Abstract
Objectives
To determine if tidal volume (V T) between 6 and 10 ml/kg body weight using pressure control ventilation affects outcome for children with acute hypoxemic respiratory failure (AHRF) or acute lung injury (ALI). To validate lung injury severity markers such as oxygenation index (OI), PaO2/FiO2 (PF) ratio, and lung injury score (LIS).
Design
Retrospective, January 2000–July 2007.
Setting
Tertiary care, 20-bed PICU.
Patients
Three hundred and ninety-eight endotracheally intubated and mechanically ventilated children with PF ratio <300. Outcomes were mortality and 28-day ventilator free days.
Measurements and main results
Three hundred and ninety-eight children met study criteria, with 20% mortality. 192 children had ALI. Using >90% pressure control ventilation, 85% of patients achieved V T less than 10 ml/kg. Median V T was not significantly different between survivors and non-survivors during the first 3 days of mechanical ventilation. After controlling for diagnostic category, age, delta P (PIP-PEEP), PEEP, and severity of lung disease, V T was not associated with mortality (P > 0.1), but higher V T at baseline and on day 1 of mechanical ventilation was associated with more ventilator free days (P < 0.05). This was particularly seen in patients with better respiratory system compliance [Crs > 0.5 ml/cmH20/kg, OR = 0.70 (0.52, 0.95)]. OI, PF ratio, and LIS were all associated with mortality (P < 0.05).
Conclusions
When ventilating children using lung protective strategies with pressure control ventilation, observed V T is between 6 and 10 ml/kg and is not associated with increased mortality. Moreover, higher V T within this range is associated with more ventilator free days, particularly for patients with less severe disease.
Similar content being viewed by others
Introduction
Acute hypoxemic respiratory failure (AHRF), acute lung injury (ALI) and acute respiratory distress syndrome (ARDS) result in morbidity and mortality for pediatric intensive care unit (PICU) patients. While contemporary ventilation strategies have improved outcome, mortality continues at 30–40% for adults [1], and 8–22% for children [2–4] with ALI or ARDS.
Several adult studies demonstrate improved outcome when patients are managed in volume control mode with set tidal volumes of 6 ml/kg predicted body weight, compared to 12 ml/kg [1, 6]. However, differences between adult and pediatric practice regarding modes of ventilation, tidal volume measurements, and developmental differences bring into question whether such strategies should be embraced universally in pediatrics [5, 13–18]. Moreover, evidence in both adults and children suggests that lung protective strategies should not be one-size fits all, but rather catered to lung disease severity [7, 8]. For further discussion, see Electronic supplementary material (ESM).
We sought to determine the effect of generated tidal volume on outcome for children with ALI or AHRF using historical data from greater than 90% of patients on pressure control ventilation. Additionally, we examined how well lung injury severity measures such as PaO2/FiO2 (PF) ratio, oxygenation index (OI), and a modified Murray pediatric lung injury score (LIS) [19] reflect mortality at various intervals during the initial days of mechanical ventilation [20].
Methods
Patient selection
Retrospective review of all admissions to a tertiary care PICU between January 2000 and July 2007 was conducted. Patients were eligible if endotracheally intubated and mechanically ventilated, and at least one PF ratio was less than 300 within 24 h after intubation. Patients were excluded for evidence of cardiac disease or incomplete ventilation data. All patients met three of four diagnostic criteria for ALI (acute onset, PF ratio <300, and no left ventricular dysfunction). The presence of bilateral infiltrates on chest radiograph (fourth ALI criteria) was handled separately. Finally, all patients with an endotracheal tube leak greater than 20% were excluded [24].
Variable selection
Data were extracted from two databases: an electronic ICU flow sheet (Philips CareVue®, Waltham, MA), and a patient database (Microsoft Access©, Redmond, WA). From these two databases, information on age, race, gender, weight, primary diagnosis, year of admission, chest X-rays, arterial blood gas values, ventilator settings, and a pediatric risk of mortality (PRISM) Score were extracted [25]. All chest X-rays were read by a pediatric radiologist blinded to the study, and reports reviewed for the presence of ALI criteria.
Ventilator strategy
While this study was non-interventional, a lung-protective strategy was in place in our PICU (see ESM).
