Abstract
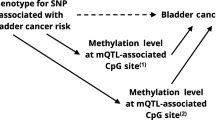
Recently, rs11892031[A] has been identified in a genome-wide association study (GWAS) to confer increased risk of urinary bladder cancer (UBC). To confirm this association and additionally study a possible relevance of exposure to urinary bladder carcinogens, we investigated the IfADo UBC study group, consisting of eight case–control series from different regions including 1,805 cases and 2,141 controls. This analysis was supplemented by a meta-analysis of all published data, including 13,395 cases and 54,876 controls. Rs11892031 A/A was significantly associated with UBC risk in the IfADo case–control series adjusted to cigarette smoking, gender, age and ethnicity (OR = 1.18; 95% CI = 1.02–1.37; P = 0.026). In the meta-analysis, a convincing association with UBC risk was obtained (OR = 1.19; 95% Cl = 1.12–1.26; P < 0.0001). Interestingly, the highest odds ratios were obtained for individual case–control series with a high degree of occupational exposure to polycyclic aromatic hydrocarbons and aromatic amines: cases with suspected occupational UBC (OR = 1.41) and cases from the highly industrialized Ruhr area (OR = 1.98) compared with Ruhr area controls (all combined OR = 1.46). Odds ratios were lower for study groups with no or a lower degree of occupational exposure to bladder carcinogens, such as the Hungary (OR = 1.02) or the ongoing West German case–control series (OR = 1.06). However, the possible association of rs11892031[A] with exposure to bladder carcinogens still should be interpreted with caution, because in contrast to the differences between the individual study groups, interview-based data on occupational exposure were not significantly associated with rs11892031. In conclusion, the association of rs11892031[A] with UBC risk could be confirmed in independent study groups.



Similar content being viewed by others
References
Babu SR, Lakshmi VM, Zenser TV, Davis BB (1994) Glucuronidation of N-acetylbenzidine by human liver. Drug Metab Dispos 22:922–927
Babu SR, Lakshmi VM, Hsu FF, Zenser TV, Davis BB (1995) Glucuronidation of N-hydroxy metabolites of N-acetylbenzidine. Carcinogenesis 16:3069–3074
Barrett JC, Fry B, Maller J, Daly MJ (2005) Haploview: analysis and visualization of LD and haplotype maps. Bioinformatics 21:263–265
Bellemare J, Rouleau M, Harvey M, Popa I, Pelletier G, Têtu B, Guillemette C (2011) Immunohistochemical expression of conjugating UGT1A-derived isoforms in normal and tumoral drug-metabolizing tissues in humans. J Pathol 223:425–435
Bretschneider F (2009) Das Ruhrgebiet im Umbruch. Strukturwandel einer Region. Essen: RVR. http://www.metropoleruhr.de/fileadmin/user_upload/metropoleruhr.de/Bilder/Metanavigation_unten/Presse/Wirtschaft_0209_neu.pdf. Accessed 23 February 2012
Carreón T, LeMasters GK, Ruder AM, Schulte PA (2006) The genetic and environmental factors involved in benzidine metabolism and bladder carcinogenesis in exposed workers. Front Biosci 11:2889–2902
Ciotti M, Lakshmi VM, Basu N, Davis BB, Owens IS, Zenser TV (1999) Glucuronidation of benzidine and its metabolites by cDNA-expressed human UDP-glucuronosyltransferases and pH stability of glucuronides. Carcinogenesis 20:1963–1969
Degen GH, Schlattjan JH, Mähler S, Föllmann W, Golka K (2004) Comparative metabolic activation of benzidine and N-acetylbenzidine by prostaglandin H synthase. Toxicol Lett 151:135–142
