org/10.1007/s00204-006-0091-3
Oda S, Matsuo K, Nakajima A, Yokoi T (2016) A novel cell-based assay for the evaluation of immune- and inflammatory-related gene expression as biomarkers for the risk assessment of drug-induced liver injury. Toxicol Lett 241:60–70. https://doi.org/10.1016/j.toxlet.2015.10.029
Onakpoya IJ, Heneghan CJ, Aronson JK (2016) Post-marketing withdrawal of 462 medicinal products because of adverse drug reactions: a systematic review of the world literature. BMC Med 14:10. https://doi.org/10.1186/s12916-016-0553-2
Ostapowicz G, Fontana RJ, Schiodt FV et al (2002) Results of a prospective study of acute liver failure at 17 tertiary care centers in the United States. Ann Intern Med 137(12):947–954. https://doi.org/10.7326/0003-4819-137-12-200212170-00007
Proctor WR, Foster AJ, Vogt J et al (2017) Utility of spherical human liver microtissues for prediction of clinical drug-induced liver injury. Arch Toxicol 91(8):2849–2863. https://doi.org/10.1007/s00204-017-2002-1
R Core Team (2019) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. URL https://www.R-project.org/
Senior JR (2014) Evolution of the Food and Drug Administration approach to liver safety assessment for new drugs: current status and challenges. Drug Saf 37(Suppl 1):S9–17. https://doi.org/10.1007/s40264-014-0182-7
Shaw PJ, Ganey PE, Roth RA (2009) Trovafloxacin enhances the inflammatory response to a Gram-negative or a Gram-positive bacterial stimulus, resulting in neutrophil-dependent liver injury in mice. J Pharmacol Exp Ther 330(1):72–78. https://doi.org/10.1124/jpet.109.151068
Smith KS, Smith PL, Heady TN, Trugman JM, Harman WD, Macdonald TL (2003) In vitro metabolism of tolcapone to reactive intermediates: relevance to tolcapone liver toxicity. Chem Res Toxicol 16(2):123–128. https://doi.org/10.1021/tx025569n
Song B, Aoki S, Liu C, Ito K (2019) A toll-like receptor 9 agonist sensitizes mice to mitochondrial dysfunction-induced hepatic apoptosis via the Fas/FasL pathway. Arch Toxicol 93(6):1573–1584. https://doi.org/10.1007/s00204-019-02454-1
Stepan AF, Walker DP, Bauman J et al (2011) Structural alert/reactive metabolite concept as applied in medicinal chemistry to mitigate the risk of idiosyncratic drug toxicity: a perspective based on the critical examination of trends in the top 200 drugs marketed in the United States. Chem Res Toxicol 24(9):1345–1410. https://doi.org/10.1021/tx200168d
Stephens C, Andrade RJ, Lucena MI (2014) Mechanisms of drug-induced liver injury. Curr Opin Allergy Clin Immunol 14(4):286–292. https://doi.org/10.1097/ACI.0000000000000070
Ter Horst R, Jaeger M, Smeekens SP et al (2016) Host and environmental factors influencing individual human cytokine responses. Cell 167(4):1111–1124. https://doi.org/10.1016/j.cell.2016.10.018
Trusolino L, Bertotti A, Comoglio PM (2010) MET signalling: principles and functions in development, organ regeneration and cancer. Nat Rev Mol Cell Biol 11(12):834–848. https://doi.org/10.1038/nrm3012
Uetrecht J (2019) Mechanistic studies of idiosyncratic DILI: clinical implications. Front Pharmacol 10:837. https://doi.org/10.3389/fphar.2019.00837
Usui T, Naisbitt DJ (2017) Human leukocyte antigen and idiosyncratic adverse drug reactions. Drug Metab Pharmacokinet 32(1):21–30. https://doi.org/10.1016/j.dmpk.2016.11.003
Vorrink SU, Zhou Y, Ingelman-Sundberg M, Lauschke VM (2018) Prediction of drug-induced hepatotoxicity using long-term stable primary hepatic 3D spheroid cultures in chemically defined conditions. Toxicol Sci 163(2):655–665. https://doi.org/10.1093/toxsci/kfy058
Weaver RJ, Blomme EA, Chadwick AE et al (2020) Managing the challenge of drug-induced liver injury: a roadmap for the development and deployment of preclinical predictive models. Nat Rev Drug Discov 19(2):131–148. https://doi.org/10.1038/s41573-019-0048-x
Wink S, Hiemstra S, Huppelschoten S et al (2014) Quantitative high content imaging of cellular adaptive stress response pathways in toxicity for chemical safety assessment. Chem Res Toxicol 27(3):338–355. https://doi.org/10.1021/tx4004038
Wuillemin N, Terracciano L, Beltraminelli H et al (2014) T cells infiltrate the liver and kill hepatocytes in HLA-B*57:01-associated floxacillin-induced liver injury. Am J Pathol 184(6):1677–1682. https://doi.org/10.1016/j.ajpath.2014.02.018
Xu JJ, Henstock PV, Dunn MC, Smith AR, Chabot JR, de Graaf D (2008) Cellular imaging predictions of clinical drug-induced liver injury. Toxicol Sci 105(1):97–105. https://doi.org/10.1093/toxsci/kfn109
Yano A, Oda S, Fukami T, Nakajima M, Yokoi T (2014) Development of a cell-based assay system considering drug metabolism and immune- and inflammatory-related factors for the risk assessment of drug-induced liver injury. Toxicol Lett 228(1):13–24. https://doi.org/10.1016/j.toxlet.2014.04.005
Acknowledgements
The authors wish to thank Dr. Tomoya Shimokata and Yuka Murasaki from Department of Clinical Oncology and Chemotherapy, and Dr. Kosuke Yoshida from Department of Drug Safety Sciences/Department of Obstetrics and Gynecology Nagoya University Graduate School of Medicine for taking blood.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest to disclose.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the 1964 Helsinki Declaration and approved by the ethics committee of Nagoya University School of Medicine (No. 2016-0094).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Oda, S., Uchida, Y., Aleo, M.D. et al. An in vitro coculture system of human peripheral blood mononuclear cells with hepatocellular carcinoma-derived cells for predicting drug-induced liver injury. Arch Toxicol 95, 149–168 (2021). https://doi.org/10.1007/s00204-020-02882-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-020-02882-4