Ventilator and arterial blood gas (ABG) variable definition
The initial PF ratio <300 after intubation was defined as “baseline,” where blood gas measurements were extracted. In addition, the closest charted ventilator settings at or prior to the ABG were extracted. Tidal volume (V T ml/kg) was calculated using exhaled tidal volume (V Te) measured at the ventilator with compensation for tubing compliance and make of the ventilator, divided by actual body weight.
Aggregate variables were created to express time-weighted averages of ventilator settings and blood gas values over the first 3 days after “baseline” (see ESM).
Lung injury severity markers
PF ratio, oxygenation index [OI = (mean airway pressure × FiO2)/(PaO2) × 100], dynamic compliance of the respiratory system [Crs = V T (ml/kg)/(PIP-PEEP)], and a modified Murray lung injury score (LIS) for pediatrics [19] were determined at “baseline” and then again aggregated over the first 3 days after baseline, as above. The modified lung injury score is a composite of PF ratio, PEEP, Crs, and involved quadrants on chest radiograph, with integers from zero to four for each component. Data on quadrants of alveolar consolidation on chest X-ray were not included in this analysis. As a result, the LIS was the average of three, not four, components.
Outcome measures
The primary outcome measure was ICU mortality. Secondary outcome was 28-day ventilator free days (VFDs) [26]. Data on outcome measures were complete and no censoring was necessary.
Statistics
Statistical analysis was performed using Statistica v. 5.5 (StatSoft, Tulsa, OK) and Stata v. 10 (StataCorp, College Station, TX). Continuous variables were analyzed with a Wilcoxon rank-sum test, as assumptions of normality could not always be satisfied. Dichotomous outcomes were analyzed using a Yates-corrected Chi-squared test. Kruskall–Wallis ANOVA was used to examine differences in median tidal volume stratified by categories. Logistic regression analysis was performed to examine the impact of the variables of interest on the outcome of mortality, and control for potential confounding variables, or effect modifiers. A multivariate logistic regression model was built incorporating variables with univariate associations with mortality (P < 0.2), with care taken to avoid terms that were collinear. Overall goodness of fit was assessed using the Hosmer–Lemeshow test and graphical evaluation for influential points. ROC plots were created and overall discrimination ability of the various predictive models was assessed using empirical estimates of the overall area under the curve (AUC).
The outcome of 28-day VFDs had an almost bimodal distribution, as reported previously. Hence, for logistic regression analysis, it was dichotomized to <14 days, or ≥14 days, as previously described [4]. However, when appropriate, comparisons of median number of VFDs between groups were analyzed using a Wilcoxon rank-sum test [26].
For more detailed description of methods and power calculations, please see ESM.
Results
Patient selection
Eight thousand two hundred and forty-six patients <18 years were admitted to the PICU during the study period, and 773 had at least two arterial PF ratios <300. Seven hundred patients were mechanically ventilated and 507 had at least one PF ratio <300 after endotracheal intubation. Nine patients were excluded for incomplete ventilator or blood gas data, 49 patients met cardiac exclusions, and 51 patients had an endotracheal tube leak >20%.
Three hundred and ninety-eight patients were included in the final analysis, of which 227 were male (57%), 194 were Latino (49%), with a median age of 4.3 years. Eighty patients died, an overall mortality of 20%. One hundred ninety-two patients (48%) had bilateral pulmonary infiltrates on chest X-ray and met full criteria for either ALI or ARDS (Table 1).
Mortality
Univariate results
Associations of diagnostic and demographic variables with mortality are presented in Table 1.
For the entire population, the median PF ratio at baseline was 138 with a lung injury score of 2.33. Patients were managed almost exclusively with pressure control ventilation (>90%, PRVC comprising the rest), with a median PEEP of 6 cmH20, V T of 7.4 ml/kg, MAP of 11 cmH20, and PIP of 26 cmH20. Eighty-four percent of patients had a PIP ≤ 35, and 94% a PIP ≤ 40. At baseline, patients that died had higher median OI, lower median PF ratio, higher median lung injury score, as well as higher FiO2, PIP, PEEP, MAP, lower pH, PaO2, and base deficit. Median V T was 7.04 ml/kg in deceased and 7.60 ml/kg in survivors (P = 0.13). These trends for blood gas, ventilator, and lung severity markers remained consistent over the first 3 days of ventilation (Table 2).
