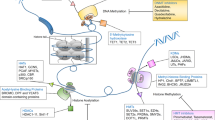
Abstract
Alteration of the chromatin orchestra seems to play a critical role in cancer. In recent years, in-depth studies of epigenetic machinery and its deregulation have led to the development and use of a wide range of modulatory molecules directed not only at chromatin enzymes (histone acetyltransferases, histone deacetylases, histone methyltransferases, histone demethylases and DNA methyltransferases) but also toward the emerging class of chromatin-associated proteins, so-called “histone readers.” Chromatin modifiers are attractive therapeutic targets for the development of new cancer therapies. Many are currently approved by the US Food and Drug Administration and used to treat different malignancies. Specifically, inhibitors of DNA methyltransferases, such as azacitidine and decitabine, have been approved for the treatment of myelodysplastic syndrome, while inhibitors of histone deacetylases, including vorinostat and romidepsin, have been approved for cutaneous T-cell lymphoma. The bromodomain and extra-terminal inhibitors JQ1, IBET762 and IBET151 have performed extremely well in preclinical settings, suggesting that they may be promising molecules for the treatment of some type of tumors. This review focuses on epidrugs and their possible application, with particular emphasis on their mechanism of action as well as their present status in clinical and preclinical trials.
Similar content being viewed by others
References
Allis CD, Berger SL, Cote J et al (2007) New nomenclature for chromatin-modifying enzymes. Cell 131(4):633–636. doi:10.1016/j.cell.2007.10.039
Amato RJ, Stephenson J, Hotte S et al (2012) MG98, a second-generation DNMT1 inhibitor, in the treatment of advanced renal cell carcinoma. Cancer Invest 30(5):415–421. doi:10.3109/07357907.2012.675381
Amiri-Kordestani L, Luchenko V, Peer CJ et al (2013) Phase I trial of a new schedule of romidepsin in patients with advanced cancers. Clin Cancer Res 19(16):4499–4507. doi:10.1158/1078-0432.CCR-13-0095
Andreadi C, Britton RG, Patel KR, Brown K (2014) Resveratrol-sulfates provide an intracellular reservoir for generation of parent resveratrol, which induces autophagy in cancer cells. Autophagy 10(3):524–525. doi:10.4161/auto.27593
Armas-Pineda C, Arenas-Huertero F, Perezpenia-Diazconti M et al (2007) Expression of PCAF, p300 and Gcn5 and more highly acetylated histone H4 in pediatric tumors. J Exp Clin Cancer Res 26(2):269–276
Audrito V, Vaisitti T, Rossi D et al (2011) Nicotinamide blocks proliferation and induces apoptosis of chronic lymphocytic leukemia cells through activation of the p53/miR-34a/SIRT1 tumor suppressor network. Cancer Res 71(13):4473–4483. doi:10.1158/0008-5472.CAN-10-4452
Balasubramanyam K, Swaminathan V, Ranganathan A, Kundu TK (2003) Small molecule modulators of histone acetyltransferase p300. J Biol Chem 278(21):19134–19140. doi:10.1074/jbc.M301580200
Balasubramanyam K, Varier RA, Altaf M et al (2004) Curcumin, a novel p300/CREB-binding protein-specific inhibitor of acetyltransferase, represses the acetylation of histone/nonhistone proteins and histone acetyltransferase-dependent chromatin transcription. J Biol Chem 279(49):51163–51171. doi:10.1074/jbc.M409024200
Bannister AJ, Kouzarides T (2011) Regulation of chromatin by histone modifications. Cell Res 21(3):381–395. doi:10.1038/cr.2011.22
Bannister AJ, Zegerman P, Partridge JF et al (2001) Selective recognition of methylated lysine 9 on histone H3 by the HP1 chromo domain. Nature 410(6824):120–124. doi:10.1038/35065138
Barber MF, Michishita-Kioi E, Xi Y et al (2012) SIRT7 links H3K18 deacetylation to maintenance of oncogenic transformation. Nature 487(7405):114–118. doi:10.1038/nature11043
Baud MG, Haus P, Leiser T, Meyer-Almes FJ, Fuchter MJ (2013) Highly ligand efficient and selective N-2-(Thioethyl)picolinamide histone deacetylase inhibitors inspired by the natural product psammaplin A. ChemMedChem 8(1):149–156. doi:10.1002/cmdc.201200450
Beaulieu N, Morin S, Chute IC, Robert MF, Nguyen H, MacLeod AR (2002) An essential role for DNA methyltransferase DNMT3B in cancer cell survival. J Biol Chem 277(31):28176–28181. doi:10.1074/jbc.M204734200
Bedalov A, Gatbonton T, Irvine WP, Gottschling DE, Simon JA (2001) Identification of a small molecule inhibitor of Sir2p. Proc Natl Acad Sci USA 98(26):15113–15118. doi:10.1073/pnas.261574398
Belinsky SA, Grimes MJ, Picchi MA et al (2011) Combination therapy with vidaza and entinostat suppresses tumor growth and reprograms the epigenome in an orthotopic lung cancer model. Cancer Res 71(2):454–462. doi:10.1158/0008-5472.CAN-10-3184
Benedetti R, Conte M, Altucci L (2014) Targeting histone deacetylases in diseases: Where are we? Antioxid Redox Signal. doi:10.1089/ars.2013.5776
Biel M, Kretsovali A, Karatzali E, Papamatheakis J, Giannis A (2004) Design, synthesis, and biological evaluation of a small-molecule inhibitor of the histone acetyltransferase Gcn5. Angew Chem 43(30):3974–3976. doi:10.1002/anie.200453879
