Abstract
Introduction and hypothesis
Pelvic organ prolapse (POP) surgery has lately gained importance in gynecological practice. This study aims to characterize the evolution of POP surgical procedures conducted in Portugal in the last decade and the impact of an FDA 2011 safety communication on mesh POP surgeries.
Methods
Trends in the surgical management of POP were assessed using the Portuguese National Medical Registry. We considered all records of women with diagnosis of genital prolapse from 1 January 2000 to 31 December 2012. Additionally, we also conducted a survey among members of the Portuguese Society of Urogynecology to evaluate current practices in the surgical management of POP.
Results
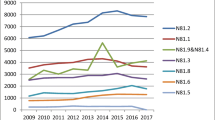
From 2000 to 2012, 46,819 diagnoses of genital prolapse were registered, with a 105 % increase during the study period (2,368 in 2000 to 4,941 in 2012). POP mesh surgery represented only 6 % of total prolapse diagnoses, but mesh use greatly increased up to 2011, when only a slight increase was registered. Among gynecologists who responded to the questionnaire, there was considerable variability on the procedures of choice to treat POP. Fifty-seven per cent of respondents performed vaginal mesh POP surgery, but only 27 % of those actually reported having changed their practice after the FDA 2011 safety communication.
Conclusions
Surgical procedures for POP conducted in Portugal greatly increased over the last decade. The use of surgical meshes is still limited, but despite FDA safety communication it has increased over the years, with a slight increase in 2012, which illustrates the need for further analyses in the coming years.



Similar content being viewed by others
Abbreviations
- FDA:
-
Food and Drug Administration
- ICD-9-CM:
-
International Classification of Diseases, 9th revision, Clinical Modification
- IUGA:
-
International Urogynecological Association
- POP:
-
Pelvic organ prolapse
- PSUG:
-
Portuguese Society of Urogynecology
- SPG:
-
Portuguese Society of Gynecology
References
Hendrix SL, Clark A, Nygaard I, Aragaki A, Barnabei V, McTiernan A (2002) Pelvic organ prolapse in the Women's Health Initiative: gravity and gravidity. Am J Obstet Gynecol 186(6):1160–1166
Smith FJ, Holman CD, Moorin RE, Tsokos N (2010) Lifetime risk of undergoing surgery for pelvic organ prolapse. Obstet Gynecol 116(5):1096–1100. doi:10.1097/AOG.0b013e3181f73729
Maher C (2013) ICI 2012: pelvic organ prolapse surgery. Int Urogynecol J 24(11):1781. doi:10.1007/s00192-013-2168-x
van Geelen JM, Dwyer PL (2013) Where to for pelvic organ prolapse treatment after the FDA pronouncements? A systematic review of the recent literature. Int Urogynecol J 24(5):707–718. doi:10.1007/s00192-012-2025-3
Skoczylas LC, Turner LC, Wang L, Winger DG, Shepherd JP (2014) Changes in prolapse surgery trends relative to FDA notifications regarding vaginal mesh. Int Urogynecol J 25(4):471–477. doi:10.1007/s00192-013-2231-7
Deprest J, Zheng F, Konstantinovic M, Spelzini F, Claerhout F, Steensma A, Ozog Y, De Ridder D (2006) The biology behind fascial defects and the use of implants in pelvic organ prolapse repair. Int Urogynecol J Pelvic Floor Dysfunct 17 [Suppl 1]:S16–S25. doi:10.1007/s00192-006-0101-2
Aslani N, Brown CJ (2010) Does mesh offer an advantage over tissue in the open repair of umbilical hernias? A systematic review and meta-analysis. Hernia 14(5):455–462. doi:10.1007/s10029-010-0705-9
Menchen LC, Wein AJ, Smith AL (2012) An appraisal of the Food and Drug Administration warning on urogynecologic surgical mesh. Curr Urol Rep 13(3):231–239. doi:10.1007/s11934-012-0244-2
Food and Drug Administration (2008) FDA public health notification: serious complications associated with transvaginal placement of surgical mesh in repair of pelvic organ prolapse and stress urinary incontinence. http://www.fda.gov/MedicalDevices/Safety/AlertsandNotices/PublicHealthNotifications/ucm061976.htm
Food and Drug Administration (2011) Urogynecologic surgical mesh: update on the safety and effectiveness of transvaginal placement for pelvic organ prolapse. http://www.fda.gov/downloads/MedicalDevices/Safety/AlertsandNotices/UCM262760.pdf. Accessed 27 February 2014
Clemons JL, Weinstein M, Guess MK, Alperin M, Moalli P, Gregory WT, Lukacz ES, Sung VW, Chen BH, Bradley CS, Committee AR (2013) Impact of the 2011 FDA transvaginal mesh safety update on AUGS members' use of synthetic mesh and biologic grafts in pelvic reconstructive surgery. Female Pelvic Med Reconstr Surg 19(4):191–198. doi:10.1097/SPV.0b013e31829099c1
