Abstract
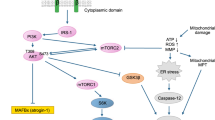
Statins are generally well tolerated, but treatment with these drugs may be associated with myopathy. The mechanisms of statin-associated myopathy are not completely understood. Statins inhibit AKT phosphorylation by an unclear mechanism, whereas insulin-like growth factor (IGF-1) activates the IGF-1/AKT signaling pathway and promotes muscle growth. The aims of the study were to investigate mechanisms of impaired AKT phosphorylation by simvastatin and to assess effects of IGF-1 on simvastatin-induced myotoxicity in C2C12 myotubes. C2C12 mouse myotubes were exposed to 10 μM simvastatin and/or 10 ng/mL IGF-1 for 18 h. Simvastatin inhibited the IGF-1/AKT signaling pathway, resulting in increased breakdown of myofibrillar proteins, impaired protein synthesis and increased apoptosis. Simvastatin inhibited AKT S473 phosphorylation, indicating reduced activity of mTORC2. In addition, simvastatin impaired stimulation of AKT T308 phosphorylation by IGF-1, indicating reduced activation of the IGF-1R/PI3K pathway by IGF-1. Nevertheless, simvastatin-induced myotoxicity could be at least partially prevented by IGF-1. The protective effects of IGF-1 were mediated by activation of the IGF-1R/AKT signaling cascade. Treatment with IGF-1 also suppressed muscle atrophy markers, restored protein synthesis and inhibited apoptosis. These results were confirmed by normalization of myotube morphology and protein content of C2C12 cells exposed to simvastatin and treated with IGF-1. In conclusion, impaired activity of AKT can be explained by reduced function of mTORC2 and of the IGF-1R/PI3K pathway. IGF-1 can prevent simvastatin-associated cytotoxicity and metabolic effects on C2C12 cells. The study gives insight into mechanisms of simvastatin-associated myotoxicity and provides potential targets for therapeutic intervention.








Similar content being viewed by others
References
Adams GR, McCue SA (1998) Localized infusion of IGF-I results in skeletal muscle hypertrophy in rats. J Appl Physiol (Bethesda, Md: 1985) 84(5):1716–1722
Alexia C, Bras M, Fallot G et al (2006) Pleiotropic effects of PI-3′ kinase/Akt signaling in human hepatoma cell proliferation and drug-induced apoptosis. Ann N Y Acad Sci 1090:1–17. doi:10.1196/annals.1378.001
Amarenco P, Bogousslavsky J, Callahan A 3rd et al (2006) High-dose atorvastatin after stroke or transient ischemic attack. N Engl J Med 355(6):549–559. doi:10.1056/NEJMoa061894
Bodine SC, Stitt TN, Gonzalez M et al (2001) Akt/mTOR pathway is a crucial regulator of skeletal muscle hypertrophy and can prevent muscle atrophy in vivo. Nat Cell Biol 3(11):1014–1019. doi:10.1038/ncb1101-1014
Bonifacio A, Sanvee GM, Bouitbir J, Krahenbuhl S (2015) The AKT/mTOR signaling pathway plays a key role in statin-induced myotoxicity. Biochim Biophys Acta 1853(8):1841–1849. doi:10.1016/j.bbamcr.2015.04.010
Bonifacio A, Mullen PJ, Mityko IS, Navegantes LC, Bouitbir J, Krahenbuhl S (2016) Simvastatin induces mitochondrial dysfunction and increased atrogin-1 expression in H9c2 cardiomyocytes and mice in vivo. Arch Toxicol 90(1):203–215. doi:10.1007/s00204-014-1378-4
Bouitbir J, Charles AL, Echaniz-Laguna A et al (2012) Opposite effects of statins on mitochondria of cardiac and skeletal muscles: a ‘mitohormesis’ mechanism involving reactive oxygen species and PGC-1. Eur Heart J 33(11):1397–1407. doi:10.1093/eurheartj/ehr224
Bouitbir J, Singh F, Charles AL et al (2016) Statins trigger mitochondrial reactive oxygen species-induced apoptosis in glycolytic skeletal muscle. Antioxid Redox Signal 24(2):84–98. doi:10.1089/ars.2014.6190
Fernandez M, Sanchez-Franco F, Palacios N, Sanchez I, Fernandez C, Cacicedo L (2004) IGF-I inhibits apoptosis through the activation of the phosphatidylinositol 3-kinase/Akt pathway in pituitary cells. J Mol Endocrinol 33(1):155–163. doi:10.1677/Jme.0.0330155
Garcia-Echeverria C, Pearson MA, Marti A et al (2004) In vivo antitumor activity of NVP-AEW541-A novel, potent, and selective inhibitor of the IGF-IR kinase. Cancer Cell 5(3):231–239
Glass DJ (2003) Signalling pathways that mediate skeletal muscle hypertrophy and atrophy. Nat Cell Biol 5(2):87–90. doi:10.1038/ncb0203-87
Hanai J, Cao P, Tanksale P et al (2007) The muscle-specific ubiquitin ligase atrogin-1/MAFbx mediates statin-induced muscle toxicity. J Clin Investig 117(12):3940–3951. doi:10.1172/jci32741