Bisht K, Wagner KH, Bulmer AC (2010) Curcumin, resveratrol and flavonoids as anti-inflammatory, cyto- and DNA-protective dietary compounds. Toxicology 278(1):88–100. doi:10.1016/j.tox.2009.11.008
Bojang P Jr, Ramos KS (2014) The promise and failures of epigenetic therapies for cancer treatment. Cancer Treat Rev 40(1):153–169. doi:10.1016/j.ctrv.2013.05.009
Bose P, Dai Y, Grant S (2014) Histone deacetylase inhibitor (HDACI) mechanisms of action: emerging insights. Pharmacol Ther. doi:10.1016/j.pharmthera.2014.04.004
Bouchard J, Momparler RL (1983) Incorporation of 5-Aza-2′-deoxycytidine-5′-triphosphate into DNA. Interactions with mammalian DNA polymerase alpha and DNA methylase. Mol Pharmacol 24(1):109–114
Bowers EM, Yan G, Mukherjee C et al (2010) Virtual ligand screening of the p300/CBP histone acetyltransferase: identification of a selective small molecule inhibitor. Chem Biol 17(5):471–482. doi:10.1016/j.chembiol.2010.03.006
Brueckner B, Garcia Boy R, Siedlecki P et al (2005) Epigenetic reactivation of tumor suppressor genes by a novel small-molecule inhibitor of human DNA methyltransferases. Cancer Res 65(14):6305–6311. doi:10.1158/0008-5472.CAN-04-2957
Bruzzone S, Parenti MD, Grozio A et al (2013) Rejuvenating sirtuins: the rise of a new family of cancer drug targets. Curr Pharm Des 19(4):614–623
Cameron EE, Bachman KE, Myohanen S, Herman JG, Baylin SB (1999) Synergy of demethylation and histone deacetylase inhibition in the re-expression of genes silenced in cancer. Nat Genet 21(1):103–107. doi:10.1038/5047
Canani RB, Costanzo MD, Leone L, Pedata M, Meli R, Calignano A (2011) Potential beneficial effects of butyrate in intestinal and extraintestinal diseases. World J Gastroenterol 17(12):1519–1528. doi:10.3748/wjg.v17.i12.1519
Candelaria M, Gallardo-Rincon D, Arce C et al (2007) A phase II study of epigenetic therapy with hydralazine and magnesium valproate to overcome chemotherapy resistance in refractory solid tumors. Ann Oncol 18(9):1529–1538. doi:10.1093/annonc/mdm204
Cea M, Soncini D, Fruscione F et al (2011) Synergistic interactions between HDAC and sirtuin inhibitors in human leukemia cells. PLoS ONE 6(7):e22739. doi:10.1371/journal.pone.0022739
Cen Y (2010) Sirtuins inhibitors: the approach to affinity and selectivity. Biochim Biophys Acta 1804(8):1635–1644. doi:10.1016/j.bbapap.2009.11.010
Chaidos A, Caputo V, Gouvedenou K et al (2014) Potent antimyeloma activity of the novel bromodomain inhibitors I-BET151 and I-BET762. Blood 123(5):697–705. doi:10.1182/blood-2013-01-478420
Challen GA, Sun D, Jeong M et al (2012) Dnmt3a is essential for hematopoietic stem cell differentiation. Nat Genet 44(1):23–31. doi:10.1038/ng.1009
Chang Y, Zhang X, Horton JR et al (2009) Structural basis for G9a-like protein lysine methyltransferase inhibition by BIX-01294. Nat Struct Mol Biol 16(3):312–317. doi:10.1038/nsmb.1560
Chen M, Shabashvili D, Nawab A et al (2012) DNA methyltransferase inhibitor, zebularine, delays tumor growth and induces apoptosis in a genetically engineered mouse model of breast cancer. Mol Cancer Ther 11(2):370–382. doi:10.1158/1535-7163.MCT-11-0458
Cheng JC, Matsen CB, Gonzales FA et al (2003) Inhibition of DNA methylation and reactivation of silenced genes by zebularine. J Natl Cancer Inst 95(5):399–409
Cherblanc FL, Chapman KL, Brown R, Fuchter MJ (2013) Chaetocin is a nonspecific inhibitor of histone lysine methyltransferases. Nat Chem Biol 9(3):136–137. doi:10.1038/nchembio.1187
Chia K, Beamish H, Jafferi K, Gabrielli B (2010) The histone deacetylase inhibitor MGCD0103 has both deacetylase and microtubule inhibitory activity. Mol Pharmacol 78(3):436–443. doi:10.1124/mol.110.065169
Coffey K, Blackburn TJ, Cook S et al (2012) Characterisation of a Tip60 specific inhibitor, NU9056, in prostate cancer. PLoS ONE 7(10):e45539. doi:10.1371/journal.pone.0045539
Collins HM, Abdelghany MK, Messmer M et al (2013) Differential effects of garcinol and curcumin on histone and p53 modifications in tumour cells. BMC Cancer 13:37. doi:10.1186/1471-2407-13-37
Conte M, Altucci L (2012) Molecular pathways: the complexity of the epigenome in cancer and recent clinical advances. Clin Cancer Res 18(20):5526–5534. doi:10.1158/1078-0432.CCR-12-2037
Daigle SR, Olhava EJ, Therkelsen CA et al (2011) Selective killing of mixed lineage leukemia cells by a potent small-molecule DOT1L inhibitor. Cancer Cell 20(1):53–65. doi:10.1016/j.ccr.2011.06.009
Daigle SR, Olhava EJ, Therkelsen CA et al (2013) Potent inhibition of DOT1L as treatment of MLL-fusion leukemia. Blood 122(6):1017–1025. doi:10.1182/blood-2013-04-497644
Dalgliesh GL, Furge K, Greenman C et al (2010) Systematic sequencing of renal carcinoma reveals inactivation of histone modifying genes. Nature 463(7279):360–363. doi:10.1038/nature08672
