Abstract
Aim
The effects of parathyroid hormone (PTH) alone or in combination with antiresorptive therapy on changes in bone mineral density (BMD) and fracture risk were studied.
Materials and methods
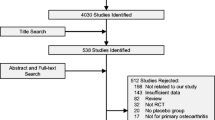
Randomised placebo controlled trials were retrieved from the PubMed, Web of Science or Embase databases.
Results
PTH alone or in combination with antiresorptive drugs reduced vertebral [relative risk (RR)=0.36, 95% confidence interval (CI): 0.28–0.47, 2p<0.01] and non-vertebral (RR=0.62, 95% CI: 0.48–0.82, 2p<0.01) fracture risk and increased spine BMD by 6.6% (95% CI: 5.2–8.1%, 2p<0.01) and hip BMD non-significantly by 1.0% (95% CI: −0.1 to 2.1%, 2p=0.08) during 11–36 months of follow-up (13 trials). The gain in spine and hip BMD tended to increase with the length of the PTH treatment. No significant effect of study duration on fracture risk could be demonstrated. The major adverse events were hypercalcaemia, nausea and discomfort at the injection sites. Only limited data are currently available on fracture risk reduction with PTH plus antiresorptive therapies.
Conclusion
Although the number of studies on non-vertebral fractures is limited, our pooled analysis revealed that PTH alone or in combination with antiresorptive drugs would appear to be able to reduce the risk of vertebral and non-vertebral fractures and to increase spine and perhaps hip BMD. However, these analyses were based on cross-sectional data – i.e. based on indirect comparisons – and further studies with a direct comparison of study duration are necessary. No studies comparing PTH, PTH plus antiresorptive drugs and antiresorptive drug versus placebo in a factorial design are available; consequently, we were unable to draw any conclusions on the superiority of PTH plus antiresorptive drug versus antiresorptive drug or PTH alone with respect to BMD or fractures.


Similar content being viewed by others
References
Johnell O (1997) The socioeconomic burden of fractures: today and in the 21st century. Am J Med 103:20S–25S
Kannus P, Niemi S, Parkkari J et al (1999) Hip fractures in Finland between 1970 and 1997 and predictions for the future (see comments). Lancet 353:802–805
Meunier PJ, Slosman DO, Delmas PD et al (2002) Strontium ranelate: dose-dependent effects in established postmenopausal vertebral osteoporosis – a 2-year randomized placebo controlled trial. J Clin Endocrinol Metab 87:2060–2066
Meunier PJ, Roux C, Seeman E et al (2004) The effects of strontium ranelate on the risk of vertebral fracture in women with postmenopausal osteoporosis. N Engl J Med 350:459–468
Neer RM, Arnaud CD, Zanchetta JR et al (2001) Effect of parathyroid hormone (1–34) on fractures and bone mineral density in postmenopausal women with osteoporosis. N Engl J Med 344:1434–1441
Orwoll ES, Scheele WH, Paul S et al (2003) The effect of teriparatide [human parathyroid hormone (1-34)] therapy on bone density in men with osteoporosis. J Bone Miner Res 18:9–17
Reginster JY, Meunier PJ (2003) Strontium ranelate phase 2 dose-ranging studies: PREVOS and STRATOS studies. Osteoporos Int 14:56–65
Reginster JY, Seeman E, de Vernejoul MC et al (2005) Strontium ranelate reduces the risk of non-vertebral fractures in postmenopausal women with osteoporosis: Treatment of Peripheral Osteoporosis (TROPOS) Study. J Clin Endocrinol Metab 90:2816–2822
Cranney A, Guyatt G, Krolicki N et al (2001) A meta-analysis of etidronate for the treatment of postmenopausal osteoporosis. Osteoporos Int 12:140–151
Cranney A, Wells G, Willan A et al (2002) Meta-analysis of alendronate for the treatment of postmenopausal women. Endocr Rev 23:508–516
Cranney A, Tugwell P, Adachi J et al (2002) Meta-analysis of risedronate for the treatment of postmenopausal osteoporosis. Endocr Rev 23:517–523
Cranney A, Tugwell P, Zytaruk N et al (2002) Meta-analysis of raloxifene for the prevention and treatment of postmenopausal osteoporosis. Endocr Rev 23:524–528
Torgerson DJ, Bell-Syer SEM (2001) Hormone replacement therapy and prevention of non-vertebral fractures. A meta-analysis of randomized trials. JAMA 285:2891–2897
Torgerson DJ, Bell-Syer SEM (2001) Hormone replacement therapy and prevention of vertebral fractures: a meta-analysis of randomised trials. BMC Musculoskelet Disord 2:7–10
Cranney A, Tugwell P, Zytaruk N et al (2002) Meta-analysis of calcitonin for the treatment of postmenopausal osteoporosis. Endocr Rev 23:540–551
Papadimitropoulos E, Wells G, Shea B et al (2002) Meta-analysis of the efficacy of vitamin D treatment in preventing osteoporosis in postmenopausal women. Endocr Rev 23:560–569
Shea B, Wells G, Cranney A et al (2002) Meta-analysis of calcium supplementation for the prevention of postmenopausal osteoporosis. Endocr Rev 23:552–559
Shea B, Wells G, Cranney A et al (2004) Calcium supplementation on bone loss in postmenopausal women (Review). Cochrane Database Syst Rev CD004526
Bischoff-Ferrari HA, Willett WC, Wong JB et al (2005) Fracture prevention with vitamin D supplementation: a meta-analysis of randomized controlled trials. JAMA 293:2257–2264
