Abstract
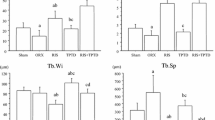
Osteoporosis is a disease of excess bone fragility that results from both the loss of bone mass and trabecular bone microarchitecture, thereby creating a very fragile skeleton. The purpose of this study was to determine whether treatment of ovariectomized (OVX) osteopenic rats with basic fibroblast growth factor (bFGF) would stimulate the production of new trabeculae, and whether the newly formed trabeculae would make physical connections with the pre-existing trabeculae after prolonged estrogen deficiency. Six-month-old Sprague Dawley rats were OVXed or sham-operated and were left untreated until day 60 post-OVX. A high resolution microscopic scan (XTM) of the right proximal tibia was performed on groups 1 and 2 on day 1 post-OVX, and was repeated in all animals on day 60 post-OVX. At day 60 groups 1 and 2 were treated with vehicle and groups 3 to 6 were injected with bFGF 200ug/kg/d intravenously for 15 days. At day 82, all animals obtained another in vivo XTM scan of the right tibia; then group 4 were treated with 17B estradiol 10ug/kg/3x a week, group 5 were treated with hPTH (1–34) at 80ug/kg/d for 35 days, group 6 were sacrificed, and groups 1 and 2 were treated with vehicle injections for 35 days. At day 110, all remaining animals were sacrificed, and repeat ex vivo XTM scans of the right proximal tibia were performed. Trabecular bone structural variables—including trabecular bone volume, connectivity, number, and thickness—were obtained from all XTM scans. Biochemical markers of bone turnover were also obtained 24 hours before each XTM scan (osteocalcin and deoxypyridinoline), and analyzed by ELISA. Animals OVXed and treated with vehicle had decreased trabecular bone volume, connectivity and number compared to sham-operated animals at both day 60 and day 110. Animals treated with bFGF from day 60–75 post-OVX had evidence of new trabeculae that physically connected with pre-existing trabeculae and also of increased trabecular bone volume seven days after the injections were discontinued. Biochemical markers of bone formation had a small and insignificant increase over baseline levels during the bFGF injections. Bone resorption markers were significantly reduced during the injection period, but returned to baseline levels after the injections were stopped. In addition, we also demonstrated that these newly formed trabecular connections could be maintained or added to with either estrogen or hPTH (1–34) treatments. Thirty-five days after ending the bFGF treatment, trabecular bone volume and connectivity was 25–80% higher in the estrogen and hPTH (1–34) treated animals compared to the untreated animals ( p <0.01). These results support continued development of bFGF as a potential treatment for severely osteoporotic individuals.


Similar content being viewed by others
References
Bouillon R, Burkhardt P, Christiansen C, Fleisch HA, Fujita T, Gennari C, Martin TJ, Mazzuoli G, Melton LJ, Ringe JD, et al. (1991) Consensus Development Conference: Prophylaxis and treatment of osteoporosis. Osteoporosis Int 1:114–117
Cummings SR, Kelsey JL, Nevitt MC, O'Dowd KJ (1985) Epidemiology of osteoporosis and osteoporotic fractures. Epidemiol Rev 7:178–208
Lindsay R, Hart DM, Clark DM (1984) The minimal effective dose of estrogen for prevention of postmenopausal bone loss. Obstet Gynecol 63:759–764
Chow J, Tobias H, Coston KW, Chambers TJ (1992) Estrogen maintains trabecular bone volume in rats not only by suppression of bone resorption but also by stimulation of bone formation. J Clin Invest 89:74–78
Lane NE, Haupt D, Kimmel DB, Modin G, Kinney JH (1999) Early estrogen replacement therapy reverses the rapid loss of trabecular bone volume and prevents further deterioration of connectivity in the rat. J Bone Miner Res 282:637–645
Black DB, Cummings SR, Karpt DB, Cauley JA, Thompson DE, Nevitt MC, Bauer DC, Genant HK, Haskett WJ, Marcus M, Ott SM, Torner JC, Quandt SA, Reiss TF, Ensrud KE (1996) Randomized trial of effect of alendronate on risk of fracture in women with existing vertebral fractures. Lancet 348:1535-1541
