Abstract
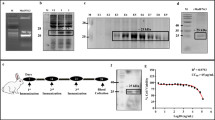
We previously constructed an arabinose-inducible recombinant Bifidobacterium longum that could efficiently express secreted IFN-α2b in vitro (Deng et al. in Arch Microbiol 191:681–686, 2009). Here, we investigated the influence of oral pBAD-SPIFN-transformed B. longum on immunomodulation and IFN-induced Mx1 gene transcription in mice. We observed enhanced serum and fecal IFN-α2b concentrations in mice orally administered recombinant B. longum, suggesting a possible Th1 pattern of induction in the spleen and Peyer’s patches. Transcription of the typically IFN-induced antiviral Mx1 gene in the hepatic and intestinal tissues of these mice was also markedly enhanced. In conclusion, oral administration of the recombinant B. longum expressing IFN-α2b might play its roles in the immunomodulation of the mice, and the potential clinical value of this bacterium in the treatment of viral infections needs to be further studied.


Similar content being viewed by others
References
Alignani D, Maletto B, Liscovsky M, Rópolo A, Morón G, Pistoresi-Palencia MC (2005) Orally administered OVA/CpG-ODN induces specific mucosal and systemic immune response in young and aged mice. J Leukocyte Biol 77:898–905
Bollati-Fogolín M, Müller W (2005) Virus free, cell-based assay for the quantification of murine type I interferons. J Immunol Methods 306:169–175
Brod SA (2002) Ingested type I interferon: a potential treatment for autoimmunity. J Interferon Cytokine Res 22:1153–1166
Brod SA, Burns DK (1994) Suppression of relapsing experimental autoimmune encephalomyelitis in the SJL/J mouse by oral administration of Type I Interferons. Neurology 44:1144–1148
Cummins JM, Krakowka GS, Thompson CG (2005) Systemic effects of interferons after oral administration in animals and humans. Am J Vet Res 66:164–176
David RL (2007–2008) CRC handbook of chemistry and physics. In: 88th edn, CRC Press, Boca Raton, pp 110
Dec M, Puchalski A (2008) Use of oromucosally administered interferon-alpha in the prevention and treatment of animal diseases. Pol J Vet Sci 11:175–186
Deng Q, Zeng W, Yu Z (2009) Signal peptide of Arabinosidase enhances secretion of interferon-alpha2b protein by Bifidobacteria longum. Arch Microbiol 191:681–686
Eid P, Meritet JF, Maury C, Lasfar A, Weill D, Tovey MG (1999) Oromucosal interferon therapy: pharmacokinetics and pharmacodynamics. J Interferon Cytokine Res 19:157–169
Forde KA, Reddy KR (2009) Hepatitis C virus infection and immunomodulatory therapies. Clin Liver Dis 13:391–401
Hu B, Kou L, Li C, Zhu LP, Fan YR, Wu ZW, Wang JJ, Xu GX (2009) Bifidobacterium longum as a delivery system of TRAIL and endostatin cooperates with chemotherapeutic drugs to inhibit hypoxic tumor growth. Cancer Gene Ther 16:655–663
Jørgensen JB, Johansen A, Hegseth MN, Zou J, Robertsen B, Collet B, Secombes CJ (2007) A recombinant CHSE-214 cell line expressing an Mx1 promoter-reporter system responds to both interferon type I and type II from salmonids and represents a versatile tool to study the IFN-system in teleost fish. Fish Shellfish Immunol 23:1294–1303
Kiliç H, Zeytin HE, Korkmaz C, Mat C, Gül A, Coşan F, Dinç A, Simşek I, Süt N, Yazici H (2009) Low-dose natural human interferon-alpha lozenges in the treatment of Behçet’s syndrome. Rheumatology (Oxford) 48:1388–1391
Meager A (2002) Biological assays for interferons. J Immunol Methods 261:21–36
Meijer JM, Pijpe J, Bootsma H, Vissink A, Kallenberg CG (2007) The future of biologic agents in the treatment of Sjögren’s syndrome. Clin Rev Allergy Immunol 32:292–297
Pachner AR, Bertolotto A, Deisenhammer F (2003) Measurement of MxA mRNA or protein as a biomarker of IFNbeta bioactivity: detection of antibody-mediated decreased bioactivity (ADB). Neurology 61(9 Suppl 5):S24–S26
Petry H, Cashion L, Szymanski P, Ast O, Orme A, Gross C, Bauzon M, Brooks A, Schaefer C, Gibson H, Qian H, Rubanyi GM, Harkins RN (2006) Mx1 and IP-10: biomarkers to measure IFN-beta activity in mice following gene-based delivery. J Interferon Cytokine Res 26:699–705
Polman C, Barkhof F, Kappos L, Pozzilli C, Sandbrink R et al (2003) Oral interferon beta-1a in relapsing -remitting multiple sclerosis: a double-blind randomized study. Mult Scler 9:342–348
Reyes Escogido ML, De León Rodríguez A, Barba de la Rosa AP (2007) A novel binary expression vector for production of human IL-10 in Escherichia coli and Bifidobacterium longum. Biotechnol Lett 29:1249–1253
Shkoporov AN, Efimov BA, Khokhlova EV, Kafarskaia LI, Smeianov VV (2008) Production of human basic fibroblast growth factor (FGF-2) in Bifidobacterium breve using a series of novel expression/secretion vectors. Biotechnol Lett 30:1983–1988
Tang W, He Y, Zhou S, Ma Y, Liu G (2009) A novel Bifidobacterium infantis-mediated TK/GCV suicide gene therapy system exhibits antitumor activity in a rat model of bladder cancer. J Exp Clin Cancer Res 28:155
Tovey MG, Maury C (1999) Oral mucosal interferon therapy: marked antiviral and antitumor activity. J Interferon Cytokine Res 19:145–155
Tupasi TE, Co VM, Clarin MS, Alesna ET, Divinagracia EM, Mangubat NV (2002) Randomized, double-blind, placebo-controlled trial of oromucosal low-dose interferon following prednisone withdrawal for chronic hepatitis B infection in Filipino patients. Int J Infect Dis 6:37–41
Yazawa K, Fujimori M, Nakamura T, Sasaki T, Amano J, Kano Y, Taniguchi S (2001) Bifidobacterium longum as a delivery system for gene therapy of chemically induced rat mammary tumors. Breast Cancer Res Treat 66:165–170
Acknowledgments
This work was supported by two grants from Shenzhen scientific Research Program of the People’s Republic of China (No. 200801020 and No. 200903277) and also supported by a grant from Ministry of Education of China (No. 200901121).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Communicated by Jorge Membrillo-Hernández.
Rights and permissions
About this article
Cite this article
Yu, Z., Zeng, Z., Huang, Z. et al. Increased mRNA expression of interferon-induced Mx1 and immunomodulation following oral administration of IFN-α2b-transformed B. longum to mice. Arch Microbiol 192, 633–638 (2010). https://doi.org/10.1007/s00203-010-0589-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00203-010-0589-1