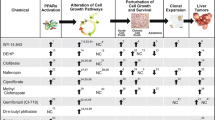
Abstract
Risk assessment based on rodent carcinogenicity data depends on the assumption of similarity between rodents and humans. While this assumption is conceivable in the case of genotoxic initiating carcinogens, considerable species differences have been observed with nongenotoxic tumor promoters. This heterogeneous group of agents increases the probability of cancer by stimulating selection and clonal expansion of cells transformed during tumor initiation. Since tumor promoters differentially affect normal tissue and preneoplastic cell clones, their action cannot be discussed without knowledge of persistent genomic and epigenetic alterations occurring during initiation and formation of preneoplastic cells. Chemical carcinogenesis, and in particular, tumor promotion, is known to be tissue specific. We focus on hepatocarcinogenesis in humans and in animal models and emphasize two different modes of action: (1) chronic cytotoxicity leading to promotion of liver carcinogenesis in both humans and animal models; (2) sustained activation of orphan receptors such as CAR, PPARα and Ah receptor leading to promotion of rodent but probably not human hepatocarcinogenesis. Further studies on the different modes of action may help to avoid overestimation of the risk of liver tumor promotion.


Similar content being viewed by others
References
Andrews NC, Schmidt PJ (2007) Iron homeostasis. Annu Rev Physiol 69:69–85
Aydinlik H, Nguyen TD, Moennikes O, Buchmann A, Schwarz M (2001) Selective pressure during tumor promotion by phenobarbital leads to clonal outgrowth of β-catenin-mutated mouse liver tumors. Oncogene 20:7812–7816
Bartsch H, Nair J (2006) Chronic inflammation and oxidative stress in the genesis and perpetuation of cancer: role of lipid peroxidation, DNA damage, and repair. Langenbecks Arch Surg 391:499–510
Beebe LE, Fornwald LW, Diwan BA, Anver MR, Anderson LM (1995) Promotion of N-nitrosodiethylamine-initiated hepatocellular tumors and hepatoblastomas by 2,3,7,8-tetrachrlorodibenzo-p-dioxin or Arochlor 1254 in C57BL/6, DBA/2 and B6D2F1 mice. Cancer Res 55:4875–4880
Bertazzi PA, Bernucci I, Brambilla G, Consonni D, Pesatori AC (1998) The Seveso studies on early and long-term effects of dioxin exposure: a review. Environ Health Perspect 106(Suppl 2):625–633
Bock KW, Köhle C (2005) Ah receptor- and TCDD-mediated liver tumor promotion: clonal selection and expansion of cells evading growth arrest and apoptosis. Biochem Pharmacol 69:1403–1408
Bock KW, Gschaidmeier H, Bock-Hennig BS, Eriksson LC (2000) Density-dependent growth of normal and nodular hepatocytes. Toxicology 144:51–56
Bosch FX, Ribes J, Borras J (1999) Epidemiology of primary liver cancer. Semin Liver Dis 19:271–285
Boyault S, Rickman DS, de Reynies A, Balabaud C, Rebouissou S, Jeannot E, Herault A, Saric J, Belghiti J, Franco D, Biolac-Sage P, Laurent-Puig P, Zucman-Rossi J (2007) Transcriptome classification of HCC is related to gene alterations and to new therapeutic targets. Hepatology 45:42–52
Buchmann A, Ziegler S, Wolf A, Robertson LW, Durham SK, Schwarz M (1991) Effects of polychlorinated biphenyls in rat liver: correlation between primary subcellular effects and promoting activity. Toxicol Appl Pharmacol 111:454–468
