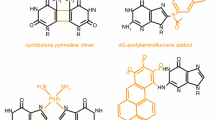
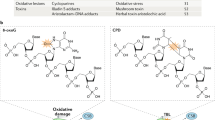
Abstract
Nucleotide excision repair (NER) is a versatile pathway that removes helix-distorting DNA lesions from the genomes of organisms across the evolutionary scale, from bacteria to humans. The serial steps in NER involve recognition of lesions, adducts or structures that disrupt the DNA double helix, removal of a short oligonucleotide containing the offending lesion, synthesis of a repair patch copying the opposite undamaged strand, and ligation, to restore the DNA to its original form. Transcription-coupled repair (TCR) is a subpathway of NER dedicated to the repair of lesions that, by virtue of their location on the transcribed strands of active genes, encumber elongation by RNA polymerases. In this review, I report on recent findings that contribute to the elucidation of TCR mechanisms in the bacterium Escherichia coli, the yeast Saccharomyces cerevisiae and human cells. I review general models for the biochemical pathways and how and when cells might choose to utilize TCR or other pathways for repair or bypass of transcription-blocking DNA alterations.



Similar content being viewed by others
References
Banerjee D, Mandal SM, Das A et al (2010) Preferential repair of oxidized base damage in the transcribed genes of mammalian cells. J Biol Chem 286(8):6006–6016. doi:10.1074/jbc.M110.198796
Beerens N, Hoeijmakers JH, Kanaar R, Vermeulen W, Wyman C (2005) The CSB protein actively wraps DNA. J Biol Chem 280(6):4722–4729. doi:10.1074/jbc.M409147200
Bhatia PK, Verhage RA, Brouwer J, Friedberg EC (1996) Molecular cloning and characterization of Saccharomyces cerevisiae RAD28, the yeast homolog of the human Cockayne syndrome A (CSA) gene. J Bacteriol 178(20):5977–5988
Bregeon D, Doddridge ZA, You HJ, Weiss B, Doetsch PW (2003) Transcriptional mutagenesis induced by uracil and 8-oxoguanine in Escherichia coli. Mol Cell 12(4):959–970
Brooks PJ (2013) Blinded by the UV light: how the focus on transcription-coupled NER has distracted from understanding the mechanisms of Cockayne syndrome neurologic disease. DNA Repair 12(8):656–671. doi:10.1016/j.dnarep.2013.04.018
Butland G, Peregrin-Alvarez JM, Li J et al (2005) Interaction network containing conserved and essential protein complexes in Escherichia coli. Nature 433(7025):531–537. doi:10.1038/nature03239:
Charlet-Berguerand N, Feuerhahn S, Kong SE et al (2006) RNA polymerase II bypass of oxidative DNA damage is regulated by transcription elongation factors. EMBO J 25(23):5481–5491. doi:10.1038/sj.emboj.7601403
Cleaver JE (1968) Defective repair replication of DNA in xeroderma pigmentosum. Nature 218(5142):652–656
Cohen SE, Walker GC (2011) New discoveries linking transcription to DNA repair and damage tolerance pathways. Transcription 2(1):37–40. doi:10.4161/trns.2.1.14228
Cohen SE, Lewis CA, Mooney RA et al (2010) Roles for the transcription elongation factor NusA in both DNA repair and damage tolerance pathways in Escherichia coli. Proc Natl Acad Sci USA 107(35):15517–15522. doi:10.1073/pnas.1005203107
Crowley DJ, Hanawalt PC (2001) The SOS-dependent upregulation of uvrD is not required for efficient nucleotide excision repair of ultraviolet light induced DNA photoproducts in Escherichia coli. Mutat Res 485(4):319–329. doi:10.1016/j.dnarep.2016.03.007
Czaja W, Mao P, Smerdon MJ (2012) The emerging roles of ATP-dependent chromatin remodeling enzymes in nucleotide excision repair. Int J Mol Sci 13(9):11954–11973. doi:10.3390/ijms130911954:
Deaconescu AM, Chambers AL, Smith AJ et al (2006) Structural basis for bacterial transcription-coupled DNA repair. Cell 124(3):507–520. doi:10.1016/j.cell.2005.11.045
