Abstract
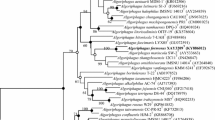
A Gram-negative, aerobic, non-motile, oxidase-positive, catalase-positive, methyl red-positive, and lipase-negative bacterium, designated A5.8T, was isolated from beach sediment of Zhairuo Island located in the East China Sea. Growth occurred at 10–40 °C (optimum, 30 °C), pH 5.5–9.5 (optimum, 7.5), and 0–2% NaCl (optimum, 1.5%). Based on 16S rRNA gene sequence analysis, strain A5.8T belongs to the genus Ancylobacter, sharing the highest similarity with Ancylobacter aquaticus JCM 20518T (98.0%). Its polar lipids mainly consist of phosphatidylethanolamine (PE) and phosphatidylcholine (PC). The predominant fatty acids are summed feature 8 (C18:1ω7c and/or C18:1ω6c, 91.0%), and the major respiratory quinone is Q-10. The DNA G + C content is 67.2 mol%. Based on above analysis, as well as digital DNA–DNA hybridization (22.5–22.9%) and average nucleotide identity (83.0–83.6%) of strain A5.8T with reference type strains of the genus Ancylobacter, strain A5.8T was suggested to represent a novel species of the genus Ancylobacter, for which the name Ancylobacter gelatini sp. nov. is proposed. The type strain is A5.8T (= MCCC 1K07167T = LMG 32566T).

Similar content being viewed by others
Abbreviations
- MM:
-
Mineral salt medium
- LB:
-
Luria–Bertani
- MCCC:
-
Marine Culture Collection of China
- BCCM/LMG:
-
Belgian Co-ordinated Collections of Microorganisms
- DSMZ:
-
Deutsche Sammlung von Mikroorganismen und Zellkulturen GmbH
- JCM:
-
Japan Collection of Microorganisms
- TEM:
-
Transmission electron microscopy
- NA:
-
Nutrient agar
- TSA:
-
Trypticase Soy agar
- R2A:
-
Reasoner's 2A agar
- MR-VP:
-
Methyl red and Voges-Proskauer
- dDDH:
-
Digital DNA–DNA hybridization
- ANI:
-
Average nucleotide identity
- GGDC:
-
Genome-to-Genome Distance Calculator
- MIDI:
-
Microbial Identification System
- HPLC:
-
High-performance liquid chromatography
- PE:
-
Phosphatidylethanolamine
- PC:
-
Phosphatidylcholine
- DPG:
-
Diphosphatidylglycerol
- PG:
-
Phosphatidylglycerol
- PL:
-
Phospholipid
- AL:
-
Aminolipid
- APL:
-
Aminophospholipid
- TLC:
-
Thin-layer chromatography
- MEGA:
-
Molecular evolutionary genetics analysis
References
Agafonova NV, Kaparullina EN, Trotsenko YA, Doronina NV (2017) Ancylobacter sonchi sp. nov., a novel methylotrophic bacterium from roots of Sonchus arvensis L. Int J Syst Evol Microbiol 67:4552–4558. https://doi.org/10.1099/ijsem.0.002330
Altschul SF, Gish W, Miller W, Myers EW, Lipman DJ (1990) Basic local alignment search tool. J Mol Biol 215:403–410. https://doi.org/10.1016/S0022-2836(05)80360-2
Auch AF, Klenk HP, Göker M (2010) Standard operating procedure for calculating genome-to-genome distances based on high-scoring segment pairs. Stand Genomic Sci 2:142–148. https://doi.org/10.4056/sigs.541628
Banik A, Mukhopadhaya SK, Dangar TK (2016) Characterization of N2-fixing plant growth promoting endophytic and epiphytic bacterial community of Indian cultivated and wild rice (Oryza spp.) genotypes. Planta 243:799–812. https://doi.org/10.1007/s00425-015-2444-8
Chemodurova AA, Kaparullina EN, Machulin AV, Sproer C, Lang E, Doronina NV (2020) Ancylobacter lacus sp. nov. and Ancylobacter plantiphilus sp. nov., novel aerobic facultative methylotrophic bacteria. Microbiology (n y) 89:35–43. https://doi.org/10.1134/S0026261720010051