Buchmann A, Stinchcombe S, Körner W, Hagenmaier H, Bock KW (1994) Effects of 2,3,7,8-tetrachloro- and 1,2,3,6,7,8-heptachlorodibenzo-p-dioxin on the proliferation of preneoplastic liver cells in the rat. Carcinogenesis 15:1143–1150
Calvisi DF, Ladu S, Factor VM, Thorgeirsson SS (2004) Activation of β-catenin provides proliferative and invasive advantages in c-myc/TGFα hepatocarcinogenesis promoted by phenobarbital. Carcinogenesis 25:901–908
Chen DS (1993) From hepatitis to hepatoma: lessons from type B viral hepatitis. Science 262:369–370
Columbano A, Ledda-Columbano GM, Pibiri M, Cossu C, Menegazzi M, Moore DD, Huang W, Tian J, Locker J (2005) Gadd45β is induced through a CAR-dependent, TNF-independent pathway in murine liver hyperplasia. Hepatology 42:1118–1126
Cougot D, Neuveut C, Buendia MA (2005) HBV induced carcinogenesis. J Clin Virol 34(Suppl 1):S75–78
Davis LM, Caspary WJ, Sakallah SA, Maronpot R, Wiseman R, Barret JC, Elliot R, Houier JC (1994) Loss of heterozygosity in spontaneous and chemically induced tumors of the B6C3F1 mouse. Carcinogenesis 15:1637–1645
Devereux TR, Anna CH, Foley JF, White CM, Sills RC, Barrett JC (1999) Mutation of beta-catenin is an early event in chemically induced mouse hepatocellular carcinogenesis. Oncogene 18:4726–4733
Dietrich A, Faust D, Budt S, Moskwa M, Kunz A, Bock KW, Oesch F (2002) 2,3,7,8-tetrachlorodibenzo-p-dioxin-dependent release from contact inhibition in WB-F344 cells: involvement of cyclin A. Toxicol Appl Pharmacol 183:117–126
Diry M, Tomkiewicz C, Koehle C, Coumoul X, Bock KW, Barouki R, Transy C (2006) Activation of the dioxin/aryl hydrocarbon receptor (AhR) modulates cell plasticity through a JNK-dependent mechanism. Oncogene 25:5570–5574
Druckrey H (1967) Quantitative aspects of chemical carcinogenesis. UICC Monogr 7:60–78
Dunsford HA, Sell S, Chisari FV (1990) Hepatocarcinogenesis due to chronic liver cell injury in hepatitis B virus transgenic mice. Cancer Res 50:3400–3407
Eriksson LC, Blank A, Bock KW, Mannervik B (1987) Metabolism of xenobiotics in hepatic nodules. Toxicol Pathol 15:27–42
Fletcher LM, Dixon JL, Purdie DM, Powell LW, Crawford DHG (2002) Excess alcohol greatly increases the prevalence of cirrhosis in hereditary hemochromatosis. Gastroenterology 122:281–289
Gold LS, Slone TH, Manley NB, Bernstein L (1991) Target organs in chronic bioassays of 533 chemical carcinogens. Environ Health Perspect 93:233–246
Gu YZ, Hogenesh JB, Bradfield CA (2000) The PAS superfamily. Sensors of environmental and developmental signals. Ann Rev Pharmacol Toxicol 40:519–561
Hanahan D, Weinberg RA (2000) The hallmarks of cancer. Cell 100:57–70
Hasmall SC, Roberts RA (1999) The perturbation of apoptosis and mitosis by drugs and xenobiotics. Pharmacol Ther 82:63–70
Hillegass JM, Murphy KA, Villano CM, White LA (2006) The impact of aryl hydrocarbon receptor signaling on matrix metabolism: implications for development and disease. Biol Chem 387:1159–1173
Hollstein M, Sidransky D, Vogelstein B, Harris CC (1991) P53 mutations in human cancers. Science 253:49–53
Holzapple MP, Pitot HC, Cohen SH, Boobis AR, Klaunig JE, Pastoor T, Dellarco VL, Dragan YP (2005) Mode of action in relation of rodent liver tumors to human cancer risk. Toxicol Sci 89:51–56