Davis AJ, Gelmon KA, Siu LL et al (2003) Phase I and pharmacologic study of the human DNA methyltransferase antisense oligodeoxynucleotide MG98 given as a 21-day continuous infusion every 4 weeks. Invest New Drugs 21(1):85–97
Dawson MA, Prinjha RK, Dittmann A et al (2011) Inhibition of BET recruitment to chromatin as an effective treatment for MLL-fusion leukaemia. Nature 478(7370):529–533. doi:10.1038/nature10509
Dawson MA, Kouzarides T, Huntly BJ (2012) Targeting epigenetic readers in cancer. N Engl J Med 367(7):647–657. doi:10.1056/NEJMra1112635
Dawson MA, Gudgin EJ, Horton SJ et al (2014) Recurrent mutations, including NPM1c, activate a BRD4-dependent core transcriptional program in acute myeloid leukemia. Leukemia 28(2):311–320. doi:10.1038/leu.2013.338
Delmore JE, Issa GC, Lemieux ME et al (2011) BET bromodomain inhibition as a therapeutic strategy to target c-Myc. Cell 146(6):904–917. doi:10.1016/j.cell.2011.08.017
Do GM, Kwon EY, Kim HJ et al (2008) Long-term effects of resveratrol supplementation on suppression of atherogenic lesion formation and cholesterol synthesis in apo E-deficient mice. Biochem Biophys Res Commun 374(1):55–59. doi:10.1016/j.bbrc.2008.06.113
Duvic M, Vu J (2007) Vorinostat in cutaneous T-cell lymphoma. Drugs Today 43(9):585–599. doi:10.1358/dot.2007.43.9.1112980
El-Khoury V, Pierson S, Szwarcbart E et al (2014) Disruption of autophagy by the histone deacetylase inhibitor MGCD0103 and its therapeutic implication in B-cell chronic lymphocytic leukemia. Leukemia. doi:10.1038/leu.2014.19
Ellis L, Pan Y, Smyth GK et al (2008) Histone deacetylase inhibitor panobinostat induces clinical responses with associated alterations in gene expression profiles in cutaneous T-cell lymphoma. Clin Cancer Res 14(14):4500–4510. doi:10.1158/1078-0432.CCR-07-4262
Erwin BG, Persson L, Pegg AE (1984) Differential inhibition of histone and polyamine acetylases by multisubstrate analogues. Biochemistry 23(18):4250–4255
Esteller M (2007) Cancer epigenomics: DNA methylomes and histone-modification maps. Nat Rev Genet 8(4):286–298. doi:10.1038/nrg2005
Esteller M (2008) Epigenetics in cancer. N Engl J Med 358(11):1148–1159. doi:10.1056/NEJMra072067
Fang XM, Sun LF, Peng JP, Dong Q, Zheng S (2003) The study of 5-Aza-2′-deoxycytidine on transcription regulation of p16/CDKN2 gene demethylation in RKO human colorectal cell line. Zhonghua yi xue za zhi 83(23):2077–2082
Filippakopoulos P, Qi J, Picaud S et al (2010) Selective inhibition of BET bromodomains. Nature 468(7327):1067–1073. doi:10.1038/nature09504
Finkel T, Deng CX, Mostoslavsky R (2009) Recent progress in the biology and physiology of sirtuins. Nature 460(7255):587–591. doi:10.1038/nature08197
Forneris F, Binda C, Vanoni MA, Mattevi A, Battaglioli E (2005) Histone demethylation catalysed by LSD1 is a flavin-dependent oxidative process. FEBS Lett 579(10):2203–2207. doi:10.1016/j.febslet.2005.03.015
Franci G, Casalino L, Petraglia F et al (2013) The class I-specific HDAC inhibitor MS-275 modulates the differentiation potential of mouse embryonic stem cells. Biol Open 2(10):1070–1077. doi:10.1242/bio.20135587
Fredly H, Gjertsen BT, Bruserud O (2013) Histone deacetylase inhibition in the treatment of acute myeloid leukemia: the effects of valproic acid on leukemic cells, and the clinical and experimental evidence for combining valproic acid with other antileukemic agents. Clin Epigenet 5(1):12. doi:10.1186/1868-7083-5-12
Furumai R, Matsuyama A, Kobashi N et al (2002) FK228 (depsipeptide) as a natural prodrug that inhibits class I histone deacetylases. Cancer Res 62(17):4916–4921
Garcia-Manero G (2012) Myelodysplastic syndromes: 2012 update on diagnosis, risk-stratification, and management. Am J Hematol 87(7):692–701. doi:10.1002/ajh.23264
George P, Bali P, Annavarapu S et al (2005) Combination of the histone deacetylase inhibitor LBH589 and the hsp90 inhibitor 17-AAG is highly active against human CML-BC cells and AML cells with activating mutation of FLT-3. Blood 105(4):1768–1776. doi:10.1182/blood-2004-09-3413
Gilbert J, Baker SD, Bowling MK et al (2001) A phase I dose escalation and bioavailability study of oral sodium phenylbutyrate in patients with refractory solid tumor malignancies. Clin Cancer Res 7(8):2292–2300
Goffin J, Eisenhauer E (2002) DNA methyltransferase inhibitors-state of the art. Ann Oncol 13(11):1699–1716
Gottlicher M, Minucci S, Zhu P et al (2001) Valproic acid defines a novel class of HDAC inhibitors inducing differentiation of transformed cells. EMBO J 20(24):6969–6978. doi:10.1093/emboj/20.24.6969
Graca I, Sousa EJ, Baptista T et al (2014) Anti-tumoral effect of the non-nucleoside DNMT inhibitor RG108 in human prostate cancer cells. Curr Pharm Des 20(11):1803–1811
Grant S, Easley C, Kirkpatrick P (2007) Vorinostat. Nat Rev Drug Discov 6(1):21–22. doi:10.1038/nrd2227