Dinant C, Ampatziadis-Michailidis G, Lans H et al (2013) Enhanced chromatin dynamics by FACT promotes transcriptional restart after UV-induced DNA damage. Mol Cell 51(4):469–479. doi:10.1016/j.molcel.2013.08.007
Epshtein V, Kamarthapu V, McGary K et al (2014) UvrD facilitates DNA repair by pulling RNA polymerase backwards. Nature 505(7483):372–377. doi:10.1038/nature12928
Fagbemi AF, Orelli B, Scharer OD (2011) Regulation of endonuclease activity in human nucleotide excision repair. DNA Repair 10(7):722–729. doi:10.1016/j.dnarep.2011.04.022
Fei J, Chen J (2012) KIAA1530 protein is recruited by Cockayne syndrome complementation group protein A (CSA) to participate in transcription-coupled repair (TCR). J Biol Chem 287(42):35118–35126. doi:10.1074/jbc.M112.398131
Fousteri M, Vermeulen W, van Zeeland AA, Mullenders LH (2006) Cockayne syndrome A and B proteins differentially regulate recruitment of chromatin remodeling and repair factors to stalled RNA polymerase II in vivo. Mol Cell 23(4):471–482. doi:10.1016/j.molcel.2006.06.029
Friedberg EC, Elledge SJ, Lehmann AR, Lindahl T, Muzi-Falconi M (eds) (2014) DNA repair, mutagenesis, and other responses to DNA damage. Cold Spring Harbor Laboratory Press, New York
Gaillard H, Aguilera A (2013) Transcription coupled repair at the interface between transcription elongation and mRNP biogenesis. Biochim Biophys Acta 1829(1):141–150. doi:10.1016/j.bbagrm.2012.09.008
Gaillard H, Wellinger RE, Aguilera A (2007) A new connection of mRNP biogenesis and export with transcription-coupled repair. Nucleic Acids Res 35(12):3893–3906. doi:10.1093/nar/gkm373
Ganesan A, Hanawalt P (2016) Photobiological origins of the field of genomic maintenance. Photochem Photobiol 92(1):52–60. doi:10.1111/php.12542
Ganesan A, Spivak G, Hanawalt PC (2012) Transcription-coupled DNA repair in prokaryotes. Prog Mol Biol Transl Sci 110:25–40. doi:10.1016/B978-0-12-387665-2.00002-X
Gao S, Drouin R, Holmquist GP (1994) DNA repair rates mapped along the human PGK1 gene at nucleotide resolution. Science 263(5152):1438–1440
Guo J, Hanawalt PC, Spivak G (2013) Comet-FISH with strand-specific probes reveals transcription-coupled repair of 8-oxoGuanine in human cells. Nucleic Acids Res 41(16):7700–7712. doi:10.1093/nar/gkt524
Hanawalt PC, Spivak G (2008) Transcription-coupled DNA repair: two decades of progress and surprises. Nat Rev Mol Cell Biol 9(12):958–970. doi:10.1038/nrm2549
Harreman M, Taschner M, Sigurdsson S et al (2009) Distinct ubiquitin ligases act sequentially for RNA polymerase II polyubiquitylation. Proc Natl Acad Sci USA 106(49):20705–20710. doi:10.1073/pnas.0907052106
Howan K, Smith AJ, Westblade LF et al (2012) Initiation of transcription-coupled repair characterized at single-molecule resolution. Nature 490(7420):431–434. doi:10.1038/nature11430
Iyama T, Wilson DM 3rd (2013) DNA repair mechanisms in dividing and non-dividing cells. DNA Repair 12(8):620–636. doi:10.1016/j.dnarep.2013.04.015
Iyama T, Wilson DM 3rd (2016) Elements that regulate the DNA damage response of proteins defective in Cockayne syndrome. J Mol Biol 428(1):62–78. doi:10.1016/j.jmb.2015.11.020
Johnson RE, Prakash S, Prakash L (1999) Efficient bypass of a thymine-thymine dimer by yeast DNA polymerase, Poleta. Science 283(5404):1001–1004
Kad NM, Van Houten B (2012) Dynamics of lesion processing by bacterial nucleotide excision repair proteins. Prog Mol Biol Transl Sci 110:1–24. doi:10.1016/B978-0-12-387665-2.00001-8
Kamarthapu V, Nudler E (2015) Rethinking transcription coupled DNA repair. Curr Opin Microbiol 24:15–20. doi:10.1016/j.mib.2014.12.005
Kirkali G, de Souza-Pinto NC, Jaruga P, Bohr VA, Dizdaroglu M (2009) Accumulation of (5′S)-8,5′-cyclo-2′-deoxyadenosine in organs of Cockayne syndrome complementation group B gene knockout mice. DNA Repair 8(2):274–278. doi:10.1016/j.dnarep.2008.09.009