Hosseinpour F, Moore R, Negishi M, Sueyoshi T (2006) Serine 202 regulates nuclear translocation of constitutive active/androstane receptor. Mol Pharmacol 69:1095–1102
Huang S, Chisari FV (1995) Strong, sustained hepatocellular proliferation precedes hepatocarcinogenesis in hepatitis B surface antigen transgenic mice. Hepatology 21:620–626
IARC (1997) Polychlorinated dibenzo-para-dioxins. In: IARC monographs on the evaluation of carcinogenic risks to humans, vol 69. IARC, Lyon, pp 33–343
IARC (1999) Hormonal contraception and post-menopausal hormone therapy. In: IARC monographs on the evaluation of carcinogenic risks to humans, vol 72. IARC, Lyon, pp 49–565
IARC (2001) Phenobarbital and its sodium salt. In: IARC monographs on the evaluation of carcinogenic risks to humans, vol 79. IARC, Lyon, pp 161–288
Jaworski M, Hailfinger S, Buchmann A, Hergenhahn M, Hollstein M, Ittrich C, Schwarz M (2005) Human p53 knock-in (hupki) mice do not differ in liver tumor response from their counterparts with murine p53. Carcinogenesis 26:1829–1834
Kekule AS, Lauer U, Weiss L, Luber B, Hofschneider PH (1993) Hepatitis B virus transactivator HBx uses a tumour promoter signalling pathway. Nature 361:742–745
Kinzler KW, Vogelstein B (1996) Lessons from hereditary colorectal cancer. Cell 87:159–170
Klaunig JE, Babich MA, Baetcke KP, Cook JC, Corton JC, David RM, Deluca JG, Lai DY, McKee RH, Peters JM, Roberts RA, Fenner-Crisp PA (2003) PPARα agonist induced rodent tumors: modes of action and human relevance. Crit Rev Toxicol 33:655–780
Knerr S, Schrenk D (2006) Carcinogenicity of ‘non-dioxinlike’ polychlorinated biphenyls. Crit Rev Toxicol 36:663–694
Kociba RJ, Keyes DG, Beyer JE, Carreon RM, Wade CE, Dittenber DA, Kalnins RP, Frauson LE, Park CN, Barnard SD, Hummel RA, Humiston CG (1978) Results of a two-year chronic toxicity and oncogenicity study of 2,3,7,8-tetrachlorodibenzo-p-dioxin in rats. Toxicol Appl Pharmacol 46:279–303
Lamminpää A, Pukkala E, Teppo L, Neuvonen PJ (2002) Cancer incidence among patients using antiepileptic drugs: a long-term follow-up of 28,000 patients. Eur J Clin Pharmacol 58:137–141
Laurent-Puig P, Legoix P, Bluteau O, Belghiti J, Franco D, Binot F, Monges G, Thomas G, Bioulac-Sage P, Zucman-Rossi J (2001) Genetic alterations associated with hepatocellular carcinomas define distinct pathways of hepatocarcinogenesis. Gastroenterology 120:1763–1773
Lee JS, Heo J, Libbrecht L, Chu IS, Kaposi-Novak P, Calvisi DF, Mikaelyan A, Roberts LR, Demetris AJ, Sun Z, Nevens F, Roskams T, Thorgeirsson SS (2006) A novel prognostic subtype of human hepatocellular carcinoma derived from hepatic progenitor cells. Nat Med 12:410–416
Levy L, Renard CA, Wie Y, Buendia MA (2002) Genetic alterations and oncogenic pathways in hepatocellular carcinoma. Ann N Y Acad Sci 963:21–36
Lieber CS (2001) Alcohol and hepatitis C. Alcohol Res Health 25:245–254
Lieber CS (2002) S-Adenosyl-l-methionine and acoholic liver disease in animal models: implications for early intervention in human beings. Alcohol 27:173–177
Lieber CS (2004) Alcoholic fatty liver: its pathogenesis and mechanism of progression to inflammation and fibrosis. Alcohol 34:9–19