Gravina GL, Festuccia C, Marampon F et al (2010) Biological rationale for the use of DNA methyltransferase inhibitors as new strategy for modulation of tumor response to chemotherapy and radiation. Mol Cancer 9:305. doi:10.1186/1476-4598-9-305
Gridelli C, Rossi A, Maione P (2008) The potential role of histone deacetylase inhibitors in the treatment of non-small-cell lung cancer. Crit Rev Oncol Hematol 68(1):29–36. doi:10.1016/j.critrevonc.2008.03.002
Gupta SC, Kannappan R, Reuter S, Kim JH, Aggarwal BB (2011) Chemosensitization of tumors by resveratrol. Ann N Y Acad Sci 1215:150–160. doi:10.1111/j.1749-6632.2010.05852.x
Gupta SC, Patchva S, Koh W, Aggarwal BB (2012) Discovery of curcumin, a component of golden spice, and its miraculous biological activities. Clin Exp Pharmacol Physiol 39(3):283–299. doi:10.1111/j.1440-1681.2011.05648.x
Harris WJ, Huang X, Lynch JT et al (2012) The histone demethylase KDM1A sustains the oncogenic potential of MLL-AF9 leukemia stem cells. Cancer Cell 21(4):473–487. doi:10.1016/j.ccr.2012.03.014
Hatziapostolou M, Iliopoulos D (2011) Epigenetic aberrations during oncogenesis. Cell Mol Life Sci 68(10):1681–1702. doi:10.1007/s00018-010-0624-z
Helin K, Dhanak D (2013) Chromatin proteins and modifications as drug targets. Nature 502(7472):480–488. doi:10.1038/nature12751
Heltweg B, Gatbonton T, Schuler AD et al (2006) Antitumor activity of a small-molecule inhibitor of human silent information regulator 2 enzymes. Cancer Res 66(8):4368–4377. doi:10.1158/0008-5472.CAN-05-3617
Herranz MC, Sanchez-Navarro JA, Aparicio F, Pallas V (2005) Simultaneous detection of six stone fruit viruses by non-isotopic molecular hybridization using a unique riboprobe or ‘polyprobe’. J Virol Methods 124(1–2):49–55. doi:10.1016/j.jviromet.2004.11.003
Herranz M, Martin-Caballero J, Fraga MF et al (2006) The novel DNA methylation inhibitor zebularine is effective against the development of murine T-cell lymphoma. Blood 107(3):1174–1177. doi:10.1182/blood-2005-05-2033
Hirao M, Posakony J, Nelson M et al (2003) Identification of selective inhibitors of NAD+-dependent deacetylases using phenotypic screens in yeast. J Biol Chem 278(52):52773–52782. doi:10.1074/jbc.M308966200
Hojfeldt JW, Agger K, Helin K (2013) Histone lysine demethylases as targets for anticancer therapy. Nat Rev Drug Discovery 12(12):917–930. doi:10.1038/nrd4154
Howitz KT, Bitterman KJ, Cohen HY et al (2003) Small molecule activators of sirtuins extend Saccharomyces cerevisiae lifespan. Nature 425(6954):191–196. doi:10.1038/nature01960
Huang H, Zhang J, Shen W et al (2007) Solution structure of the second bromodomain of Brd2 and its specific interaction with acetylated histone tails. BMC Struct Biol 7:57. doi:10.1186/1472-6807-7-57
Isham CR, Tibodeau JD, Jin W, Xu R, Timm MM, Bible KC (2007) Chaetocin: a promising new antimyeloma agent with in vitro and in vivo activity mediated via imposition of oxidative stress. Blood 109(6):2579–2588. doi:10.1182/blood-2006-07-027326
Ito S, Shen L, Dai Q et al (2011) Tet proteins can convert 5-methylcytosine to 5-formylcytosine and 5-carboxylcytosine. Science 333(6047):1300–1303. doi:10.1126/science.1210597
Jair KW, Bachman KE, Suzuki H et al (2006) De novo CpG island methylation in human cancer cells. Cancer Res 66(2):682–692. doi:10.1158/0008-5472.CAN-05-1980
Jin SG, Jiang Y, Qiu R et al (2011) 5-Hydroxymethylcytosine is strongly depleted in human cancers but its levels do not correlate with IDH1 mutations. Cancer Res 71(24):7360–7365. doi:10.1158/0008-5472.CAN-11-2023
Jones PA, Baylin SB (2007) The epigenomics of cancer. Cell 128(4):683–692. doi:10.1016/j.cell.2007.01.029
Jung-Hynes B, Ahmad N (2009) SIRT1 controls circadian clock circuitry and promotes cell survival: a connection with age-related neoplasms. FASEB J 23(9):2803–2809. doi:10.1096/fj.09-129148
Juttermann R, Li E, Jaenisch R (1994) Toxicity of 5-aza-2′-deoxycytidine to mammalian cells is mediated primarily by covalent trapping of DNA methyltransferase rather than DNA demethylation. Proc Natl Acad Sci USA 91(25):11797–11801
Kadia TM, Jabbour E, Kantarjian H (2011) Failure of hypomethylating agent-based therapy in myelodysplastic syndromes. Semin Oncol 38(5):682–692. doi:10.1053/j.seminoncol.2011.04.011
Karthik S, Sankar R, Varunkumar K, Ravikumar V (2014) Romidepsin induces cell cycle arrest, apoptosis, histone hyperacetylation and reduces matrix metalloproteinases 2 and 9 expression in bortezomib sensitized non-small cell lung cancer cells. Biomed Pharmacother 68(3):327–334. doi:10.1016/j.biopha.2014.01.002
Khan O, La Thangue NB (2012) HDAC inhibitors in cancer biology: emerging mechanisms and clinical applications. Immunol Cell Biol 90(1):85–94. doi:10.1038/icb.2011.100
Kimura A, Matsubara K, Horikoshi M (2005) A decade of histone acetylation: marking eukaryotic chromosomes with specific codes. J Biochem 138(6):647–662. doi:10.1093/jb/mvi184
