Abstract
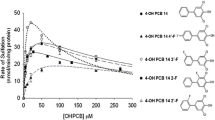
Quantitative structure–activity relationships (QSARs), which relate the glucuronidation of hydroxyl polychlorinated biphenyls (OH-PCBs)—catalyzed by the uridine diphosphate glucuronosyltransferases (UGTs)—to their physicochemical properties and molecular structural parameters, can be used to predict the rate constants and interpret the mechanism of glucuronidation. In this study, QSARs have been developed that use 23 semi-empirical calculated quantum chemical descriptors to predict the logarithms of the constants 1/Km and Vmax, related to enzyme kinetics. A partial least squares regression method was used to select the optimal set of descriptors to minimize the multicollinearity between the descriptors, as well as to maximize the cross-validated coefficient (Q2 cum) values. The key descriptors affecting log(1/Km) were Elumo− Ehomo (the energy gap between the lowest unoccupied molecular orbital and the highest occupied molecular orbital) and q −C (the largest negative net atomic charge on a carbon atom), while the key descriptors affecting logVmax were the polarizability α, the Connolly solvent-excluded volume (CSEV), and logP (the logarithm of the partition coefficient for octanol/water). From the results obtained it can be concluded that hydrophobic and electronic aspects of OH-PCBs are important in the glucuronidation of OH-PCBs.



Similar content being viewed by others
References
Berger U, Herzke D, Sandanger TM (2004) Two trace analytical methods for determination of hydroxylated PCBs and other halogenated phenolic compounds in eggs from Norwegian birds of prey. Anal Chem 76:441–452
Bock KW (2003) Vertebrate UDP-glucuronosyltransferases: functional and evolutionary aspects. Biochem Pharmacol 66:691–696
Brenner RC, Magar VS, Ickes JA, Foote EA, Abbott JE, Bingler LS, Crecelius EA (2004) Long-term recovery of PCB-contaminated surface sediments at the Sangamo-Weston/Twelvemile Creek/Lake Hartwell Superfund site. Environ Sci Technol 38:2328–2337
Brouwer A, Morse DC, Lans MC, Schuur AG, Murk AJ, Klasson-Wehler E, Bergman A, Visser T (1998) Interaction of persistent environmental organohalogens with the thyroid hormone system: mechanism and possible consequences for animal and human health. J Toxicol Ind Health 14:59–84
Campbell LM, Muir DCG, Whittle DM, Backus S, Norstrom RJ, Fisk AT (2003) Hydroxylated PCBs and other chlorinated phenolic compounds in Lake Trout (Salvelinus namaycush) blood plasma from the Great Lakes region. Environ Sci Technol 37:1720–1725
Cronin MTD, Schultz TW (2001) Development of quantitative structure–activity relationships for the toxicity of aromatic compounds to Tetrahymena pyriformis: comparative assessment of the methodologies. Chem Res Toxicol 14:1284–1295
Dimov D, Nedyalkova Z, Haladjova S, Schüürmann G, Mekenyan O (2001) QSAR modeling of antimycobacterial activity and activity against other bacteria of 3-formyl rifamycin SV derivatives. QSAR Comb Sci 20:298–318
Faucon JC, Bureau R, Faisant J, Briens F, Rault S (1999) Prediction of the fish acute toxicity from heterogeneous data coming from notification files. Chemosphere 38:3261–3276
Fischer LJ, Seegal RF, Ganey PE, Pessah IN, Kodavanti PRS (1998) Symposium overview: toxicity of non-coplanar PCBs. Toxicol Sci 41:49–61
Gobas FAPC, Wilcockson JB, Russell RW, Haffner GD (1999) Mechanism of biomagnification in fish under laboratory and field conditions. Environ Sci Technol 33:133–141
Hoffman B, Cho SJ, Zheng W, Wyrick S, Nichols DE, Mailman RB, Tropsha A (1999) Quantitative structure–activity relationship modeling of dopamine D1 antagonists using comparative molecular field analysis, genetic algorithms-partial least-squares, and k nearest neighbor methods. J Med Chem 42:3217–3226