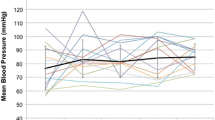
Seventy percent of patients were managed with V T between 6 and 10 ml/kg throughout the first 3 days of ventilation (Fig. 1). There was a non-significant trend for improved survival with higher V T. Patients with higher LIS had lower median V T than those with lower LIS (K–W ANOVA P < 0.001, Fig. 2). However, the relationship between V T and mortality was similar when stratified as a function of LIS (Fig. 3), even though mortality was higher for patients with higher LIS.
Tidal volume and mortality by day of mechanical ventilation. There is a general trend for higher mortality with lower tidal volumes both at the beginning of mechanical ventilation, and throughout the first 3 days. Most patients were ventilated between 6 and 10 ml/kg body weight. Mortality expressed as a proportion
Markers of lung injury including PF ratio, OI, and LIS were all significantly associated with mortality at baseline (Table 2) with similar overall classification in a logistic regression model (Table 3). Further, there was a trend for stronger degrees of association with mortality with each additional day the patient remained on mechanical ventilation (Fig. 4). Notably, patients with “severe ARDS,” defined as baseline LIS > 2.5 [16], had significantly higher mortality than those with LIS ≤ 2.5 [53/163 (32.5%) vs. 26/225 (11.5%), P < 0.001, Fig. 3]. Respiratory system compliance was associated with mortality when averaged (time-weighted) for days 1–3, although not at baseline.
Lung injury by day and mortality. Odds ratio (OR) for death per one unit increase in OI, one unit increase in LIS, 0.1 unit decrease in Crs, and 20 point decrease in PF ratio. LIS and OI are associated with mortality at baseline and over the first 3 days (P < 0.05). Crs over the first 3 days and PF ratio at baseline, second and third day are associated with mortality (P < 0.05).  95% CI,
95% CI,  odds ratio
odds ratio
Bilateral pulmonary infiltrates
One hundred and ninety-two patients had bilateral pulmonary infiltrates and met all four ALI criteria. Patients with bilateral pulmonary infiltrates had higher PIP, PEEP, OI, and lower median V T (7.13 vs. 7.84 ml/kg, P = 0.02) than those without bilateral infiltrates. Moreover, 44/192 (22.8%) patients with bilateral infiltrates died compared to 36/206 (17.5%) without bilateral infiltrates (P = 0.2). However, there was no difference between median initial V T and mortality when limited to patients with bilateral pulmonary infiltrates [n = 192, 7.3 (survivors) vs. 7.0 (deceased) ml/kg, P = 0.27]. There was also no difference for aggregated V T over days 1–3 (P > 0.1).
Multivariate results
Multivariate logistic regression modeling was performed to determine whether severity of lung disease or systemic illness impacted the relationship between V T and mortality. V T was not associated with mortality after controlling for age, Delta P (PIP-PEEP), PEEP, admission diagnosis, and severity of lung disease using LIS, PF ratio, or OI (all V T coefficients P > 0.1, H–L test P > 0.2). Moreover, there were no significant interaction effects between V T and presence of bilateral infiltrates on chest X-ray, diagnostic category of admission, or severity of lung disease as measured by OI, PF ratio, Crs (<0.5 or ≥0.5 ml/cmH20/kg), or LIS and mortality (all interaction term P values >0.1, see ESM). Finally, due to concerns of colinearity between PRISM score and individual components of the model, a separate model including V T, PRISM score, age, Delta P, and PEEP was built. It also demonstrated initial as well as day 1–3 V T had no effect on mortality (P > 0.1 all V T coefficients).
Bilateral pulmonary infiltrates
When limited to patients with bilateral pulmonary infiltrates, V T had no effect on mortality after controlling for age, Delta P, PEEP, admission diagnosis, and severity of lung disease using LIS, PF ratio, or OI (all V T coefficients P > 0.1, H–L test P > 0.2). The same results were seen using the model incorporating PRISM score (all V T coefficients P > 0.2, H–L test P > 0.2).