Klein CJ, Botuyan MV, Wu Y et al (2011) Mutations in DNMT1 cause hereditary sensory neuropathy with dementia and hearing loss. Nat Genet 43(6):595–600. doi:10.1038/ng.830
Klisovic RB, Stock W, Cataland S et al (2008) A phase I biological study of MG98, an oligodeoxynucleotide antisense to DNA methyltransferase 1, in patients with high-risk myelodysplasia and acute myeloid leukemia. Clin Cancer Res 14(8):2444–2449. doi:10.1158/1078-0432.CCR-07-1320
Knutson SK, Warholic NM, Wigle TJ et al (2013) Durable tumor regression in genetically altered malignant rhabdoid tumors by inhibition of methyltransferase EZH2. Proc Natl Acad Sci USA 110(19):7922–7927. doi:10.1073/pnas.1303800110
Kooistra SM, Helin K (2012) Molecular mechanisms and potential functions of histone demethylases. Nat Rev Mol Cell Biol 13(5):297–311. doi:10.1038/nrm3327
Kouzarides T (2007) Chromatin modifications and their function. Cell 128(4):693–705. doi:10.1016/j.cell.2007.02.005
Kruidenier L, Chung CW, Cheng Z et al (2012) A selective jumonji H3K27 demethylase inhibitor modulates the proinflammatory macrophage response. Nature 488(7411):404–408. doi:10.1038/nature11262
Kubicek S, O’Sullivan RJ, August EM et al (2007) Reversal of H3K9me2 by a small-molecule inhibitor for the G9a histone methyltransferase. Mol Cell 25(3):473–481. doi:10.1016/j.molcel.2007.01.017
Kurundkar D, Srivastava RK, Chaudhary SC et al (2013) Vorinostat, an HDAC inhibitor attenuates epidermoid squamous cell carcinoma growth by dampening mTOR signaling pathway in a human xenograft murine model. Toxicol Appl Pharmacol 266(2):233–244. doi:10.1016/j.taap.2012.11.002
Kuttan R, Sudheeran PC, Josph CD (1987) Turmeric and curcumin as topical agents in cancer therapy. Tumori 73(1):29–31
Lau OD, Kundu TK, Soccio RE et al (2000) HATs off: selective synthetic inhibitors of the histone acetyltransferases p300 and PCAF. Mol Cell 5(3):589–595
Lee BH, Yegnasubramanian S, Lin X, Nelson WG (2005a) Procainamide is a specific inhibitor of DNA methyltransferase 1. J Biol Chem 280(49):40749–40756. doi:10.1074/jbc.M505593200
Lee WJ, Shim JY, Zhu BT (2005b) Mechanisms for the inhibition of DNA methyltransferases by tea catechins and bioflavonoids. Mol Pharmacol 68(4):1018–1030. doi:10.1124/mol.104.008367
Lee JH, Choy ML, Ngo L, Foster SS, Marks PA (2010) Histone deacetylase inhibitor induces DNA damage, which normal but not transformed cells can repair. Proc Natl Acad Sci USA 107(33):14639–14644. doi:10.1073/pnas.1008522107
Lee JH, Choy ML, Ngo L, Venta-Perez G, Marks PA (2011) Role of checkpoint kinase 1 (Chk1) in the mechanisms of resistance to histone deacetylase inhibitors. Proc Natl Acad Sci USA 108(49):19629–19634. doi:10.1073/pnas.1117544108
Leone G, Teofili L, Voso MT, Lubbert M (2002) DNA methylation and demethylating drugs in myelodysplastic syndromes and secondary leukemias. Haematologica 87(12):1324–1341
Ley TJ, Ding L, Walter MJ et al (2010) DNMT3A mutations in acute myeloid leukemia. N Engl J Med 363(25):2424–2433. doi:10.1056/NEJMoa1005143
Liu F, Chen X, Allali-Hassani A et al (2009) Discovery of a 2,4-diamino-7-aminoalkoxyquinazoline as a potent and selective inhibitor of histone lysine methyltransferase G9a. J Med Chem 52(24):7950–7953. doi:10.1021/Jm901543m
Liu F, Chen X, Allali-Hassani A et al (2010) Protein lysine methyltransferase G9a inhibitors: design, synthesis, and structure activity relationships of 2,4-diamino-7-aminoalkoxy-quinazolines. J Med Chem 53(15):5844–5857. doi:10.1021/jm100478y
Liu F, Barsyte-Lovejoy D, Li F et al (2013) Discovery of an in vivo chemical probe of the lysine methyltransferases G9a and GLP. J Med Chem 56(21):8931–8942. doi:10.1021/jm401480r
Maes T, Tirapu I, Mascaro C et al (2013) Preclinical characterization of a potent and selective inhibitor of the histone demethylase KDM1A for MLL leukemia. J Clin Oncol 31(15):e13543
Mai A (2007) The therapeutic uses of chromatin-modifying agents. Expert Opin Ther Targets 11(6):835–851. doi:10.1517/14728222.11.6.835
Mai A, Altucci L (2009) Epi-drugs to fight cancer: from chemistry to cancer treatment, the road ahead. Int J Biochem Cell Biol 41(1):199–213. doi:10.1016/j.biocel.2008.08.020
Mantelingu K, Kishore AH, Balasubramanyam K et al (2007) Activation of p300 histone acetyltransferase by small molecules altering enzyme structure: probed by surface-enhanced Raman spectroscopy. J Phys Chem B 111(17):4527–4534. doi:10.1021/jp067655s
Marcu MG, Jung YJ, Lee S et al (2006) Curcumin is an inhibitor of p300 histone acetylatransferase. Med Chem 2(2):169–174
Marks PW (2012) Decitabine for acute myeloid leukemia. Expert Rev Anticancer Ther 12(3):299–305. doi:10.1586/era.11.207
Marks PA, Richon VM, Breslow R, Rifkind RA (2001) Histone deacetylase inhibitors as new cancer drugs. Curr Opin Oncol 13(6):477–483
McCabe MT, Ott HM, Ganji G et al (2012) EZH2 inhibition as a therapeutic strategy for lymphoma with EZH2-activating mutations. Nature 492(7427):108–112. doi:10.1038/nature11606