Luebeck EG, Buchmann A, Stinchcombe S, Moolgavkar SH, Schwarz M (2000) Effects of 2,3,7,8-tetrachlorodibenzo-p-dioxin on initiation and promotion of GST-P-positive foci in rat liver: a quantitative analysis of experimental data using a stochastic model. Toxicol Appl Pharmacol 167:63–73
Moennikes O, Buchmann A, Romualdi A, Ott T, Werringloer J, Willeke K, Schwarz M (2000) Lack of phenobarbital-mediated promotion of hepatocarcinogenesis in connexin32-null mice. Cancer Res 60:5087–5091
Moennikes O, Loeppen S, Buchmann A, Andersson P, Ittrich C, Poellinger L, Schwarz M (2004) A constitutively active dioxin/aryl hydrocarbon receptor promotes hepatocarcinogenesis in mice. Cancer Res 64:4707–4710
Moolgavkar SH, Luebeck EG, Buchmann A, Bock KW (1996) Quantitative analysis of enzyme-altered liver foci in rats initiated with diethylnitrosamine and promoted with 2,3,7,8-tetrachlorodibenzo-p-dioxin or 1,2,3,4,6,7,8-heptachlorodibenzo-p-dioxin. Toxicol Appl Pharmacol 138:31–42
Müller O, Alonso A, Buchmann A, Bauer-Hofmann R, Bock KW, Schwarz M (1990) Detection of genomic alterations in carcinogen-induced mouse liver tumors by DNA fingerprint analysis. Mol Carcinog 3:330–334
Münzel P, Bock-Hennig B, Schieback S, Gschaidmeier H, Beck-Gschaidmeier S, Bock KW (1996) Growth modulation of hepatocytes and rat liver epithelial cells (WB-F344) by 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD). Carcinogenesis 17:197–202
Okey AB, Franc MA, Moffat ID, Tijet N, Boutros PC, Korkalainen M, Tuomisto M, Pohjanvirta R (2005) Toxicological implications of polymorphisms in receptors for xenobiotic chemicals: the case of the aryl hydrocarbon receptor. Toxicol Appl Pharmacol 207:S43–S51
Olsen JH, Schulgen G, Boice JD, Whysner J, Travis LB, Williams GM, Johnson FB, McGee JOD (1995) Antiepileptic treatment and risk for hepatobiliary cancer and malignant lymphoma. Cancer Res 55:294–297
Park KT, Mitchell KA, Huang G, Elferink CJ (2005) The aryl hydrocarbon receptor predisposes hepatocytes to Fas-mediated apoptosis. Mol Pharmacol 67:612–622
Parzefall W, Erber E, Sedivy R, Schulte-Hermann R (1991) Testing for induction of DNA synthesis in human hepatocyte primary cultures by rat liver tumor promoters. Cancer Res 51:1143–1147
Parzefall W, Berger W, Kainzbauer E, Teufelhofer O, Schulte-Hermann R, Thurman RG (2001) Peroxisome proliferators do not increase DNA synthesis in purified rat hepatocytes. Carcinogenesis 22:519–523
Pitot HC, Goldsworthy TL, Campbell HA, Poland A (1980) Quantitative evaluation of the promotion by 2,3,7,8-tetrachlorodibenzo-p-dioxin of hepatocarcinogenesis from diethylnitrosamine. Cancer Res 40:3616–3620
Pitot HC (1990) Altered hepatic foci: their role in murine hepatocarcinogenesis. Annu Rev Pharmacol Toxicol 30:465–500
Roberts RA, Ganey PE, Ju C, Kamendulis LM, Rusyn I, Klaunig JE (2007) Role of Kupffer cell in mediating hepatic toxicity and carcinogenesis. Toxicol Sci 96:2–15
Roncalli M, Bianchi P, Bruni B, Laghi L, Destro A, Di Gioia S, Gennari L, Tommasini M, Malesci A, Coggi G (2002) Methylation framework of cell cycle gene inhibitors in cirrhosis and associated hepatocellular carcinoma. Hepatology 36:427–432
Schrenk D, Schäfer S, Bock KW (1994) 2,3,7,8-tetrachlorodibenzo-p-dioxin as growth modulator in mouse hepatocytes with high and low affinity Ah receptor. Carcinogenesis 17:197–202