Analysis of non-survivors
For the 80 patients that died, refractory respiratory failure [28] was responsible for death in 29/80 (36%) patients. Twenty-five (31%) patients met neurologic criteria for death or had a hopeless neurological condition. Shock or cardiac arrest not from refractory respiratory failure was responsible for 22 (27%) deaths. Four patients (5%) died from other causes. Fifty-two (69%) patients had at least three dysfunctional organs prior to death. Thirty-four (43%) patients had a documented limitation of care or do not resuscitate order.
28 day ventilator free days (VFDs)
Univariate analysis
The entire cohort had a median of 17.4 VFDs (IQR 0, 24.4). Lower baseline V T and day 1 V T were both associated with fewer VFDs (Table 4). Moreover, all variables that had a univariate association with mortality were also associated with fewer VFDs. Except for V T at baseline and day 1, variables not associated with mortality were also not associated with fewer VFDs (analysis not shown). Patients with bilateral pulmonary infiltrates had significantly fewer VFDs (median 16.4 vs. 20.8, P = 0.02), although this was not significant if VFDs were dichotomized at 14 days.
Multivariate analysis
This relationship between lower V T on day 1 of ventilator management and fewer VFDs held after controlling for age, Delta P, PEEP, admission diagnosis, and measures of lung disease severity such as OI, PF ratio, or LIS (P < 0.05 with each measure of lung disease severity). The same results were seen with the multivariate model incorporating PRISM score, age, Delta P, and PEEP [OR = 0.88 (0.78, 0.98)]. Further, when stratifying patients by dynamic compliance of the respiratory system <0.5 or ≥0.5 ml/cmH20/kg, higher V T on day 1 was associated with more VFDs for patients with respiratory system compliance ≥0.5 [n = 76, OR = 0.73 (0.56,0.95)] than for patients with compliance <0.5 [n = 254, OR = 0.90 (0.80, 1.01)]. This relationship held after controlling for Delta P, PEEP, and LIS [n = 76, OR = 0.70 (0.52, 0.95)].
Bilateral pulmonary infiltrates
Finally, when limiting the analysis to the 192 patients with bilateral infiltrates, the association between higher V T on day 1 and more VFDs held true [OR = 0.82 (0.70, 0.96)]; however this did not reach statistical significance when controlling for age, Delta P, PEEP, admission diagnosis, and LIS [n = 156, OR = 0.85 (0.70, 1.03)].
Discussion
This study demonstrates when using a pressure control lung protective ventilation strategy, V T in the typical range of 6–10 ml/kg measured at the mechanical ventilator does not appear to be associated with mortality for children with ALI or AHRF. This represents true clinical practice as the ventilator variables are composites capturing patient exposure to particular settings, rather than predetermined intermittent snapshots. This echoes the findings of Flori et al. [4], showing no difference in V T between survivors and non-survivors with pediatric ALI.
Previous work has shown that a universal approach to V T selection for patients with ALI may not be best. Higher V T (>6 ml/kg) may be associated with better outcome for patients with less severe lung disease by improving alveolar recruitment, independent of PEEP. Here, the association between higher V T on day 1 and more VFDs was seen more clearly in patients with less severe lung disease (Crs ≥ 0.5 ml/cmH20/kg). One can speculate that patients with less severe lung disease may therefore benefit from a higher V T strategy [7, 8, 12]. We advocate V T choice be tailored to lung disease severity, rather than universally being 6 ml/kg for all children.
Inherently, pressure control ventilation facilitates changing V T based on compliance, as patients with poor compliance attain a smaller V T for a given pressure than patients with better compliance. This is seen in Fig. 2, where patients with severe disease, measured by LIS, were managed with lower median V T. Moreover, the decelerating flow of pressure limited ventilation imparts additional benefit over the constant flow of volume modes of ventilation for patients with non-homogenous lung disease [29].