McDonough MA, Loenarz C, Chowdhury R, Clifton IJ, Schofield CJ (2010) Structural studies on human 2-oxoglutarate dependent oxygenases. Curr Opin Struct Biol 20(6):659–672. doi:10.1016/j.sbi.2010.08.006
McGeary RP, Bennett AJ, Tran QB, Cosgrove KL, Ross BP (2008) Suramin: clinical uses and structure-activity relationships. Mini Rev Med Chem 8(13):1384–1394
Meng F, Sun G, Zhong M, Yu Y, Brewer MA (2013) Anticancer efficacy of cisplatin and trichostatin A or 5-aza-2′-deoxycytidine on ovarian cancer. Br J Cancer 108(3):579–586. doi:10.1038/bjc.2013.10
Mertz JA, Conery AR, Bryant BM et al (2011) Targeting MYC dependence in cancer by inhibiting BET bromodomains. Proc Natl Acad Sci USA 108(40):16669–16674. doi:10.1073/pnas.1108190108
Milne JC, Lambert PD, Schenk S et al (2007) Small molecule activators of SIRT1 as therapeutics for the treatment of type 2 diabetes. Nature 450(7170):712–716. doi:10.1038/nature06261
Mirguet O, Gosmini R, Toum J et al (2013) Discovery of epigenetic regulator I-BET762: lead optimization to afford a clinical candidate inhibitor of the BET bromodomains. J Med Chem 56(19):7501–7515. doi:10.1021/jm401088k
Momparler RL, Bovenzi V (2000) DNA methylation and cancer. J Cell Physiol 183(2):145–154. doi:10.1002/(SICI)1097-4652(200005)183:2<145::AID-JCP1>3.0.CO;2-V
Mummaneni P, Shord SS (2014) Epigenetics and oncology. Pharmacotherapy 34(5):495–505. doi:10.1002/phar.1408
Muraoka M, Konishi M, Kikuchi-Yanoshita R et al (1996) p300 gene alterations in colorectal and gastric carcinomas. Oncogene 12(7):1565–1569
Nakagawa H, Hasumi K, Woo JT, Nagai K, Wachi M (2004) Generation of hydrogen peroxide primarily contributes to the induction of Fe(II)-dependent apoptosis in Jurkat cells by (−)-epigallocatechin gallate. Carcinogenesis 25(9):1567–1574. doi:10.1093/carcin/bgh168
Nakamura K, Aizawa K, Nakabayashi K et al (2013) DNA methyltransferase inhibitor zebularine inhibits human hepatic carcinoma cells proliferation and induces apoptosis. PLoS ONE 8(1):e54036. doi:10.1371/journal.pone.0054036
Napper AD, Hixon J, McDonagh T et al (2005) Discovery of indoles as potent and selective inhibitors of the deacetylase SIRT1. J Med Chem 48(25):8045–8054. doi:10.1021/jm050522v
Nebbioso A, Pereira R, Khanwalkar H et al (2011) Death receptor pathway activation and increase of ROS production by the triple epigenetic inhibitor UVI5008. Mol Cancer Ther 10(12):2394–2404. doi:10.1158/1535-7163.MCT-11-0525
Nebbioso A, Carafa V, Benedetti R, Altucci L (2012) Trials with ‘epigenetic’ drugs: an update. Mol Oncol 6(6):657–682. doi:10.1016/j.molonc.2012.09.004
Oike T, Ogiwara H, Torikai K, Nakano T, Yokota J, Kohno T (2012) Garcinol, a histone acetyltransferase inhibitor, radiosensitizes cancer cells by inhibiting non-homologous end joining. Int J Radiat Oncol Biol Phys 84(3):815–821. doi:10.1016/j.ijrobp.2012.01.017
Okano M, Bell DW, Haber DA, Li E (1999) DNA methyltransferases Dnmt3a and Dnmt3b are essential for de novo methylation and mammalian development. Cell 99(3):247–257
Oki Y, Buglio D, Fanale M et al (2013) Phase I study of panobinostat plus everolimus in patients with relapsed or refractory lymphoma. Clin Cancer Res 19(24):6882–6890. doi:10.1158/1078-0432.CCR-13-1906
Olsen EA, Kim YH, Kuzel TM et al (2007) Phase IIb multicenter trial of vorinostat in patients with persistent, progressive, or treatment refractory cutaneous T-cell lymphoma. J Clin Oncol 25(21):3109–3115. doi:10.1200/JCO.2006.10.2434
Outeiro TF, Kontopoulos E, Altmann SM et al (2007) Sirtuin 2 inhibitors rescue alpha-synuclein-mediated toxicity in models of Parkinson’s disease. Science 317(5837):516–519. doi:10.1126/science.1143780
Pacholec M, Bleasdale JE, Chrunyk B et al (2010) SRT1720, SRT2183, SRT1460, and resveratrol are not direct activators of SIRT1. J Biol Chem 285(11):8340–8351. doi:10.1074/jbc.M109.088682
Patutina OA, Mironova NL, Vlassov VV, Zenkova MA (2009) New approaches for cancer treatment: antitumor drugs based on gene-targeted nucleic acids. Acta Nat 1(2):44–60
Perego P, Zuco V, Gatti L, Zunino F (2012) Sensitization of tumor cells by targeting histone deacetylases. Biochem Pharmacol 83(8):987–994. doi:10.1016/j.bcp.2011.11.010
Pfeifer GP, Kadam S, Jin SG (2013) 5-hydroxymethylcytosine and its potential roles in development and cancer. Epigenetics Chromatin 6(1):10. doi:10.1186/1756-8935-6-10
Pili R, Salumbides B, Zhao M et al (2012) Phase I study of the histone deacetylase inhibitor entinostat in combination with 13-cis retinoic acid in patients with solid tumours. Br J Cancer 106(1):77–84. doi:10.1038/bjc.2011.527
Pina IC, Gautschi JT, Wang GY et al (2003) Psammaplins from the sponge Pseudoceratina purpurea: inhibition of both histone deacetylase and DNA methyltransferase. J Org Chem 68(10):3866–3873. doi:10.1021/jo034248t
Qian DZ, Kachhap SK, Collis SJ et al (2006) Class II histone deacetylases are associated with VHL-independent regulation of hypoxia-inducible factor 1 alpha. Cancer Res 66(17):8814–8821. doi:10.1158/0008-5472.CAN-05-4598