Schulte-Hermann R, Grasl-Kraupp B, Bursch W (1995) Apoptosis and hepatocarcinogenesis. In: Jirtle RL (ed) Liver regeneration and carcinogenesis. Academic Press, New York, pp 141–178
Schwarz M (1995) Tumor promotion in liver. Birkhäuser, Boston
Schwarz M, Pearson D, Port R, Kunz W (1984) Promoting effect of 4-dimethylaminoazobenzene on enzyme altered foci induced in rat liver by N-nitrosodiethanolamine. Carcinogenesis 5:725–730
Schwienbacher C, Gramentieri L, Scelfo R, Veronese A, Calin GA, Bolondi L (2000) Gain of imprinting at the chromosome 11p15: a pathogenic mechanism identified in human hepatocarcinomas. Proc Natl Acad Sci USA 97:5445–5449
Solt D, Farber E (1976) New principle for the analysis of chemical carcinogenesis. Nature 263:701–703
Stahl S, Ittrich C, Marx-Stölting P, Köhle C, Altug-Teber Ö, Riess O, Bonin M, Jobst J, Kaiser S, Buchmann A, Schwarz M (2005) Genotype–phenotype relationships in hepatocellular tumors from mice and man. Hepatology 42:353–361
Stinchcombe S, Buchmann A, Bock KW, Schwarz M (1995) Inhibition of apoptosis during 2,3,7,8-tetrachlorodibenzo-p-dioxin-mediated tumour promotion in rat liver. Carcinogenesis 16:1271–1275
Strathmann J, Schwarz M, Tharappel JC, Glauert HP, Spear BT, Robertson LW, Appel KE, Buchmann A (2006) PCB 153, a non-dioxin-like tumor promoter, selects for beta-catenin (Catnb)-mutated mouse liver tumors. Toxicol Sci 93:34–40
Thorgeirsson SS, Grisham JW (2002) Molecular pathogenesis of human hepatocellular carcinoma. Nat Genet 31:339–346
Unger C, Buchmann A, Buenemann CL, Kress S, Schwarz M (1998) Wild-type function of the p53 tumor suppressor protein is not required for apoptosis of mouse hepatoma cells. Cell Death Differ 5:87–95
Watson MA, Devereux TR, Malarkey DE, Anderson MW, Maronpot RR (1995) H-ras oncogene mutation spectra in B6C3F1 and C57BL/6 mouse liver tumors provide evidence for TCDD promotion of spontaneous and vinyl carbamate-initiated liver cells. Carcinogenesis 16:1705–1710
Williams GM (1997) Chemicals with carcinogenic activity in rodent liver; mechanistic evaluation of human risk. Cancer Lett 117:175–188
Xu XR, Huang J, Xu ZG, Qian BZ, Zhu ZD, Yan Q, Cai T, Zhang X, Xiao HS, Qu J, Liu F, Huang QH, Cheng ZH, Li NG, Du JJ, Hu W, Shen KT, Lu G, Fu G, Zhong M, Xu SH, Gu WY, Huang W, Zhao XT, Hu GX, Gu JR, Chen Z, Han ZG (2001) Insight into hepatocellular carcinogenesis at transcriptome level by comparing gene expression profiles of hepatocellular carcinoma with those of corresponding noncancerous liver. Proc Natl Acad Sci USA 98:15089–15094
Yager JD, Liehr JG (1996) Molecular mechanisms of estrogen carcinogenesis. Annu Rev Pharmacol Toxicol 36:203–232
Yamamoto Y, Moore R, Goldsworthy TL, Negishi M, Maronpot RR (2004) The orphan nuclear receptor constitutive active/androstane receptor is essential for liver tumor promotion by phenobarbital in mice. Cancer Res 64:7197–7200
Acknowledgments
We apologize for having often referred to reviews to keep the number of references to a minimum.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Köhle, C., Schwarz, M. & Bock, K.W. Promotion of hepatocarcinogenesis in humans and animal models. Arch Toxicol 82, 623–631 (2008). https://doi.org/10.1007/s00204-007-0273-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-007-0273-7