Given the almost universal use of pressure control ventilation and the strategy to limit PIP < 40 cmH20, the observed relationship between higher V T and more VFDs is likely explained by lung disease severity. Patients with severe disease will attain a lower V T than those with better compliance, given the same Delta P. As such, V T and compliance really measure the same things: severity of lung disease. We controlled for disease severity using OI, PF ratio, LIS, PRISM, as well as pressure to attain V T, and found the relationship between higher first day V T (up to 10 ml/kg) and more VFDs to hold. Short of a randomized controlled trial where different V T between 6 and 10 ml/kg are targeted using pressure control ventilation, it is difficult to definitively answer whether higher V T is in some way protective. However, this data demonstrates a pressure control strategy with a restricted upper pressure limit targeting V T less than 10 ml/kg measured at the mechanical ventilator, is safe for children with ALI or AHRF.
A caveat to these findings is V T measurements were not made at the airway using a proximal flow sensor, but rather at the ventilator with compensation for tubing compliance. This could result in overestimation of V T, particularly for small children [16]. Unfortunately, proximal measurements were not available for a large number of patients, and such measurements are not routinely used clinically in most ICUs.
With the hope of finding larger variability in ventilator and V T strategies, we analyzed the presence of bilateral pulmonary infiltrates on chest radiograph separately, although all patients met other diagnostic criteria for ALI. Patients with bilateral infiltrates had more severe lung disease, a trend towards higher mortality, and fewer VFDs than those without bilateral infiltrates. However, the relationship between V T and outcome was not different in these two groups, and bilateral infiltrates did not confound or modify the relationship between V T and mortality or VFDs. In fact, the same trends were seen in the 192 patients who met all four criteria for ALI, although some power was lost. With the 192 patients in the ALI group we could detect a 7% reduction in mortality with a power of 0.8, or a 9% reduction in >14 VFDs. With the entire cohort of 398 we could detect a 4.5% reduction in mortality, or a 6.5% reduction in >14 VFDs. While ALI and ARDS represent unique physiologic cohorts, we believe these conclusions regarding V T and outcome are applicable to patients with either AHRF or ALI.
This study also demonstrates that OI, PF ratio, and a pediatric modification of the Murray lung injury score are all associated with mortality for children with AHRF or ALI. In contrast to adult studies [28, 30–32], initial degree of oxygen impairment is associated with mortality for children, validating previous pediatric results [4, 33]. In general, mortality for ALI in adults is higher, and they tend to die of other organ dysfunction, not irreversible respiratory failure. Approximately 40% of deaths in our study were due to refractory respiratory failure, with 70% of patients having at least three organ dysfunctions at death. This reflects the findings of Trachsel et al. [33], and contrasts with adult studies where mortality from irreversible respiratory failure was responsible for 16% of deaths [28].
Further, OI has a stronger association with mortality each day the patient remains ventilated, as previously reported [33]. This is likely due to alveolar recruitment shortly after intubation, and helps explain why initial measures of lung disease severity often are not robust predictors of outcome. Nonetheless, we have demonstrated that any of these measures (OI, PF, LIS) can be used in future studies on ALI or AHRF to assess disease severity and stratify risk.
There are several shortcomings to this retrospective study. First, V T measurements were made at the mechanical ventilator, as discussed earlier. Second, consistency in charting may be variable depending on personnel, patient, or unit acuity. While this cannot be controlled for retrospectively, most information was captured electronically in real-time and human transcription minimized. Third, this was not a randomized trial, so we could not control for all confounding variables.
Conclusions
Ventilating children with ALI or AHRF using a pressure limited strategy with a reasonable upper pressure limit, targeting V T < 10 ml/kg measured at the mechanical ventilator, is safe. Further, severity of lung disease measures such as OI, PF ratio, and a pediatric modification of the Murray lung injury score are all associated with mortality, both on initiation and throughout the first 3 days of ventilator support. This data is retrospective from a single center, and should be confirmed with a multicenter trial.