Chesnut CH, Silverman S, Andriano K, Genant HK, Gimona A, Harris S, Kiel D, LeBoff M, Maricic M, Miller P, Moniz C, Peacock M, Richardson P, Watts N, Baylink D for PROOF Study Investigators (2000) A randomized trial of nasal spray salmon calcitonin in postmenopausal women with established osteoporosis : 'The prevent recurrence of osteoporotic fracture studies'. Amer J Med 109:267–276
Delmas PD, Bjarnason NH, Mitlak BH, et al. (1996) Effects of raloxifene on bone mineral density, serum cholesterol concentrations, and uterine endometrium in postmenopausal women. N Engl J Med 337:1641–1647
Chavassieux PM, Arlot ME, Reda C, Wei L, Yates J, Menuier PJ (1997) Histomorphometric assessment of the long-term effects of alendronate on bone quality and remodeling in patients with osteoporosis. J Clin Invest 100(6):1475–1480
Lane NE, Thompson JM, Strewler GJ, Kinney JH (1995) Intermittent treatment with human parathyroid hormone (hPTH[1–34]) increased trabecular bone volume but not connectivity in osteopenic rats. J Bone Miner Res 10 (10):1470–1477
Mosekilde L, Danielson CC, Gasser J (1994) The effect on vertebral bone mass and strength of long-term treatment with anti-resorptive agents (estrogen and calcitonin), human parathyroid hormone-(hPTH[1–38]), and combination therapy, assessed in aged ovariectomized rats. Endocrinology 134(5):2126–2134
Wronski TJ, Yen C-F, Qi H, Dann LM (1993) Parathyroid hormone is more effective than estrogen or bisphosphonates for restoration of lost bone mass in ovariectomized rats. Endocrinology 132:823–831
Lindsay R, Nieves J, Formica C, et al. (1997) Randomized controlled study of the effect of PTH on vertebral bone mass and fracture incidence among postmenopausal women on oestrogen with osteoporosis. Lancet 350:550–555
Recker RR (1993) Architecture and vertebral fracture. Calcif Tiss Int 53 (Suppl. 1):139–142
Liang H, Pun S, Wronski TJ (1999) Bone anabolic effects of basic fibroblast growth factor in ovariectomized rat. Endocrinology 140 (12):5780–5788
Pun S, Floria CL, Wronski TJ (2000) Anabolic effects of basic fibroblast growth factor in the tibial diaphysis of ovariectomized rats. Bone 27(2):197–202
Canalis E, Centrella M, McCarthy T (1988) Effects of fibroblast growth factor on bone formation in vitro. J Clin Invest 81:1572–1577
Dunstan CR, Boyce R, Boyce BF, Garrett IR, Izbicka E, Burgess WH, Mundy GR (1999) Systemic administration of acidic fibroblast growth factor (FGF-1) prevents bone loss and increases new bone formation in ovariectomized rats. J Bone Miner Res 14:953–959
Noff D, Pitaru S, Savion N (1989) Basic fibroblast growth factor enhances the capacity of bone marrow cells to form bone-like nodules in vitro. FEBS Letter 250:619–621
Rodan SB, Wesolowski G, Thomas KA, Yoon K, Rodan GA (1989) Effects of acidic and basic fibroblast growth factors on osteoblastic cells. Connect Tissue Res 20:283–288
Pitaru S, Kotev-Emeth S, Noff D, Kaffuler S, Savion N (1988) Effect of basic fibroblast growth factor on the growth and differentiation of adult stromal bone marrow cells: enhanced development of mineralized bone-like cells in culture. Bone 23:111–117
Hanada K, Dennis JE, Caplan AI (1997) Stimulatory effects of basic fibroblast growth factor and bone morphogenic protein-2 on osteogenic differentiation of rat bone marrow-derived mesenchymal stem cells. J Bone Miner Res 12:1606–1614
Waynforth HB (1980) Experimental and surgical techniques in the rat. Academic Press, New York
Kalu KN. (1991) The ovariectomized rat model of postmenopausal bone loss. Bone Miner 15:175–192
Kinney JH, Lane NE, Haupt D (1995) Three dimensional in vivo microscopy of sequential changes in trabecular architecture following estrogen depletion in female rats. J Bone Miner Res 10:264–270
Lane NE, JM Thompson, D Haupt, DB Kimmel, G Modin, JH Kinney (1998) Acute changes in trabecular bone connectivity and osteoclast activity in the ovariectomized rat in vivo. J Bone Min Res 13(2):229–236
Kinney JH, Haupt DL, Balooch M, Ladd AJC, Ryaby JT, Lane NE (2000) Three-dimensional morphometry of the L6 vertebrae in the ovariectomized rat model in osteoporosis: biomechanical implications. J Bone Min Res 15(10):1981–1991