Kisker C, Kuper J, Van Houten B (2013) Prokaryotic nucleotide excision repair. Cold Spring Harb Perspect Biol 5(3):a012591. doi:10.1101/cshperspect.a012591
Koeppel F, Poindessous V, Lazar V, Raymond E, Sarasin A, Larsen AK (2004) Irofulven cytotoxicity depends on transcription-coupled nucleotide excision repair and is correlated with XPG expression in solid tumor cells. Clin Cancer Res 10(16):5604–5613. doi:10.1158/1078-0432.CCR-04-0442
Kuraoka I, Ito S, Wada T et al (2008) Isolation of XAB2 complex involved in pre-mRNA splicing, transcription, and transcription-coupled repair. J Biol Chem 283(2):940–950. doi:10.1074/jbc.M706647200
Lans H, Marteijn JA, Vermeulen W (2012) ATP-dependent chromatin remodeling in the DNA-damage response. Epigenetics Chromatin 5:4. doi:10.1186/1756-8935-5-4
Lee YC, Cai Y, Mu H et al (2014) The relationships between XPC binding to conformationally diverse DNA adducts and their excision by the human NER system: is there a correlation? DNA Repair 19:55–63. doi:10.1016/j.dnarep.2014.03.026
Leela JK, Syeda AH, Anupama K, Gowrishankar J (2013) Rho-dependent transcription termination is essential to prevent excessive genome-wide R-loops in Escherichia coli. Proc Natl Acad Sci USA 110(1):258–263. doi:10.1073/pnas.1213123110
Lehmann AR (2011) DNA polymerases and repair synthesis in NER in human cells. DNA Repair 10(7):730–733. doi:10.1016/j.dnarep.2011.04.023
Li S, Smerdon MJ (2002) Rpb4 and Rpb9 mediate subpathways of transcription-coupled DNA repair in Saccharomyces cerevisiae. EMBO J 21(21):5921–5929
Li B, Carey M, Workman JL (2007) The role of chromatin during transcription. Cell 128(4):707–719. doi:10.1016/j.cell.2007.01.015
Li W, Selvam K, Rahman SA, Li S (2016) Sen1, the yeast homolog of human senataxin, plays a more direct role than Rad26 in transcription coupled DNA repair. Nucleic Acids Res. doi:10.1093/nar/gkw428
Manelyte L, Kim YI, Smith AJ, Smith RM, Savery NJ (2010) Regulation and rate enhancement during transcription-coupled DNA repair. Mol Cell 40(5):714–724. doi:10.1016/j.molcel.2010.11.012:
Martin HA, Pedraza-Reyes M, Yasbin RE, Robleto EA (2011) Transcriptional de-repression and Mfd are mutagenic in stressed Bacillus subtilis cells. J Mol Microbiol Biotechnol 21(1–2):45–58. doi:10.1159/000332751
Mathieu N, Kaczmarek N, Ruthemann P, Luch A, Naegeli H (2013) DNA quality control by a lesion sensor pocket of the xeroderma pigmentosum group D helicase subunit of TFIIH. Curr Biol 23(3):204–212. doi:10.1016/j.cub.2012.12.032
McKibbin PL, Fleming AM, Towheed MA, Van Houten B, Burrows CJ, David SS (2013) Repair of hydantoin lesions and their amine adducts in DNA by base and nucleotide excision repair. J Am Chem Soc 135(37):13851–13861. doi:10.1021/ja4059469
Mellon I, Champe GN (1996) Products of DNA mismatch repair genes mutS and mutL are required for transcription-coupled nucleotide-excision repair of the lactose operon in Escherichia coli. Proc Natl Acad Sci USA 93(3):1292–1297
Mellon I, Spivak G, Hanawalt PC (1987) Selective removal of transcription-blocking DNA damage from the transcribed strand of the mammalian DHFR gene. Cell 51(2):241–249
Menoni H, Gasparutto D, Hamiche A et al (2007) ATP-dependent chromatin remodeling is required for base excision repair in conventional but not in variant H2A.Bbd nucleosomes. Mol Cell Biol 27(17):5949–5956. doi:10.1128/MCB.00376-07
Menoni H, Hoeijmakers JH, Vermeulen W (2012) Nucleotide excision repair-initiating proteins bind to oxidative DNA lesions in vivo. J Cell Biol 199(7):1037–1046. doi:10.1083/jcb.201205149
Mooney RA, Davis SE, Peters JM, Rowland JL, Ansari AZ, Landick R (2009) Regulator trafficking on bacterial transcription units in vivo. Mol Cell 33(1):97–108. doi:10.1016/j.molcel.2008.12.021