Chun J, Rainey FA (2014) Integrating genomics into the taxonomy and systematics of the Bacteria and Archaea. Int J Syst Evol Microbiol 64:316–324. https://doi.org/10.1099/ijs.0.054171-0
Collins MD, Jones D (1981) Distribution of isoprenoid quinone structural types in bacteria and their taxonomic implications. Microbiol Rev 45:316–354. https://doi.org/10.1128/Mmbr.45.2.316-354.1981
Debnath SC, Chen C, Liu SX, Di YN, Zheng DQ, Li XY, Xu XW, Xu JZ, Wang PM (2019) Flavobacterium sharifuzzamanii sp. nov., isolated from the sediments of the East China Sea. Curr Microbiol 76:297–303. https://doi.org/10.1007/s00284-018-1609-7
Dong X, Cai M (2001) Determinative manual for routine bacteriology. Scientific Press, Beijing
Edgar RC (2004) MUSCLE: a multiple sequence alignment method with reduced time and space complexity. BMC Bioinform 5:113. https://doi.org/10.1186/1471-2105-5-113
Felsenstein J (1981) Evolutionary trees from DNA sequences: a maximum likelihood approach. J Mol Evol 17:368–376. https://doi.org/10.1007/Bf01734359
Felsenstein J (1985) Confidence limits on phylogenies: an approach using the bootstrap. Evolution 39:783–791. https://doi.org/10.2307/2408678
Firsova J, Doronina N, Lang E, Spröer C, Vuilleumier S, Trotsenko Y (2009) Ancylobacter dichloromethanicus sp. nov.—a new aerobic facultatively methylotrophic bacterium utilizing dichloromethane. Syst Appl Microbiol 32:227–232. https://doi.org/10.1016/j.syapm.2009.02.002
Goris J, Konstantinidis KT, Klappenbach JA, Coenye T, Vandamme P, Tiedje JM (2007) DNA–DNA hybridization values and their relationship to whole-genome sequence similarities. Int J Syst Evol Microbiol 57:81–91. https://doi.org/10.1099/ijs.0.64483-0
Jain C, Rodriguez-R LM, Phillippy AM, Konstantinidis KT, Aluru S (2018) High throughput ANI analysis of 90K prokaryotic genomes reveals clear species boundaries. Nat Commun 9:5114. https://doi.org/10.1038/s41467-018-07641-9
Kämpfer P, Kroppenstedt RM (1996) Numerical analysis of fatty acid patterns of coryneform bacteria and related taxa. Can J Microbiol 42:989–1005. https://doi.org/10.1139/m96-128
Komagata K, Suzuki K (1988) 4 Lipid and cell-wall analysis in bacterial systematics. Method Microbiol 19:161–207. https://doi.org/10.1016/S0580-9517(08)70410-0
Kumar S, Stecher G, Li M, Knyaz C, Tamura K (2018) MEGA X: molecular evolutionary genetics analysis across computing platforms. Mol Biol Evol 35:1547–1549. https://doi.org/10.1093/molbev/msy096
Lang E, Swiderski J, Stackebrandt E, Schumann P, Sproer C, Sahin N (2008) Description of Ancylobacter oerskovii sp. nov. and two additional strains of Ancylobacter polymorphus. Int J Syst Evol Microbiol 58:1997–2002. https://doi.org/10.1099/ijs.0.65666-0
Lu P, Jin L, Liang B, Zhang J, Li S, Feng Z, Huang X (2011) Study of biochemical pathway and enzyme involved in metsulfuron-methyl degradation by Ancylobacter sp. XJ-412-1 isolated from soil. Curr Microbiol 62:1718–1725. https://doi.org/10.1007/s00284-011-9919-z
Ma L, Du SJ, Gong Y, Du ZJ (2021) Robertkochia sediminum sp. nov., isolated from coastal sediment. Arch Microbiol 204:13. https://doi.org/10.1007/s00203-021-02605-y