Ragno R, Simeoni S, Castellano S et al (2007) Small molecule inhibitors of histone arginine methyltransferases: homology modeling, molecular docking, binding mode analysis, and biological evaluations. J Med Chem 50(6):1241–1253. doi:10.1021/jm061213n
Rhodes LV, Nitschke AM, Segar HC et al (2012) The histone deacetylase inhibitor trichostatin A alters microRNA expression profiles in apoptosis-resistant breast cancer cells. Oncol Rep 27(1):10–16. doi:10.3892/or.2011.1488
Richon VM (2008) A new path to the cancer epigenome. Nat Biotechnol 26(6):655–656. doi:10.1038/nbt0608-655
Richon VM, Emiliani S, Verdin E et al (1998) A class of hybrid polar inducers of transformed cell differentiation inhibits histone deacetylases. Proc Natl Acad Sci USA 95(6):3003–3007
Robertson KD (2001) DNA methylation, methyltransferases, and cancer. Oncogene 20(24):3139–3155. doi:10.1038/sj.onc.1204341
Robertson KD, Uzvolgyi E, Liang G et al (1999) The human DNA methyltransferases (DNMTs) 1, 3a and 3b: coordinate mRNA expression in normal tissues and overexpression in tumors. Nucleic Acids Res 27(11):2291–2298
Rocha-Gonzalez HI, Ambriz-Tututi M, Granados-Soto V (2008) Resveratrol: a natural compound with pharmacological potential in neurodegenerative diseases. CNS Neurosci Ther 14(3):234–247. doi:10.1111/j.1755-5949.2008.00045.x
Rodriguez-Paredes M, Esteller M (2011) Cancer epigenetics reaches mainstream oncology. Nat Med 17(3):330–339. doi:10.1038/nm.2305
Rosato R, Hock S, Dent P, Dai Y, Grant S (2012) LBH-589 (panobinostat) potentiates fludarabine anti-leukemic activity through a JNK- and XIAP-dependent mechanism. Leuk Res 36(4):491–498. doi:10.1016/j.leukres.2011.10.020
Ruthenburg AJ, Li H, Patel DJ, Allis CD (2007) Multivalent engagement of chromatin modifications by linked binding modules. Nat Rev Mol Cell Biol 8(12):983–994. doi:10.1038/nrm2298
Ryan QC, Headlee D, Acharya M et al (2005) Phase I and pharmacokinetic study of MS-275, a histone deacetylase inhibitor, in patients with advanced and refractory solid tumors or lymphoma. J Clin Oncol 23(17):3912–3922. doi:10.1200/JCO.2005.02.188
Sagar V, Zheng W, Thompson PR, Cole PA (2004) Bisubstrate analogue structure-activity relationships for p300 histone acetyltransferase inhibitors. Bioorg Med Chem 12(12):3383–3390. doi:10.1016/j.bmc.2004.03.070
Sakane C, Okitsu T, Wada A, Sagami H, Shidoji Y (2014) Inhibition of lysine-specific demethylase 1 by the acyclic diterpenoid geranylgeranoic acid and its derivatives. Biochem Biophys Res Commun 444(1):24–29. doi:10.1016/j.bbrc.2013.12.144
Savickiene J, Treigyte G, Jazdauskaite A, Borutinskaite VV, Navakauskiene R (2012) DNA methyltransferase inhibitor RG108 and histone deacetylase inhibitors cooperate to enhance NB4 cell differentiation and E-cadherin re-expression by chromatin remodelling. Cell Biol Int 36(11):1067–1078. doi:10.1042/CBI20110649
Schaefer M, Lyko F (2010) Solving the Dnmt2 enigma. Chromosoma 119(1):35–40. doi:10.1007/s00412-009-0240-6
Schaefer M, Pollex T, Hanna K, Tuorto F, Meusburger M, Helm M, Lyko F (2010) RNA methylation by Dnmt2 protects transfer RNAs against stress-induced cleavage. Genes Dev 24(15):1590–1595. doi: 10.1101/gad.586710
Schuetz A, Min J, Antoshenko T et al (2007) Structural basis of inhibition of the human NAD+-dependent deacetylase SIRT5 by suramin. Structure 15(3):377–389. doi:10.1016/j.str.2007.02.002
Segura-Pacheco B, Trejo-Becerril C, Perez-Cardenas E et al (2003) Reactivation of tumor suppressor genes by the cardiovascular drugs hydralazine and procainamide and their potential use in cancer therapy. Clin Cancer Res 9(5):1596–1603
Shukla Y, Singh R (2011) Resveratrol and cellular mechanisms of cancer prevention. Ann N Y Acad Sci 1215:1–8. doi:10.1111/j.1749-6632.2010.05870.x
Sikandar S, Dizon D, Shen X, Li Z, Besterman J, Lipkin SM (2010) The class I HDAC inhibitor MGCD0103 induces cell cycle arrest and apoptosis in colon cancer initiating cells by upregulating Dickkopf-1 and non-canonical Wnt signaling. Oncotarget 1(7):596–605
Silvestri L, Ballante F, Mai A, Marshall GR, Ragno R (2012) Histone deacetylase inhibitors: structure-based modeling and isoform-selectivity prediction. J Chem Inf Model 52(8):2215–2235. doi:10.1021/ci300160y
Skliris GP, Munot K, Bell SM et al (2003) Reduced expression of oestrogen receptor beta in invasive breast cancer and its re-expression using DNA methyl transferase inhibitors in a cell line model. J Pathol 201(2):213–220. doi:10.1002/path.1436
Smith BC, Denu JM (2009) Chemical mechanisms of histone lysine and arginine modifications. Biochim Biophys Acta 1789(1):45–57. doi:10.1016/j.bbagrm.2008.06.005
Song S, Yu B, Wei Y, Wientjes MG, Au JL (2004) Low-dose suramin enhanced paclitaxel activity in chemotherapy-naive and paclitaxel-pretreated human breast xenograft tumors. Clin Cancer Res 10(18 Pt 1):6058–6065. doi:10.1158/1078-0432.CCR-04-0595