Mullenders L (2015) DNA damage mediated transcription arrest: step back to go forward. DNA Repair 36:28–35. doi:10.1016/j.dnarep.2015.09.005
Nakatsu Y, Asahina H, Citterio E et al (2000) XAB2, a novel tetratricopeptide repeat protein involved in transcription-coupled DNA repair and transcription. J Biol Chem 275(45):34931–34937. doi:10.1074/jbc.M004936200
Nakazawa Y, Sasaki K, Mitsutake N et al (2012) Mutations in UVSSA cause UV-sensitive syndrome and impair RNA polymerase IIo processing in transcription-coupled nucleotide-excision repair. Nat Genet 44(5):586–592. doi:10.1038/ng.2229
Nouspikel TP, Hyka-Nouspikel N, Hanawalt PC (2006) Transcription domain-associated repair in human cells. Mol Cell Biol 26(23):8722–8730. doi:10.1128/MCB.01263-06
Ogi T, Limsirichaikul S, Overmeer RM et al (2010) Three DNA polymerases, recruited by different mechanisms, carry out NER repair synthesis in human cells. Mol Cell 37(5):714–727. doi:10.1016/j.molcel.2010.02.009
Pettijohn D, Hanawalt P (1964) Evidence for repair-replication of ultraviolet damaged DNA in bacteria. J Mol Biol 9:395–410
Prakash S, Prakash L (2000) Nucleotide excision repair in yeast. Mutat Res 451(1–2):13–24
Reagan MS, Friedberg EC (1997) Recovery of RNA polymerase II synthesis following DNA damage in mutants of Saccharomyces cerevisiae defective in nucleotide excision repair. Nucleic Acids Res 25(21):4257–4263
Reed SH (2005) Nucleotide excision repair in chromatin: the shape of things to come. DNA Repair 4(8):909–918. doi:10.1016/j.dnarep.2005.04.009
Reis AM, Mills WK, Ramachandran I, Friedberg EC, Thompson D, Queimado L (2012) Targeted detection of in vivo endogenous DNA base damage reveals preferential base excision repair in the transcribed strand. Nucleic Acids Res 40(1):206–219. doi:10.1093/nar/gkr704
Ross C, Pybus C, Pedraza-Reyes M, Sung HM, Yasbin RE, Robleto E (2006) Novel role of mfd: effects on stationary-phase mutagenesis in Bacillus subtilis. J Bacteriol 188(21):7512–7520. doi:10.1128/JB.00980-06
Sarker A, Tsutakawa S, Kostek S et al (2005) Recognition of RNA polymerase II and transcription bubbles by XPG, CSB, and TFIIH insights for transcription-coupled repair and Cockayne syndrome. Mol Cell 20:187–198
Saxowsky TT, Meadows KL, Klungland A, Doetsch PW (2008) 8-Oxoguanine-mediated transcriptional mutagenesis causes Ras activation in mammalian cells. Proc Natl Acad Sci USA 105(48):18877–18882. doi:10.1073/pnas.0806464105
Schalow BJ, Courcelle CT, Courcelle J (2012) Mfd is required for rapid recovery of transcription following UV-induced DNA damage but not oxidative DNA damage in Escherichia coli. J Bacteriol 194(10):2637–2645. doi:10.1128/JB.06725-11
Scharer OD (2013) Nucleotide excision repair in eukaryotes. Cold Spring Harb Perspect Biol 5(10):a012609. doi:10.1101/cshperspect.a012609
Schwertman P, Lagarou A, Dekkers DH et al (2012) UV-sensitive syndrome protein UVSSA recruits USP7 to regulate transcription-coupled repair. Nat Genet 44(5):598–602. doi:10.1038/ng.2230
Schwertman P, Vermeulen W, Marteijn JA (2013) UVSSA and USP7, a new couple in transcription-coupled DNA repair. Chromosoma 122(4):275–284. doi:10.1007/s00412-013-0420-2
Selby CP, Sancar A (1993) Molecular mechanism of transcription-repair coupling. Science 260(5104):53–58
Selby CP, Sancar A (1997) Cockayne syndrome group B protein enhances elongation by RNA polymerase II. Proc Natl Acad Sci USA 94(21):11205–11209
Selby CP, Witkin EM, Sancar A (1991) Escherichia coli mfd mutant deficient in “mutation frequency decline” lacks strand-specific repair: in vitro complementation with purified coupling factor. Proc Natl Acad Sci USA 88(24):11574–11578
Setlow RB, Carrier WL (1964) The disappearance of thymine dimers from DNA: an error-correcting mechanism. Proc Natl Acad Sci USA 51:226–231