Meier-Kolthoff JP, Auch AF, Klenk HP, Göker M (2013) Genome sequence-based species delimitation with confidence intervals and improved distance functions. BMC Bioinform 14:60. https://doi.org/10.1186/1471-2105-14-60
Minnikin DE, O’donnell AG, Goodfellow M, Alderson G, Athalye M, Schaal A, Parlett JH (1984) An integrated procedure for the extraction of bacterial isoprenoid quinones and polar lipids. J Microbiol Methods 2:233–241. https://doi.org/10.1016/0167-7012(84)90018-6
Nie W, Zheng X, Wang S, Ahmad I, Zhu B (2022) Genome resource of Ancylobacter pratisalsi E130T: a novel plant-growth-promoting bacterium isolated from the rhizosphere. Phytopathology 112:729–731. https://doi.org/10.1094/PHYTO-06-21-0251-A
Nurk S, Bankevich A, Antipov D, Gurevich A, Korobeynikov A, Lapidus A, Prjibelsky A, Pyshkin A, Sirotkin A and Sirotkin Y (2013) Assembling genomes and mini-metagenomes from highly chimeric reads. In: Annual international conference on research in computational molecular biology. Springer, pp 158–170
Ørskov J (1928) Beschreibung eines neuen Mikroben, Microcyclus aquaticus. mit eigentuemlicher Morphologie. Zentralbl Bakteriol Parasitenkd Infektionskr Hyg Abt I 107:180–184
Poroshina M, Doronina N, Kaparullina E, Kovalevskaya N, Trotsenko YA (2013) Halophilic and halotolerant aerobic methylobacteria from the technogenic Solikamsk biotopes. Microbiology (n y) 82:490–498. https://doi.org/10.1134/S0026261713040097
Qi YB, Wang CY, Lv CY, Lun ZM, Zheng CG (2017) Removal capacities of polycyclic aromatic hydrocarbons (PAHs) by a newly isolated strain from oilfield produced water. Int J Env Res Pub He 14:215. https://doi.org/10.3390/ijerph14020215
Raj HD (1983) Proposal of Ancylobacter gen. nov. as a substitute for the bacterial genus Microcyclus Ørskov 1928. Int J Syst Evol Microbiol 33:397–398. https://doi.org/10.1099/00207713-33-2-397
Raj HD (1981). The genus Microcyclus and related bacteria. In: Starr MP, Stolp H, Trüper HG, Balows A and Schlegel HG (ed) The prokaryotes: a Handbook on Habitats, Isolation, and Identification of Bacteria. Springer, New York, pp 630–644. https://doi.org/10.1007/978-3-662-13187-9_55
Rzhetsky A, Nei M (1992) A simple method for estimating and testing minimum-evolution trees. Mol Biol Evol 9:945–967
Saccardo P (1905) Pyrenomyceteae, Dothideaceae, Hyalodidymae, Microcyclus. Sylloge Fungorum omnium hucusque Cognitorum. Supplementum Universale 17:844–845
Saitou N, Nei M (1987) The neighbor-joining method—a new method for reconstructing phylogenetic trees. Mol Biol Evol 4:406–425. https://doi.org/10.1093/oxfordjournals.molbev.a040454
Sasser M (1990) Identification of bacteria by gas chromatography of cellular fatty acids, MIDI technical note 101. MIDI Inc, Newark
Suarez C, Ratering S, Schäfer J, Schnell S (2017) Ancylobacter pratisalsi sp. nov. with plant growth promotion abilities from the rhizosphere of Plantago winteri Wirtg. Int J Syst Evol Microbiol 67:4500–4506. https://doi.org/10.1099/ijsem.0.002320
Tamura K, Nei M, Kumar S (2004) Prospects for inferring very large phylogenies by using the neighbor-joining method. Proc Natl Acad Sci USA 101:11030–11035. https://doi.org/10.1073/pnas.0404206101