Huang K, Fingar DC (2014) Growing knowledge of the mTOR signaling network. Semin Cell Dev Biol 36:79–90. doi:10.1016/j.semcdb.2014.09.011
Joy TR, Hegele RA (2009) Narrative review: statin-related myopathy. Ann Intern Med 150(12):858–868
Kaufmann P, Torok M, Zahno A, Waldhauser KM, Brecht K, Krahenbuhl S (2006) Toxicity of statins on rat skeletal muscle mitochondria. Cell Mol Life Sci 63(19–20):2415–2425. doi:10.1007/S00018-006-6235-Z
Link E, Parish S, Armitage J et al (2008) SLCO1B1 variants and statin-induced myopathy–a genomewide study. N Engl J Med 359(8):789–799. doi:10.1056/NEJMoa0801936
Ma XM, Blenis J (2009) Molecular mechanisms of mTOR-mediated translational control. Nat Rev Mol Cell Biol 10(5):307–318. doi:10.1038/nrm2672
Mullen PJ, Luscher B, Scharnagl H, Krahenbuhl S, Brecht K (2010) Effect of simvastatin on cholesterol metabolism in C2C12 myotubes and HepG2 cells, and consequences for statin-induced myopathy. Biochem Pharmacol 79(8):1200–1209. doi:10.1016/j.bcp.2009.12.007
Mullen PJ, Zahno A, Lindinger P et al (2011) Susceptibility to simvastatin-induced toxicity is partly determined by mitochondrial respiration and phosphorylation state of Akt. Biochimica Et Biophysica Acta-Mol Cell Res 1813(12):2079–2087. doi:10.1016/J.Bbamcr.2011.07.019
Neuvonen PJ, Kantola T, Kivisto KT (1998) Simvastatin but not pravastatin is very susceptible to interaction with the CYP3A4 inhibitor itraconazole. Clin Pharmacol Ther 63(3):332–341. doi:10.1016/s0009-9236(98)90165-5
Nicholls SJ, Ballantyne CM, Barter PJ et al (2011) Effect of two intensive statin regimens on progression of coronary disease. N Engl J Med 365(22):2078–2087. doi:10.1056/NEJMoa1110874
Roten L, Schoenenberger RA, Krahenbuhl S, Schlienger RG (2004) Rhabdomyolysis in association with simvastatin and amiodarone. Ann Pharmacother 38(6):978–981. doi:10.1345/aph.1D498
Sacheck JM, Ohtsuka A, McLary SC, Goldberg AL (2004) IGF-I stimulates muscle growth by suppressing protein breakdown and expression of atrophy-related ubiquitin ligases, atrogin-1 and MuRF1. Am J Physiol Endocrinol Metab 287(4):E591–E601. doi:10.1152/ajpendo.00073.2004
Sandri M, Sandri C, Gilbert A et al (2004) Foxo transcription factors induce the atrophy-related ubiquitin ligase atrogin-1 and cause skeletal muscle atrophy. Cell 117(3):399–412
Sarbassov DD, Guertin DA, Ali SM, Sabatini DM (2005) Phosphorylation and regulation of Akt/PKB by the rictor-mTOR complex. Science (New York, NY) 307(5712):1098–1101. doi:10.1126/science.1106148
Schiaffino S, Mammucari C (2011) Regulation of skeletal muscle growth by the IGF1-Akt/PKB pathway: insights from genetic models. Skelet Muscle 1(1):4. doi:10.1186/2044-5040-1-4
Sugita H, Kaneki M, Sugita M, Yasukawa T, Yasuhara S, Martyn JA (2005) Burn injury impairs insulin-stimulated Akt/PKB activation in skeletal muscle. Am J Physiol Endocrinol Metab 288(3):E585–E591. doi:10.1152/ajpendo.00321.2004
Svanberg E, Frost RA, Lang CH et al (2000) IGF-I/IGFBP-3 binary complex modulates sepsis-induced inhibition of protein synthesis in skeletal muscle. Am J Physiol Endocrinol Metab 279(5):E1145–E1158
Wang RH, Kim HS, Xiao C, Xu X, Gavrilova O, Deng CX (2011) Hepatic Sirt1 deficiency in mice impairs mTorc2/Akt signaling and results in hyperglycemia, oxidative damage, and insulin resistance. J Clin Investig 121(11):4477–4490. doi:10.1172/JCI46243
Zheng B, Ohkawa S, Li H, Roberts-Wilson TK, Price SR (2010) FOXO3a mediates signaling crosstalk that coordinates ubiquitin and atrogin-1/MAFbx expression during glucocorticoid-induced skeletal muscle atrophy. FASEB J: Off Publ Fed Am Soc Exp Biol 24(8):2660–2669. doi:10.1096/fj.09-151480
Zhou H, Li XM, Meinkoth J, Pittman RN (2000) Akt regulates cell survival and apoptosis at a postmitochondrial level. J Cell Biol 151(3):483–494
Acknowledgments
The authors thank Dr Giulia Minetti for the aid with the measurements of myotube diameters.
Funding
SK was supported by a grant from the Swiss National Science Foundation (31003A_156270).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Bonifacio, A., Sanvee, G.M., Brecht, K. et al. IGF-1 prevents simvastatin-induced myotoxicity in C2C12 myotubes. Arch Toxicol 91, 2223–2234 (2017). https://doi.org/10.1007/s00204-016-1871-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-016-1871-z