Dupont WD, Plummer WD (1990) Power and sample size calculations: A review and computer program. Control Clin Trials 11:116–128
Dupont WD, Plummer WD (1998) Power and sample size calculations for studies involving linear regression. Control Clin Trials 19:589–601
Freudenthal RI, Stephens E, Anderson DP (1999) Determining the potential of aromatic amines to induce cancer of the urinary bladder. Int J Toxicol 18:353–359
Garcia-Closas M, Ye Y, Rothman N et al (2011) A genome-wide association study of bladder cancer identifies a new susceptibility locus within SLC14A1, a urea transporter gene on chromosome 18q12.3. Hum Mol Genet 20:4282–4289
Giuliani L, Gazzaniga P, Caporuscio F, Ciotti M, Frati L, Aglianò AM (2001) Can down-regulation of UDP-glucuronosyltransferases in the urinary bladder tissue impact the risk of chemical carcinogenesis? Int J Cancer 91:141–143
Giuliani L, Ciotti M, Stoppacciaro A et al (2005) UDP-glucuronosyltransferases 1A expression in human urinary bladder and colon cancer by immunohistochemistry. Oncol Rep 13:185–191
Golka K, Hermes M, Selinski S et al (2009) Susceptibility to urinary bladder cancer: relevance of rs9642880[T], GSTM1 0/0 and occupational exposure. Pharmacogenet Genomics 19:903–906
Golka K, Selinski S, Lehmann ML et al (2011) Genetic variants in urinary bladder cancer: collective power of the “wimp SNPs”. Arch Toxicol 85:539–554
Green MD, Tephly TR (1998) Glucuronidation of amine substrates by purified and expressed UDP-glucuronosyltransferase proteins. Drug Metab Dispos 26:860–867
IARC GLOBOCAN (2008) http://globocan.iarc.fr. Accessed 23 February 2012
Iida K, Itoh K, Kumagai Y et al (2004) Nrf2 is essential for the chemopreventive efficacy of oltipraz against urinary bladder carcinogenesis. Cancer Res 64:6424–6431
Iida K, Itoh K, Maher JM et al (2007) Nrf2 and p53 cooperatively protect against BBN-induced urinary bladder carcinogenesis. Carcinogenesis 28:2398–2403
Iida K, Mimura J, Itoh K et al (2010) Suppression of AhR signaling pathway is associated with the down-regulation of UDP-glucuronosyltransferases during BBN-induced urinary bladder carcinogenesis in mice. J Biochem 147:353–360
Izumi K, Zheng Y, Hsu JW, Chang C, Miyamoto H (2011) Androgen receptor signals regulate UDP-glucuronosyltransferases in the urinary bladder: a potential mechanism of androgen-induced bladder carcinogenesis. Mol Carcinog (2011 Nov 15) (Epub ahead of print)
Kaivosaari S, Finel M, Koskinen M (2011) N-glucuronidation of drugs and other xenobiotics by human and animal UDP-glucuronosyltransferases. Xenobiotica 41:652–669
Kalthoff S, Ehmer U, Freiberg N, Manns MP, Strassburg CP (2010) Interaction between oxidative stress sensor Nrf2 and xenobiotic-activated aryl hydrocarbon receptor in the regulation of the human phase II detoxifying UDP-glucuronosyltransferase 1A10. J Biol Chem 285:5993–6002
Kiemeney LA, Thorlacius S, Sulem P et al (2008) Sequence variant on 8q24 confers susceptibility to urinary bladder cancer. Nat Genet 40:1307–1312
Kiemeney LA, Sulem P, Besenbacher S et al (2010) A sequence variant at 4p16.3 confers susceptibility to urinary bladder cancer. Nat Genet 42:415–419
Lewis CM (2002) Genetic association studies: design, analysis and interpretation. Brief Bioinform 3:146–153
Lin GF, Guo WC, Chen JG et al (2005) An association of UDP-glucuronosyltransferase 2B7 C802T (His268Tyr) polymorphism with bladder cancer in benzidine-exposed workers in China. Toxicol Sci 85:502–506