References
ARDSnet (2000) Ventilation with lower tidal volumes as compared with traditional tidal volumes for acute lung injury and the acute respiratory distress syndrome. The Acute Respiratory Distress Syndrome Network. N Engl J Med 342:1301–1308
Curley MA, Hibberd PL, Fineman LD, Wypij D, Shih MC, Thompson JE, Grant MJ, Barr FE, Cvijanovich NZ, Sorce L, Luckett PM, Matthay MA, Arnold JH (2005) Effect of prone positioning on clinical outcomes in children with acute lung injury: a randomized controlled trial. JAMA 294:229–237
Willson DF, Thomas NJ, Markovitz BP, Bauman LA, DiCarlo JV, Pon S, Jacobs BR, Jefferson LS, Conaway MR, Egan EA and the Pediatric Acute Lung Injury and Sepsis Investigators (2005) Effect of exogenous surfactant (calfactant) in pediatric acute lung injury: a randomized controlled trial. JAMA 293:470–476
Flori HR, Glidden DV, Rutherford GW, Matthay MA (2005) Pediatric acute lung injury: prospective evaluation of risk factors associated with mortality. Am J Respir Crit Care Med 171:995–1001
Farias JA, Frutos F, Esteban A, Flores JC, Retta A, Baltodano A, Alia I, Hatzis T, Olazarri F, Petros A, Johnson M (2004) What is the daily practice of mechanical ventilation in pediatric intensive care units? A multicenter study. Intensive Care Med 30:918–925
Amato MB, Barbas CS, Medeiros DM, Magaldi RB, Schettino GP, Lorenzi-Filho G, Kairalla RA, Deheinzelin D, Munoz C, Oliveira R, Takagaki TY, Carvalho CR (1998) Effect of a protective-ventilation strategy on mortality in the acute respiratory distress syndrome. N Engl J Med 338:347–354
Tobin MJ (2000) Culmination of an era in research on the acute respiratory distress syndrome. N Engl J Med 342:1360–1361
Deans KJ, Minneci PC, Cui X, Banks SM, Natanson C, Eichacker PQ (2005) Mechanical ventilation in ARDS: one size does not fit all. Crit Care Med 33:1141–1143
Stewart TE, Meade MO, Cook DJ, Granton JT, Hodder RV, Lapinsky SE, Mazer CD, McLean RF, Rogovein TS, Schouten BD, Todd TR, Slutsky AS (1998) Evaluation of a ventilation strategy to prevent barotrauma in patients at high risk for acute respiratory distress syndrome. Pressure- and Volume-Limited Ventilation Strategy Group. N Engl J Med 338:355–361
Brochard L, Roudot-Thoraval F, Roupie E, Delclaux C, Chastre J, Fernandez-Mondejar E, Clementi E, Mancebo J, Factor P, Matamis D, Ranieri M, Blanch L, Rodi G, Mentec H, Dreyfuss D, Ferrer M, Brun-Buisson C, Tobin M, Lemaire F (1998) Tidal volume reduction for prevention of ventilator-induced lung injury in acute respiratory distress syndrome. The Multicenter Trail Group on tidal volume reduction in ARDS. Am J Respir Crit Care Med 158:1831–1838
Brower RG, Shanholtz CB, Fessler HE, Shade DM, White P Jr, Wiener CM, Teeter JG, Dodd-o JM, Almog Y, Piantadosi S (1999) Prospective, randomized, controlled clinical trial comparing traditional versus reduced tidal volume ventilation in acute respiratory distress syndrome patients. Crit Care Med 27:1492–1498
Eichacker PQ, Gerstenberger EP, Banks SM, Cui X, Natanson C (2002) Meta-analysis of acute lung injury and acute respiratory distress syndrome trials testing low tidal volumes. Am J Respir Crit Care Med 166:1510–1514
Randolph AG, Meert KL, O’Neil ME, Hanson JH, Luckett PM, Arnold JH, Gedeit RG, Cox PN, Roberts JS, Venkataraman ST, Forbes PW, Cheifetz IM, and the Pediatric Acute Lung Injury and Sepsis Investigators (2003) The feasibility of conducting clinical trials in infants and children with acute respiratory failure. Am J Respir Crit Care Med 167:1334–1340
Curley MA, Arnold JH, Thompson JE, Fackler JC, Grant MJ, Fineman LD, Cvijanovich N, Barr FE, Molitor-Kirsch S, Steinhorn DM, Matthay MA, Hibberd PL, Pediatric Prone Positioning Study Group (2006) Clinical trial design—effect of prone positioning on clinical outcomes in infants and children with acute respiratory distress syndrome. J Crit Care 21:23–32
