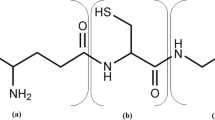
Abstract
The well-known probiotic GRAS Saccharomyces boulardii (CNCM I-745) was used for the first time to produce glutathione (GSH). The culture conditions affecting GSH biosynthesis were screened using a Plackett–Burman design (PBD). Analyzing the regression coefficients for 12 tested variables, yeast extract, glucose, peptone, cysteine, temperature and agitation rate had a positive significant effect on GSH production with a maximum yeild 192 mg/L. The impact of kinetics of adding cysteine was investigated in 19 experiments during the growth time course (0–36 h), and the maximum yield of glutathione (235 mg/L) was obtained by addition of cysteine after 8 h post-inoculation. The most significant variables were further explored at five levels using central composite rotatable design (CCRD), giving a maximum production of GSH (552 mg/L). Using baffled flasks, the yield of GSH was increased to 730 mg/L, i.e., 1.32-fold increment. The two rate-limiting genes of GSH biosynthesis “γ-glutamyl cysteine synthetase (GSH1) and GSH-synthetase (GSH2)” were amplified and sequenced to validate the GSH biosynthetic potency of S. boulardii. The sequences of genes showed 99% similarity with GSH1 and GSH2 genes of S. cerevisiae. Glutathione peroxidase was purified and characterized from S. boulardii with molecular mass and subunit structure of 80 kDa and 35 kDa as revealed from native and SDS-PAGE, ensuring its homodimeric identity. The activity of GPx was reduced by 2.5-fold upon demetallization confirming its metalloproteinic identity. The GPx was strongly inhibited by hydroxylamine and DTNB, ensuring the implication of surface lysine and cysteine residues on the enzyme active site domains.





Similar content being viewed by others
References
Abdel-Monem OA, El-Baz AF, Shetaia YM, El-Sabbagh SM (2012) Production and application of thermostable cellulase-free xylanase by aspergillus fumigatus from agricultural wastes. Ind Biotechn 8:152–161
Alfafara C, Miura K, Shimizu H, Shioya S, Suga KI (1992) Cysteine addition strategy for maximum glutathione production in fed-batch culture of Saccharomyces cerevisiae. Appl Microbiol Biotechnol 37:141–146
Bachhawat AK, Ganguli D, Kaur J, Kasturia N, Thakur A, Kaur H, Kumar A, Yadav A (2009) Glutathione production in Yeast. In: Satyanarayana T, Kunze G (eds) Yeast biotechnology: diversity and applications. Springer, Dordrecht, pp 259–280
Ballatori N, Krance SM, Notenboom S, Shi S, Tieu K, Hammond CL (2009) Glutathione dysregulation and the etiology and progression of human diseases. Biol Chem 390(3):191–214
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Brigelius-Flohé R, Maiorino M (2013) Glutathione peroxidases. Biochim Biophys Acta 1830:3289–3303
Cha JY, Park JC, Jeon BS, Lee YC, Cho YS (2004) Optimal fermentation conditions for enhanced glutathione production by Saccharomyces cerevisiae FF-8. J Microbiol Seoul 42(1):51–55
Cui X, Wan J, Zhang X, Wu H, Li Z, Ye Q (2019) Efficient glutathione production in metabolically engineered Escherichia coli strains using constitutive promoters. J Biotechnol 289:39–45
Dan LIU, Yiman QI, Ning ZH, Oufang CAO, Junnan XU, Mingtao FAN (2020) Multivariate analysis reveals effect of glutathione-enriched inactive dry yeast on amino acids and volatile components of kiwi wine. Food Chem 329:127086
Du LP, Hao RX, Xiao DG, Guo LL, Gai WD (2012) Research on the characteristics and culture conditions of Saccharomyces boulardii. Adv Mater Res 343:594–598 (Trans Tech Publications Ltd)
El Baz AF, Shetaia YM, Elkhouli RR (2011a) Xylitol production by Candida tropicalis under different statistically optimized growth conditions. Afr J Biotechnol 10(68):15353–15363
El Baz AF, Shetaia YM, Elkhouli RR (2011b) Kinetic behavior of Candida tropicalis during xylitol production using semi-synthetic and hydrolysate based media. Afr J Biotechnol 10:16617–16625
El Baz AF, Sorour NM, Shetaia YM (2016) Trichosporon jirovecii–mediated synthesis of cadmium sulfide nanoparticles. J Basic Microbiol 56(5):520–530