Spannhoff A, Hauser AT, Heinke R, Sippl W, Jung M (2009) The emerging therapeutic potential of histone methyltransferase and demethylase inhibitors. ChemMedChem 4(10):1568–1582. doi:10.1002/cmdc.200900301
Sroczynska P, Cruickshank VA, Bukowski JP et al (2014) shRNA screening identifies JMJD1C as being required for leukemia maintenance. Blood 123(12):1870–1882. doi:10.1182/blood-2013-08-522094
Stewart DJ, Donehower RC, Eisenhauer EA et al (2003) A phase I pharmacokinetic and pharmacodynamic study of the DNA methyltransferase 1 inhibitor MG98 administered twice weekly. Ann Oncol 14(5):766–774
Stimson L, Rowlands MG, Newbatt YM et al (2005) Isothiazolones as inhibitors of PCAF and p300 histone acetyltransferase activity. Mol Cancer Ther 4(10):1521–1532. doi:10.1158/1535-7163.MCT-05-0135
Stolfa DA, Stefanachi A, Gajer JM et al (2012) Design, synthesis, and biological evaluation of 2-aminobenzanilide derivatives as potent and selective HDAC inhibitors. ChemMedChem 7(7):1256–1266. doi:10.1002/cmdc.201200193
Suzuki H, Gabrielson E, Chen W et al (2002) A genomic screen for genes upregulated by demethylation and histone deacetylase inhibition in human colorectal cancer. Nat Genet 31(2):141–149. doi:10.1038/ng892
Sweis RF, Pliushchev M, Brown PJ et al (2014) Discovery and development of potent and selective inhibitors of histone methyltransferase g9a. ACS Med Chem Lett 5(2):205–209. doi:10.1021/ml400496h
Tan J, Yang X, Zhuang L et al (2007) Pharmacologic disruption of polycomb-repressive complex 2-mediated gene repression selectively induces apoptosis in cancer cells. Genes Dev 21(9):1050–1063. doi:10.1101/gad.1524107
Taverna SD, Li H, Ruthenburg AJ, Allis CD, Patel DJ (2007) How chromatin-binding modules interpret histone modifications: lessons from professional pocket pickers. Nat Struct Mol Biol 14(11):1025–1040. doi:10.1038/nsmb1338
Terao Y, Nishida J, Horiuchi S et al (2001) Sodium butyrate induces growth arrest and senescence-like phenotypes in gynecologic cancer cells. Int J Cancer J Int Du Cancer 94(2):257–267
Timmers S, Auwerx J, Schrauwen P (2012) The journey of resveratrol from yeast to human. Aging 4(3):146–158
Tsuji N, Kobayashi M, Nagashima K, Wakisaka Y, Koizumi K (1976) A new antifungal antibiotic, trichostatin. J Antibiot 29(1):1–6
Ueda R, Suzuki T, Mino K et al (2009) Identification of cell-active lysine specific demethylase 1-selective inhibitors. J Am Chem Soc 131(48):17536–17537. doi:10.1021/ja907055q
van Haaften G, Dalgliesh GL, Davies H et al (2009) Somatic mutations of the histone H3K27 demethylase gene UTX in human cancer. Nat Genet 41(5):521–523. doi:10.1038/ng.349
Vannini A, Volpari C, Filocamo G et al (2004) Crystal structure of a eukaryotic zinc-dependent histone deacetylase, human HDAC8, complexed with a hydroxamic acid inhibitor. Proc Natl Acad Sci USA 101(42):15064–15069. doi:10.1073/pnas.0404603101
Villalba JM, Alcain FJ (2012) Sirtuin activators and inhibitors. BioFactors 38(5):349–359. doi:10.1002/biof.1032
Villar-Garea A, Esteller M (2003) DNA demethylating agents and chromatin-remodelling drugs: which, how and why? Curr Drug Metab 4(1):11–31
Walter MJ, Ding L, Shen D et al (2011) Recurrent DNMT3A mutations in patients with myelodysplastic syndromes. Leukemia 25(7):1153–1158. doi:10.1038/leu.2011.44
Wang H, Yang YJ, Qian HY, Zhang Q, Xu H, Li JJ (2012) Resveratrol in cardiovascular disease: what is known from current research? Heart Fail Rev 17(3):437–448. doi:10.1007/s10741-011-9260-4
Whetstine JR, Nottke A, Lan F et al (2006) Reversal of histone lysine trimethylation by the JMJD2 family of histone demethylases. Cell 125(3):467–481. doi:10.1016/j.cell.2006.03.028
Xu S, Ren J, Chen HB et al (2014) Cytostatic and apoptotic effects of DNMT and HDAC inhibitors in endometrial cancer cells. Curr Pharm Des 20(11):1881–1887
Yang Q, Wang B, Zang W et al (2013) Resveratrol inhibits the growth of gastric cancer by inducing G1 phase arrest and senescence in a Sirt1-dependent manner. PLoS ONE 8(11):e70627. doi:10.1371/journal.pone.0070627
Zuber J, Shi J, Wang E et al (2011) RNAi screen identifies Brd4 as a therapeutic target in acute myeloid leukaemia. Nature 478(7370):524–528. doi:10.1038/nature10334
Acknowledgments
This work was supported by: EU, Blueprint project no. 282510; the Italian Flag Project: EPIGEN; AIRC No. 11812; PRIN-2012. We apologize to the authors whose work we could not cite due to reference restrictions. We wish to thank C. Fisher for linguistic editing of the manuscript.
Conflict of interest
The authors declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Di Costanzo, A., Del Gaudio, N., Migliaccio, A. et al. Epigenetic drugs against cancer: an evolving landscape. Arch Toxicol 88, 1651–1668 (2014). https://doi.org/10.1007/s00204-014-1315-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-014-1315-6