Greenspan SL, Bone HG, Marriott TB et al (2005)Preventing the first vertebral fracture in postmenopausal women with low bone mass using PTH(1–84): results from the TOP study (Abstract). J Bone Miner Res 20:S56
Lindsay R, Nieves J, Formica C et al (1997) Randomised controlled study of effect of parathyroid hormone on vertebral-bone mass and fracture incidence among postmenopausal women on oestrogen with osteoporosis. Lancet 350:550–555
Black DM, Bilezikian JP, Ensrud KE et al (2005) One year of alendronate after one year of parathyroid hormone (1-84) for osteoporosis. N Engl J Med 353:555–565
Black DM, Greenspan SL, Ensrud KE et al (2003) The effects of parathyroid hormone and alendronate alone or in combination in postmenopausal osteoporosis. N Engl J Med 349:1207–1215
Cosman F, Nieves J, Woelfert L et al (2001) Parathyroid hormone added to established hormone therapy: effects on vertebral fracture and maintenance of bone mass after parathyroid hormone withdrawal. J Bone Miner Res 16:925–931
Cosman F, Nieves J, Zion M et al (2005) Daily and cyclic parathyroid hormone in women receiving alendronate. N Engl J Med 353:566–575
Finkelstein JS, Klibanski A, Arnold AL et al (1998) Prevention of estrogen deficiency-related bone loss with human parathyroid hormone(1-34). JAMA 280:1067–1073
Finkelstein JS, Arnold AL (1999) Increases in bone mineral density after discontinuation of daily human parathyroid hormone and gonadotropin-releasing hormone analog administration in women with endometriosis. J Clin Endocrinol Metab 84:1214–1219
Finkelstein JS, Hayes A, Hunzelman JL et al (2003) The effects of parathyroid hormone, alendronate, or both in men with osteoporosis. N Engl J Med 349:1216–1226
Jadad AR, Moore RA, Carroll D et al (1996) Assessing the quality of reports of randomized clinical trials: is blinding necessary? Control Clin Trials 17:1–12
Böhning D (2000) Computer-assisted analysis of mixtures and applications: meta-analysis, disease mapping and others. Chapman&Hall/CRC, Boca Raton
Bucher HC, Guyatt GH, Griffith LE, Walter SD (1997) The results of direct and indirect treatment comparisons in meta-analysis of randomized controlled trials. J Clin Epidemiol 50:683–691
Hodsman AB, Hanley DA, Ettinger MP et al (2003) Efficacy and safety of human parathyroid hormone-(1-84) in increasing bone mineral density in postmenopausal osteoporosis. J Clin Endocrinol Metab 88:5212–5220
Kurland ES, Cosman F, McMahon DJ et al (2000) Parathyroid hormone as a therapy for idiopathic osteoporosis in men: effects on bone mineral density and bone markers. J Clin Endocrinol Metab 85:3069–3076
Genant HK, Halse J, Briney WG et al (2005) The effects of teriparatide on the incidence of back pain in postmenopausal women with osteoporosis. Curr Med Res Opin 21:1027–1034
Lundkvist J, Johnell O, Cooper C, Sykes D (2006) Economic evaluation of parathyroid hormone (PTH) in the treatment of osteoporosis in postmenopausal women. Osteoporos Int 17:201–211
Rosner AJ, Grima DT, Torrance GW et al (1998) Cost effectiveness of multi-therapy treatment strategies in the prevention of vertebral fractures in postmenopausal women with osteoporosis. Pharmacoeconomics 14:559–573
Liu H, Michaud K, Nayak S et al (2006) The cost-effectiveness of therapy with teriparatide and alendronate in women with severe osteoporosis. Arch Intern Med 166:1209–1217
Delmas PD, Li Z, Cooper C (2004) Relationship between changes in bone mineral density and fracture risk reduction with antiresorptive drugs: some issues with meta-analyses. J Bone Miner Res 19:330–337
Delmas PD, Seeman E (2004) Changes in bone mineral density explain little of the reduction in vertebral or non-vertebral fracture risk with anti-resorptive therapy. Bone 34:599–604
Wehren LE, Hosking D, Hochberg MC (2004) Putting evidence-based medicine into clinical practice: comparing anti-resorptive agents for the treatment of osteoporosis. Curr Med Res Opin 20:525–531
Kanis JA, Johnell O, De Laet C et al (2004) A meta-analysis of previous fracture and subsequent fracture risk. Bone 35:375–382
Lane NE, Sanchez S, Modin GW et al (1998) Parathyroid hormone treatment can reverse corticosteroid-induced osteoporosis. Results of a randomized controlled clinical trial. J Clin Invest 102:1627–1633
Body JJ, Gaich GA, Scheele WH et al (2002) A randomized double-blind trial to compare the efficacy of teriparatide [recombinant human parathyroid hormone (1–34)] with alendronate in postmenopausal women with osteoporosis. J Clin Endocrinol Metab 87:4528–4535
McClung MR, San Martin J, Miller PD et al (2005) Opposite bone remodeling effects of teriparatide and alendronate in increasing bone mass. Arch Intern Med 165:1762–1768
Acknowledgement
Research librarian ms. Edith Clausen is acknowledged for skillful help with the references.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Vestergaard, P., Jorgensen, N.R., Mosekilde, L. et al. Effects of parathyroid hormone alone or in combination with antiresorptive therapy on bone mineral density and fracture risk – a meta-analysis. Osteoporos Int 18, 45–57 (2007). https://doi.org/10.1007/s00198-006-0204-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-006-0204-0