Ellington DR, Richter HE (2013) The role of vaginal mesh procedures in pelvic organ prolapse surgery in view of complication risk. Obstet Gynecol Int 2013:356960. doi:10.1155/2013/356960
Freitas A, Silva-Costa T, Lopes F, Garcia-Lema I, Teixeira-Pinto A, Brazdil P, Costa-Pereira A (2012) Factors influencing hospital high length of stay outliers. BMC Health Serv Res 12:265. doi:10.1186/1472-6963-12-265
Administração Central do Sistema de Saúde (2011) Relatório anual de auditoria externa à codificação clínica—Ano 2010. http://www.acss.min-saude.pt/Portals/0/Relat%C3%B3rio_anual_2010_%20V%201%200_auditorias%20codif%20clin02.09.pdf
Centers for Disease Control and Prevention (2013) Conversion table of new ICD-9-CM codes, October 2013. http://www.cdc.gov/nchs/data/icd/ICD-9-CM_FY14_CNVTBL_Final.pdf. Accessed 27 February 2014
Bradley SL, Weidner AC, Siddiqui NY, Gandhi MP, Wu JM (2011) Shifts in national rates of inpatient prolapse surgery emphasize current coding inadequacies. Female Pelvic Med Reconstr Surg 17(4):204–208. doi:10.1097/SPV.0b013e3182254cf1
Detollenaere RJ, den Boon J, Kluivers KB, Vierhout ME, van Eijndhoven HW (2013) Surgical management of pelvic organ prolapse and uterine descent in the Netherlands. Int Urogynecol J 24(5):781–788. doi:10.1007/s00192-012-1934-5
Jones KA, Shepherd JP, Oliphant SS, Wang L, Bunker CH, Lowder JL (2010) Trends in inpatient prolapse procedures in the United States, 1979–2006. Am J Obstet Gynecol 202(5):501–507. doi:10.1016/j.ajog.2010.01.017
Instituto Nacional de Estatística (2013) Estatísticas Demográficas 2012
Luber KM, Boero S, Choe JY (2001) The demographics of pelvic floor disorders: current observations and future projections. Am J Obstet Gynecol 184(7):1496–1501, discussion 1501–1493
Jha S, Moran P (2011) The UK national prolapse survey: 5 years on. Int Urogynecol J 22(5):517–528. doi:10.1007/s00192-011-1379-2
Babalola EO, Bharucha AE, Melton LJ 3rd, Schleck CD, Zinsmeister AR, Klingele CJ, Gebhart JB (2008) Utilization of surgical procedures for pelvic organ prolapse: a population-based study in Olmsted County, Minnesota, 1965–2002. Int Urogynecol J Pelvic Floor Dysfunct 19(9):1243–1250. doi:10.1007/s00192-008-0613-z
Wu MP, Huang KH, Long CY, Tsai EM, Tang CH (2010) Trends in various types of surgery for hysterectomy and distribution by patient age, surgeon age, and hospital accreditation: 10-year population-based study in Taiwan. J Minim Invasive Gynecol 17(5):612–619. doi:10.1016/j.jmig.2010.04.010
Rogo-Gupta L, Rodriguez LV, Litwin MS, Herzog TJ, Neugut AI, Lu YS, Raz S, Hershman DL, Wright JD (2012) Trends in surgical mesh use for pelvic organ prolapse from 2000 to 2010. Obstet Gynecol 120(5):1105–1115. doi:10.1097/AOG.0b013e31826ebcc2
Erekson EA, Lopes VV, Raker CA, Sung VW (2010) Ambulatory procedures for female pelvic floor disorders in the United States. Am J Obstet Gynecol 203(5):497. doi:10.1016/j.ajog.2010.06.055
Jonsson Funk M, Edenfield AL, Pate V, Visco AG, Weidner AC, Wu JM (2013) Trends in use of surgical mesh for pelvic organ prolapse. Am J Obstet Gynecol 208(1):79
Reynolds WS, Gold KP, Ni S, Kaufman MR, Dmochowski RR, Penson DF (2013) Immediate effects of the initial FDA notification on the use of surgical mesh for pelvic organ prolapse surgery in medicare beneficiaries. Neurourol Urodyn 32(4):330–335. doi:10.1002/nau.22318
Acknowledgements
The authors would like to thank the Central Administration of the National Health System (Administração Central do Sistema de Saúde) for providing access to the Portuguese National Medical Registry (clinical/administrative data).
Funding
This study received no external funding.
Financial disclaimer/conflict of interest
None.
Ethics/institutional review board approval
This study is not subject to approval by the ethics/institutional review board, since data were retrieved from a national database (the Portuguese National Medical Registry, clinical/administrative data) from which patient-identifying information is not available to the research team (all patient-identifying information is encrypted).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mascarenhas, T., Mascarenhas-Saraiva, M., Ricon-Ferraz, A. et al. Pelvic organ prolapse surgical management in Portugal and FDA safety communication have an impact on vaginal mesh. Int Urogynecol J 26, 113–122 (2015). https://doi.org/10.1007/s00192-014-2480-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00192-014-2480-0