Karelson M, Lobanov VS, Katritzky AR (1996) Quantum chemical descriptors in QSAR/QSPR studies. Chem Rev 96:1027–1043
King CD, Rios GR, Assouline JA, Tephly TR (1999) Expression of UDP-glucuronosyltransferases (UGTs) 2B7 and 1A6 in the human brain and identification of 5-hydroxytryptamine as a substrate. Arch Biochem Biophys 365:156–162
Letcher RJ, Norstrom RJ, Muir DCG (1998) Biotransformation versus bioaccumulation: sources of methyl sulfone PCB and 4,4′-DDE metabolites in the polar bear food chain. Environ Sci Technol 32:1656–1661
Li H, Drouillard KG, Bennett E, Haffner GD, Letcher RJ (2003) Plasma-associated halogenated phenolic contaminants in benthic and pelagic fish species from the Detroit river. Environ Sci Technol 37:832–839
Luco JM, Salinas AP, Torriero AAJ, Vazquez RN, Raba J, Marchevsky E (2003) Immobilized artificial membrane chromatography: quantitative structure–retention relationships of structurally diverse drugs. J Chem Inf Comput Sci 43:2129–2136
Mancera RL, Gómez AG, Pisanty A (1995) Quantitative structure–activity relationships of competitive inhibitors of phosphoenolpyruvate carboxylase. Bioorgan Med Chem 3:217–225
Miners JO, McKinnon RA, Mackenzie PI (2002) Genetic polymorphisms of UDP-glucuronosyltransferases and their functional significance. Toxicology 181–182:453–456
Muszynski IC, Scapozza L, Kovar KA, Folkers G (1999) Quantitative structure–activity relationships of phenyltropanes as inhibitors of three monoamine transporters: classical and CoMFA studies. QSAR Comp Sci 18:342–353
Netzeva TI,Dearden JC, Edwards R, Worgan ADP, Cronin MTD (2004) QSAR analysis of the toxicity of aromatic compounds to chlorella vulgaris in a novel short-term assay. J Chem Inf Comput Sci 44:258–265
Parr RG, Pearson RG (1983) Absolute hardness: companion parameter to absolute electronegativity. J Am Chem Soc 105:7512–7516
Pearson RG (1986) Absolute electronegativity and hardness correlated with molecular orbital theory. Proc Natl Acad Sci USA 83:8440–8441
Priolo N, Arribére CM, Caffin N, Barberis S, Vázquez RN, Lucod JM (2001) Isolation and purification of cysteine peptidases from the latex of Araujia hortorum fruits: study of their esterase activities using partial least-squares (PLS) modeling. J Mol Catal B Enzyme 15:177–189
Sacco JC, James M (2004) Glucuronidation in the polar bear (Ursus maritimus). Mar Environ Res 58:475–479
Soffers AEMF, Boersma MG, Vaes WHJ, Vervoort J, Tyrakowka B, Hermens JLM, Rietjens IMCM (2001) Computer-modeling-based QSARs for analyzing experimental data on biotransformation and toxicity. Toxicol In Vitro 15:539–551
Stanton DT (2003) On the physical interpretation of QSAR models. J Chem Inf Comput Sci 43:1423–1433
Svante W, Michael S, Lennart E (2001) PLS-regression: a basic tool of chemometrics. Chemom Intell Lab 58:109–130
Tampal N, Lehmler HJ, Espandiari P, Malmberg T, Robertson LW (2002) Glucuronidation of hydroxylated polychlorinated biphenyls (PCBs). Chem Res Toxicol 15:1259–1266
Waller CL (2004) A comparative QSAR study using CoMFA, HQSAR, and FRED/SKEYS paradigms for estrogen receptor binding affinities of structurally diverse compounds. J Chem Inf Comput Sci 44:758–765
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, D. The uridine diphosphate glucuronosyltransferases: quantitative structure–activity relationships for hydroxyl polychlorinated biphenyl substrates. Arch Toxicol 79, 554–560 (2005). https://doi.org/10.1007/s00204-005-0671-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-005-0671-7