El Baz FN, Gamal RF, ElBaz AF, Ibrahim NE, ElMekawy A (2017) Biochemical and biotechnological studies on a novel purified bacillus cholesterol oxidase tolerant to solvent and thermal stress. Biocat Biotransform 35:205–214
El Baz AF, El-Enshasy HA, Shetaia YM, Mahrous H, Othman NZ, Yousef AE (2018) Semi-industrial scale production of a new yeast with probiotic traits, Cryptococcus sp. YMHS, isolated from the red sea. Probi Antimicrob Proteins 10(1):77–88
El Mekawy A, Hegab HM, El-Baz A, Hudson SM (2013) Kinetic properties and role of bacterial chitin deacetylase in the bioconversion of chitin to chitosan. Recent Patent Biotechnol 7:234–241
El-Sayed ASA, Ali GS (2020) Aspergillus flavipes is a novel efficient biocontrol agent of Phytophthora parasitica. Biol Control 40:104072
El-Sayed ASA, Abdel-Azim S, Ibrahim H, Yassin MA, Abdel-Ghany S, Esener S, Ali GS (2015) Biochemical stability and molecular dynamic characterization of Aspergillus fumigatus cystathionine γ-Lyase in response to various reaction effectors. Enzyme Microb Technol 81:31–46
El-Sayed ASA, Hassan AEA, Shindia AA, Mohamed SG, Sitohy MZ (2016) Aspergillus flavipes methionine γ-lyase dextran conjugates with enhanced structural proteolytic stability and anticancer efficiency. J Mol Catal B-Enzym 133:S15–S24
El-Sayed ASA, Shindia AA, Abou-Zaid AA, Yassin AM (2019a) Aspergillus nidulans thermostable arginine deiminase- Dextran conjugates with enhanced molecular stability, proteolytic resistance, pharmacokinetic properties and anticancer activity. Enz Microb Technol 131(12):109432
El-Sayed ASA, Ali DMI, Yassin MA, Zayed RW, Ali GS (2019b) Sterol inhibitor “Fluconazole” enhance the Taxol yield and molecular expression of its encoding genes cluster from Aspergillus flavipes. Process Biochem 76:55–67
Fietto JL, Araújo RS, Valadão FN, Fietto LG, Brandão RL, Neves M, Castro M (2004) Molecular and physiological comparisons between Saccharomyces cerevisiae and Saccharomyces boulardii. Can J Microbiol 50(8):615–621
Ge X, Rao G (2012) Real-time monitoring of shake flask fermentation and off gas using triple disposable noninvasive optical sensors. Biotechnol Prog 28:872–877
Hassan A, Sorour NM, El Baz AF, Shetaia YM (2019) Simple synthesis of bacterial cellulose/magnetite nanoparticles composite for the removal of antimony from aqueous solution. Int J Environ Sci Technol 16(3):1433–1448
Hong JY, Son SH, Hong SP, Yi SH, Kang SH, Lee NK, Paik HD (2019) Production of β-glucan, glutathione, and glutathione derivatives by probiotic Saccharomyces cerevisiae isolated from cucumber jangajji. LWT 100:114–118
Huang CS, He W, Meister A, Anderson ME (1995) Amino acid sequence of rat kidney glutathione synthetase. Proc Natl Acad Sci 92(4):1232–1236
Johnston RB, Bloch K (1951) Enzymatic synthesis of glutathione. J Biol Chem 188:221–240
Kebeish R, El-Sayed ASA, Fahmy H, Abdel-Ghany A (2016) Molecular cloning, biochemical characterization, and antitumor properties of a novel L-asparaginase from Synechococcus elongates PCC6803. Biochem 81:1173–1181
Laemmli U (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Lei Z, Chen H, Huang D, Zhai Y, Shu G (2016) Optimization of medium compositions for Saccharomyces Boulardii by Box-Behnken design. Sci Study Res Chem Chem Engin Biotechnol Food Ind 17(4):405
Li S, Yan T, Yang J-Q, Oberley TD, Oberley LW (2000) The role of cellular glutathione peroxidase redox regulation in the suppression of tumor cell growth by manganese superoxide dismutase. Cancer Res 60:3927–3939
Li Y, Wei G, Chen J (2004) Glutathione: a review on biotechnological production. Appl Microbiol Biotechnol 66:233–242
Li C, Xia JY, Chu J, Wang YH, Zhuang YP, Zhang SL (2013) CFD analysis of the turbulent flow in baffled shake flasks. Biochem Eng J 70:140–150
Liang G, Liao X, Du G, Chen J (2009) A new strategy to enhance glutathione production by multiple H2O2-induced oxidative stresses in Candida utilis. Bioresour Technol 100:350–355