Albuali WH, Singh RN, Fraser DD, Seabrook JA, Kavanagh BP, Parshuram CS, Kornecki A (2007) Have changes in ventilation practice improved outcome in children with acute lung injury? Pediatr Crit Care Med 8:324–330
Al-Majed SI, Thompson JE, Watson KF, Randolph AG (2004) Effect of lung compliance and endotracheal tube leakage on measurement of tidal volume. Crit Care 8:R398–R402
Khemani RG, Bart RD, Newth CJ (2007) Respiratory monitoring during mechanical ventilation. Paediatr Child Health 17:193–201
Komecki AKB (2007) Mechanical ventilation. In: Wheeler DS, Wong HR, Shanley TP (eds) Pediatric critical care medicine: basic science and clinical evidence. Springer, London, pp 412–425
Hammer J, Numa A, Newth CJ (1997) Acute respiratory distress syndrome caused by respiratory syncytial virus. Pediatr Pulmonol 23:176–183
Khemani RG, Newth CJ, Bart RD (2008) Pediatric hypoxemic respiratory failure: risk factors for mortality. American Thoracic Society international conference abstracts issue: d104.3129 (abstract)
Wheeler A P, Wickersham N, Ancukiewicz M, Brower R, Thomspon T, Bernard G (2000) Low tidal volume (VT) ventilation reduces plasma cytokines in human acute lung injury (ALI). American Thoracic Society international conference abstracts issue: A83 (abstract)
Klein D, Morris AH (2000) Variations in tidal volume due to predicted body weight equations. American Thoracic Society international conference abstracts issue: C91 (abstract)
Lanken PN, Schoenfeld D, Thompson T (2000) Evaluation of early changes in PaO2/FiO2 (P/F) or oxygenation index (OI) as surrogate markers for survival in patients with acute lung injury (ALI) or ARDS. American Thoracic Society international conference abstracts issue: A82
Main E, Castle R, Stocks J, James I, Hatch D (2001) The influence of endotracheal tube leak on the assessment of respiratory function in ventilated children. Intensive Care Med 27:1788–1797
Pollack MM, Ruttimann UE, Getson P (1988) Pediatric risk of mortality (PRISM) score. Crit Care Med 16:1110–1116
Schoenfeld DA, Bernard GR, Network A (2002) Statistical evaluation of ventilator-free days as an efficacy measure in clinical trials of treatments for acute respiratory distress syndrome. Crit Care Med 30:1772–1777
Hosmer DW, Lemeshow S (2000) Applied logistic regression, 2nd edn. Wiley, New York
Montgomery AB, Stager MA, Carrico CJ, Hudson LD (1985) Causes of mortality in patients with the adult respiratory distress syndrome. Am Rev Respir Dis 132:485–489
Heulitt MJ, Gerhard K, Arnold JH (2008) Mechanical ventilation. In: Nichols DG (ed) Rogers’ textbook of pediatric intensive care, 4th edn. Lippincott Williams and Wilkins, Philadelphia, pp 519–520
Zilberberg MD, Epstein SK (1998) Acute lung injury in the medical ICU: comorbid conditions, age, etiology, and hospital outcome. Am J Respir Crit Care Med 157:1159–1164
Doyle RL, Szaflarski N, Modin GW, Wiener-Kronish JP, Matthay MA (1995) Identification of patients with acute lung injury. Predictors of mortality. Am J Respir Crit Care Med 152:1818–1824
Nuckton TJ, Alonso JA, Kallet RH, Daniel BM, Pittet JF, Eisner MD, Matthay MA (2002) Pulmonary dead-space fraction as a risk factor for death in the acute respiratory distress syndrome. N Engl J Med 346:1281–1286
Trachsel D, McCrindle BW, Nakagawa S, Bohn D (2005) Oxygenation index predicts outcome in children with acute hypoxemic respiratory failure. Am J Respir Crit Care Med 172:206–211
Acknowledgments
Christopher J. L. Newth was partially supported by NIH 5U10 HD050012-04.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Khemani, R.G., Conti, D., Alonzo, T.A. et al. Effect of tidal volume in children with acute hypoxemic respiratory failure. Intensive Care Med 35, 1428–1437 (2009). https://doi.org/10.1007/s00134-009-1527-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00134-009-1527-z




 Median value,
Median value,  25–75%,
25–75%,  minimum−maximum.
minimum−maximum.