Sidorenko VS, Yeo JE, Bonala RR, Johnson F, Scharer OD, Grollman AP (2012) Lack of recognition by global-genome nucleotide excision repair accounts for the high mutagenicity and persistence of aristolactam-DNA adducts. Nucleic Acids Res 40(6):2494–2505. doi:10.1093/nar/gkr1095
Sin Y, Tanaka K, Saijo M (2016) The C-terminal region and SUMOylation of cockayne syndrome group B protein play critical roles in transcription-coupled nucleotide excision repair. J Biol Chem 291(3):1387–1397. doi:10.1074/jbc.M115.683235
Singh A, Compe E, Le May N, Egly JM (2015) TFIIH subunit alterations causing xeroderma pigmentosum and trichothiodystrophy specifically disturb several steps during transcription. Am J Hum Genet 96(2):194–207. doi:10.1016/j.ajhg.2014.12.012
Soria G, Polo SE, Almouzni G (2012) Prime, repair, restore: the active role of chromatin in the DNA damage response. Mol Cell 46(6):722–734. doi:10.1016/j.molcel.2012.06.002
Spivak G (2015) Nucleotide excision repair in humans. DNA Repair 36:13–18. doi:10.1016/j.dnarep.2015.09.003
Spivak G, Ganesan AK (2014) The complex choreography of transcription-coupled repair. DNA Repair 19:64–70. doi:10.1016/j.dnarep.2014.03.025
Spivak G, Hanawalt PC (2006) Host cell reactivation of plasmids containing oxidative DNA lesions is defective in Cockayne syndrome but normal in UV-sensitive syndrome fibroblasts. DNA Repair 5(1):13–22. doi:10.1016/j.dnarep.2005.06.017
Spivak G, Hanawalt PC (2015) Photosensitive human syndromes. Mutat Res Fundam Mol Mech Mutagen 776:24–30
Stantial N, Dumpe J, Pietrosimone K, Baltazar F, Crowley DJ (2016) Transcription-coupled repair of UV damage in the halophilic archaea. DNA Repair 41:63–68. doi:10.1016/j.dnarep.2016.03.007
Tatum D, Li S (2011) Nucleotide Excision repair in S. cerevisiae. In: Storici F (ed) DNA repair—on the pathway to fixing DNA damage and errors. InTech. www.intechopen.com/books/dna-repair-on-the-pathways-to-fixing-dna-damage-and-errors/nucleotide-excision-repair-in-s-cerevisiae
Tornaletti S, Reines D, Hanawalt PC (1999) Structural characterization of RNA polymerase II complexes arrested by a cyclobutane pyrimidine dimer in the transcribed strand of template DNA. J Biol Chem 274(34):24124–24130
van Gool AJ, Verhage R, Swagemakers SM et al (1994) RAD26, the functional S. cerevisiae homolog of the Cockayne syndrome B gene ERCC6. EMBO J 13(22):5361–5369
Verhage RA, Heyn J, van de Putte P, Brouwer J (1997) Transcription elongation factor S-ii is not required for transcription-coupled repair in yeast. Mol Gen Genet 254(3):284–290
Vermeulen W, Fousteri M (2013) Mammalian transcription-coupled excision repair. Cold Spring Harbor Perspect Biol. doi:10.1101/cshperspect.a012625
Wei L, Lan L, Yasui A et al (2011) BRCA1 contributes to transcription-coupled repair of DNA damage through polyubiquitination and degradation of Cockayne syndrome B protein. Cancer Sci 102(10):1840–1847. doi:10.1111/j.1349-7006.2011.02037.x
Wimberly H, Shee C, Thornton PC, Sivaramakrishnan P, Rosenberg SM, Hastings PJ (2013) R-loops and nicks initiate DNA breakage and genome instability in non-growing Escherichia coli. Nat Commun 4:2115. doi:10.1038/ncomms3115
Zhang X, Horibata K, Saijo M et al (2012) Mutations in UVSSA cause UV-sensitive syndrome and destabilize ERCC6 in transcription-coupled DNA repair. Nat Genet 44(5):593–597. doi:10.1038/ng.2228
Acknowledgments
Supported by National Institute of Environmental Health Sciences grant 5RO1 CA077712. We apologize to those whose work has not been cited.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Spivak, G. Transcription-coupled repair: an update. Arch Toxicol 90, 2583–2594 (2016). https://doi.org/10.1007/s00204-016-1820-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-016-1820-x