Lienkamp AC, Heine T, Tischler D (2020) Glutathione: a powerful but rare cofactor among actinobacteria. Advances Appl Microbiol 110:181–217
Liu L, Mao SZ, Liu XM, Huang X, Xu JY, Liu JQ, Luo GM, Shen JC (2008) Functional mimicry of the active site of glutathione peroxidase by glutathione imprinted selenium-containing protein. Biomacromolecules 9(1):363–368
Lorenz E, Schmacht M, Stahl U, Senz M (2015) Enhanced incorporation yield of cysteine for glutathione overproduction by fed-batch fermentation of Saccharomyces cerevisiae. J Biotechnol 216:131–139
McFarland LV, Bernasconi P (1993) Saccharomyces boulardii’. A review of an innovative biotherapeutic agent. Microb Ecol Health Dis 6(4):157–171
Meister A, Anderson ME (1983) Glutathione. Annu Rev Biochem 52(1):711–760
Mutoh N, Nakagawa CW, Hayashi Y (1995) Molecular cloning and nucleotide sequencing of the γ-glutamylcysteine synthetase gene of the fission yeast Schizosaccharomyces pombe. J Biochem 117(2):283–288
Myers RH, Montgomery DC, Anderson-Cook CM (2016) Response surface methodology: process and product optimization using designed experiments. John Wiley & Sons, New York
Shi C, Wang X, Xiao Z, Wang R, Qiao Y, Kan G (2020) Cloning, characterization and expression analysis of glutathione S-transferase from the Antarctic yeast Rhodotorula mucilaginosa AN5. Protein Expr Purif 167:105518
Takahashi M, Aoyagi H (2020) Analysis and effect of conventional flasks in shaking culture of Escherichia coli. AMB Expr 10:77
Tamura K, Peterson D, Peterson N, Stecher G, Nei M, Kumar S (2011) MEGA5: molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol Biol Evol 28(10):2731–2739
Tietze F (1969) Enzymic method for quantitative determination of nanogram amounts of total and oxidized glutathione: applications to mammalian blood and other tissues. Anal Biochem 27:502–522
Udeh KO, Achremowicz B (1997) High-glutathione containing yeast Saccharomyces cerevisiae: optimization of production. Acta Microbiol Polonica 46(1):105–114
Wang C, Zhang J, Wu H, Li Z, Ye Q (2015) Heterologous gshF gene expression in various vector systems in Escherichia coli for enhanced glutathione production. J Biotechnol 214:63–68
Wang D, Wang C, Wu H, Li Z, Ye Q (2016) Glutathione production by recombinant Escherichia coli expressing bifunctional glutathione synthetase. J Ind Microbiol Biotechnol 43(1):45–53
Wen S, Zhang T, Tan T (2005) Optimization of the amino acid composition in glutathione fermentation. Process Biochem 40(11):3474–3479
Yoshida H, Hara KY, Kiriyama K, Nakayama H, Okazaki F, Matsuda F, Kondo A (2011) Enzymatic glutathione production using metabolically engineered Saccharomyces cerevisiae as a whole-cell biocatalyst. Appl Microbiol Biotechnol 91(4):1001–1006
Zhang T, Wen S, Tan T (2007) Optimization of the medium for glutathione production in Saccharomyces cerevisiae. Process Biochem 42(3):454–458
Zhang J, Quan C, Wang C, Wu H, Li Z, Ye Q (2016) Systematic manipulation of glutathione metabolism in Escherichia coli for improved glutathione production. Microb Cell Fact 15(1):38
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
All authors declare that there are no financial/commercial conflicts of interest.
Additional information
Communicated by Erko Stackebrandt.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.


203_2021_2584_MOESM3_ESM.jpg
Supplementary Fig. S2 The phylogenetic analysis of Gsp1p (A) and Gsp2p (B) genes sequence of the Egy-B1 isolate along with their amino acid sequences retrieved from NCBI using MEGA 5 with neighbor-joining method using 1000 bootstrap replicates (JPG 180 KB)
Rights and permissions
About this article
Cite this article
Badr, H., El-Baz, A., Mohamed, I. et al. Bioprocess optimization of glutathione production by Saccharomyces boulardii: biochemical characterization of glutathione peroxidase. Arch Microbiol 203, 6183–6196 (2021). https://doi.org/10.1007/s00203-021-02584-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00203-021-02584-0