Nuzzo S, Peyrin F, Cloetens P, Baruchel J, Bolvin G (2002) Quantification of the degree of mineralization of bone in three dimensions using synchrotron radiation microtomography. Medical Physics 29:2672–2681
Parfitt AM (1983) The physiologic and clinical significance of bone histomorphometric data. In: Recker RR (ed) Bone histomorphometry: techniques and interpretation. CRC Press, Boca Raton, FL, pp 143–224
Parfitt AM, Drezner MK, Glorieux FH, Kanis JA, et al. (1987) Bone histomorphometry: standardization of nomenclature, symbols, and units. J Bone Miner Res 2:595–610
Odgaard A, Gundersen HJG (1993) Quantification of connectivity in cancellous bone with special emphasis on 3-D reconstructions. Bone 14:173–182
Seyedin SM, Kung VT, Daniloff YN, Hesley RP, Gomez B, Nielsen LA, Rosen HN, Zuk RF (1993) Immunoassay for urinary pyridinoline: the new marker of bone resorption. J Bone Miner Res 8:635–641
Delmas PJ, Schlemmer A, Gineyts E, Riis B, Christiansen C (1991) Urinary excretion of pyridinoline crosslinks correlates with bone turnover measured on iliac crest biopsy in patients with osteoporosis. J Bone Miner Res 6:639–644
Sokal R, Rohlf FJ (1969) Biometry, Freeman, San Francisco, CA
Mundy GR, Boyce B, Hughes D, Wright K, Bonewald L, Dallas S, Harris S, Ghosh-Choudhury N, Chen D, Dunstand C, Izbicka E, Yoneda T (1995) The effects of cytokines and growth factors on osteoblastic cells. Bone 17:71S-75S
Hurley MM, Abreu C, Harrison JR, Lichtler AC, Raisz LG, Kream BE (1993) Basic fibroblast growth factor inhibits type I collagen gene expression in osteoblastic MC3T3-E1 cells. J Biol Chem 268:5588–5593
Globus RK, Plouet J, Gospodarowicz D (1989) Cultured bovine bone cells synthesize basic fibroblast growth factor and store it in their extracellular matrix. Endocrinology 124:1539–1547
Leboy PS, Beresford JN, Devlin C, Owen ME (1991) Dexamethasone induction of osteoblast mRNA in rat marrow stromal cell cultures. J Cell Physiol 146:370–378
Scutt A, Bertram P (1999) Basic fibroblast growth factor in the presence of dexamethasone stimulates colony formation, expansion, and osteoblastic differentiation by rat bone marrow stromal cells. Calcif Tissue Int 64:69–77
Beresford JN, Bennett JH, Devlin C, Leboy PS, Owen ME (1992) Evidence for an inverse relationship between the differentiation of adipocytic and osteogenic cells in rat marrow stromal cell cultures. J Cell Sci 102:341–351.
Hurley MM, Lee SK Riasz LG, Benecker P, Lorenzo J (1998) Basic fibroblast growth factor induces osteoclast formation in murine bone marrow cultures. Bone 22(4):309–316
Noda M, Camilliere JJ (1989) In vivo stimulation of bone formation by transforming growth factor beta. Endocrinology 124:2991–2994
Nakamura K, Kurokawa T, Aoyama I, Hanada K, Tamura M, Kawaguchi H (1998) Stimulation of bone formation by intraosseous injection of basic fibroblast growth factor in ovariectomized rats. Int Ortho 22:49–54
Hofbauer LC, Khosla S, Dunstan CR, Lacey DL, Boyle WJ, Riggs BL (2000) The roles of osteoprotegerin and osteoprotegerin ligand in the paracrine regulation of bone resorption. J Bone Miner Res 15(1):2–12
Wronski TJ, Ratkus AM, Thomsen JS, Vulcan TQ, Mosekilde L (2001) Sequential treatment with basic fibroblast growth factor and parathyroid hormone restores lost cancellous bone mass and strength in the proximal tibia of aged ovariectomized rats. J Bone Miner Res 16(8):1399–1407
Acknowledgements
The authors would like to acknowledge David Haupt for assistance in obtaining the XTM images. This work was supported by grants from the NIH 1R01AR43052, the Research Evaluation and Allocation Committee of UCSF and the Rosalind Russell Arthritis Research Center.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lane, N.E., Kumer, J., Yao, W. et al. Basic fibroblast growth factor forms new trabeculae that physically connect with pre-existing trabeculae, and this new bone is maintained with an anti-resorptive agent and enhanced with an anabolic agent in an osteopenic rat model. Osteoporos Int 14, 374–382 (2003). https://doi.org/10.1007/s00198-003-1374-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-003-1374-7