Tatusova T, DiCuccio M, Badretdin A, Chetvernin V, Nawrocki EP, Zaslavsky L, Lomsadze A, Pruitt KD, Borodovsky M, Ostell J (2016) NCBI prokaryotic genome annotation pipeline. Nucleic Acids Res 44:6614–6624. https://doi.org/10.1093/nar/gkw569
Urakami T, Komagata K (1986) Methanol-utilizing Ancylobacter strains and comparison of their cellular fatty-acid compositions and quinone systems with those of Spirosoma, Flectobacillus, and Runella species. Int J Syst Bacteriol 36:415–421. https://doi.org/10.1099/00207713-36-3-415
van den Wijngaard AJ, van der Kamp KW, van der Ploeg J, Pries F, Kazemier B, Janssen DB (1992) Degradation of 1,2-dichloroethane by Ancylobacter aquaticus and other facultative methylotrophs. Appl Environ Microbiol 58:976–983. https://doi.org/10.1128/aem.58.3.976-983.1992
van den Wijngaard AJ, Prins J, Smal AJ, Janssen DB (1993) Degradation of 2-chloroethylvinylether by Ancylobacter aquaticus AD25 and AD27. Appl Environ Microbiol 59:2777–2783. https://doi.org/10.1128/aem.59.9.2777-2783.1993
Xin YH, Zhou YG, Zhou HL, Chen WX (2004) Ancylobacter rudongensis sp nov., isolated from roots of Spartina anglica. Int J Syst Evol Microbiol 54:385–388. https://doi.org/10.1099/ijs.0.02466-0
Yoon SH, Ha SM, Kwon S, Lim J, Kim Y, Seo H, Chun J (2017) Introducing EzBioCloud: a taxonomically united database of 16S rRNA gene sequences and whole-genome assemblies. Int J Syst Evol Microbiol 67:1613–1617. https://doi.org/10.1099/ijsem.0.001755
Yu HX, Yu GZ, Wen XC (2006) Ancylobacter polymorphus sp. nov. and Ancylobacter vacuolatus sp. nov. Int J Syst Evol Microbiol 56:1185–1188. https://doi.org/10.1099/ijs.0.64118-0
Acknowledgements
We thank Dr. Xu Youping (Analysis Center of Agrobiology and Environmental Sciences, Faculty of Agriculture, Life and Environment Sciences, Zhejiang University) for fatty acid analysis.
Funding
This study was supported by a grant from Hainan Provincial Natural Science Foundation of China (321QN273) and Fundamental Research Funds for the Central Universities (2021XZZX018).
Author information
Authors and Affiliations
Contributions
PW, HS, and KZ performed the experiments and wrote the manuscript; YH and SCD isolated strains from the beach sediment sample; CY analyzed the polar lipids; KL and GC analyzed the genomic data; PW and JX guided the experiments and revised the manuscript; FW and ZG collected the beach sediment sample; ZG and DZ designed the study. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that there are no conflicts of interest.
Additional information
Communicated by Erko Stackebrandt.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Repositories: The GenBank/EMBL/DDBJ accession number for the 16S rRNA gene sequence of Ancylobacter gelatini (A5.8 T) is MW287270. The GenBank accession number for the genome sequence of Ancylobacter gelatini (A5.8 T) is JAKVIL000000000.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wang, P., Sheng, H., Zheng, K. et al. Ancylobacter gelatini sp. nov., isolated from beach sediment of Zhairuo Island, China. Arch Microbiol 204, 430 (2022). https://doi.org/10.1007/s00203-022-03048-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00203-022-03048-9