Nakamura A, Nakajima M, Yamanaka H, Fujiwara R, Yokoi T (2008) Expression of UGT1A and UGT2B mRNA in human normal tissues and various cell lines. Drug Metab Dispos 36:1461–1464
NCBI Gene (2012) http://www.ncbi.nlm.nih.gov/sites/entrez?db=gene&cmd=Retrieve&dopt=Graphics&list_uids=54575. Accessed 23 February 2012
Rafnar T, Sulem P, Stacey SN et al (2009) Sequence variants at the TERT-CLPTM1L locus associate with many cancer types. Nat Genet 41:221–227
Rafnar T, Vermeulen SH, Sulem P et al (2011) European genome-wide association study identifies SLC14A1 as a new urinary bladder cancer susceptibility gene. Hum Mol Genet 20:4268–4281
Rothman N, Garcia-Closas M, Chatterjee N et al (2010) A multi-stage genome-wide association study of bladder cancer identifies multiple susceptibility loci. Nat Genet 42:978–984
Saravana Devi S, Vinayagamoorthy N, Agrawal M et al (2008) Distribution of detoxifying genes polymorphism in Maharastrian population of central India. Chemosphere 70:1835–1839
Strassburg CP, Oldhafer K, Manns MP, Tukey RH (1997) Differential expression of the UGT1A locus in human liver, biliary, and gastric tissue: identification of UGT1A7 and UGT1A10 transcripts in extrahepatic tissue. Mol Pharmacol 52:212–220
Strassburg CP, Strassburg A, Nguyen N, Li Q, Manns MP, Tukey RH (1999) Regulation and function of family 1 and family 2 UDP-glucuronosyltransferase genes (UGT1A, UGT2B) in human oesophagus. Biochem J 338:489–498
Tang W, Fu YP, Figueroa JD et al (2012) Mapping of the UGT1A locus identifies an uncommon coding variant that affects mRNA expression and protects from bladder cancer. Hum Mol Genet 21:1918–1930
Tukey RH, Strassburg CP (2000) Human UDP-glucuronosyltransferases: metabolism, expression, and disease. Annu Rev Pharmacol Toxicol 40:581–616
Wikipedia (2012a) Coal mining plant Minister Stein. http://de.wikipedia.org/wiki/Zeche_Minister_Stein. Accessed 23 February 2012
Wikipedia (2012b) Coke oven plant Kaiserstuhl. http://de.wikipedia.org/wiki/Kokerei_Kaiserstuhl. Accessed 23 February 2012
Wikipedia (2012c) West Yorkshire. http://en.wikipedia.org/wiki/West_Yorkshire. Accessed 24 February 2012
Wikipedia (2012d) Los Angeles. http://en.wikipedia.org/wiki/Los_Angeles. Accessed 24 February 2012
Wu X, Ye Y, Kiemeney LA et al (2009) Genetic variation in the prostate stem cell antigen gene PSCA confers susceptibility to urinary bladder cancer. Nat Genet 41:991–995
Zenser TV, Lakshmi VM, Hsu FF, Davis BB (2002) Metabolism of N-acetylbenzidine and initiation of bladder cancer. Mutat Res 506–507:29–40
Zimmermann A, Blaszkewicz M, Roth G et al (2008) UDP-glucuronosyltransferase 2B7 C802T (His268Tyr) polymorphism in bladder cancer cases. J Toxicol Environ Health A 71:911–914
Acknowledgments
We would like to thank Rothman et al. for providing genotype frequency data by study for the meta-analysis of rs11892031, as published in Nature Genetics 2010 Nov;42(11):978–84. The authors thank Ms Kirsten Liesenhoff-Henze, Ms Marion Page and Ms Claudia Schulte-Dahmann for excellent technical support.
Author information
Authors and Affiliations
Corresponding author
Additional information
Jan G. Hengstler and Klaus Golka: shared senior authorship.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Selinski, S., Lehmann, ML., Blaszkewicz, M. et al. Rs11892031[A] on chromosome 2q37 in an intronic region of the UGT1A locus is associated with urinary bladder cancer risk. Arch Toxicol 86, 1369–1378 (2012). https://doi.org/10.1007/s00204-012-0854-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-012-0854-y