Abstract
Objectives
Any delay in adequate antibiotic treatment compromises the outcome of ventilator-associated pneumonia (VAP). However, the diagnosis and optimal treatment of VAP remain a challenge for intensivists. We assessed the potential impact of using results of routine weekly endotracheal aspirate (EA) cultures to guide initial antibiotic treatment for VAP.
Design and setting
Retrospective analysis of prospectively collected data in a medical–surgical intensive care unit (ICU) of a university hospital.
Patients and methods
We studied 113 VAP episodes and evaluated the concordance between the latest EA and the broncho-alveolar lavage (BAL). We stratified patients into three groups: concordant EA–BAL (concordant group), discordant EA–BAL (discordant group) and EA not performed group. We then compared the adequacy of the antibiotic prescribed initially and outcomes between the three groups.
Measurements and main results
Ninety assessable EA–BAL samples were evaluated. When guided by EA, the initial antibiotic regimen was adequate in 85% of situations, a proportion significantly superior (P < 0.05) to that resulting from application of the ATS guidelines (73%). When clinicians did not have a pre-VAP EA to guide their treatment (EA not performed group), only 61% of treatments were adequate. No significant difference was observed between the three groups for length of mechanical ventilation, length of ICU stay, nonpulmonary nosocomial infections and mortality.
Conclusion
Once-a-week routine quantitative EA cultures may help to improve the adequacy of empiric antibiotic therapy for VAP.
Similar content being viewed by others
Introduction
Ventilator-associated pneumonia (VAP) is the most frequent nosocomial infection in the intensive care unit (ICU). Although its attributable mortality remains controversial [1–4], VAP is associated with an increased length of ICU stay and duration of mechanical ventilation [3–5]. Approximately 50% of all antibiotics are administered for respiratory tract infections [6], and the additional cost of evaluating and treating a patient with VAP is between 8,000 and 10,000 euros per episode [2, 7]. Delaying adequate treatment is associated with a greater risk of hospital mortality [8–10]. To improve the prognosis of VAP, clinicians should prescribe “early and adequate” antibiotic therapy. However, the systematic prescription of broad-spectrum antibiotics to achieve this goal stimulates the emergence of multidrug-resistant pathogens [11].
Recently, Michel et al. [12] proposed twice-weekly examinations of quantitative cultures of endotracheal aspiration (EA) in all mechanically ventilated patients as a means of assisting the choice of initial antibiotic therapies for VAP. The result of the most recently performed EA was similar to that of broncho-alveolar lavage (BAL) in 83% of assessable cases. This strategy, performed in a medical ICU, allowed an initially adequate treatment in 95% of situations and was superior to therapy derived from use of the ATS guidelines [1]. To our knowledge, no study has evaluated the impact of routine once-weekly EA to guide initial treatment in both medical and surgical patients. We therefore evaluated the impact of routine weekly EA in patients who later had VAP confirmed by a BAL. We then described the outcome of these patients. We tested the hypothesis that therapeutic use of EA results obtained prior to the BAL diagnosis would allow an “early and adequate” treatment in a larger proportion of patients with suspected VAP than that obtained by using current guidelines. This study has been presented in abstract form [13].
Methods
This study was conducted from January 1997 to December 2004 in a 12-bed medical–surgical ICU at the Montpellier University Hospital. We performed a retrospective analysis of prospectively collected data. In accordance with the French law, informed consent was not mandatory, given that this observational study did not modify current diagnostic or therapeutic strategies.
Study population
Patients >18 years of age who had received mechanical ventilation for >48 h were prospectively included if VAP was confirmed by a BAL. Their analysis was performed retrospectively. If VAP was clinically suspected, BAL was performed as described elsewhere [1, 14]. The threshold for diagnosing pneumonia was 104 or 103 cfu/ml when an antibiotic therapy was initiated 48 h before BAL [15].
Endotracheal aspirates
Routine surveillance including microbiological analysis of endotracheal aspirates (EAs) was performed using a validated technique upon the patient’s admission and then every Tuesday [1, 14]. The EA analysis was quantitative. Only EAs above the threshold of 104 cfu/ml were compared with BAL.
Our VAP antibiotic policy
Antibiotic therapy, initiated after EA and BAL sampling, was guided by the last available EA specimen culture and then adjusted according to the BAL culture results. For patients with risk factors for multiresistant pathogens [1] and in whom EA has not been performed, the empiric regimen was guided by the ATS guidelines and consisted of a combination of two antipseudomonal antibiotics (noncarbapenem betalactam, fluoroquinolone or aminoglycoside) and a glycopeptide if methicillin-resistant Staphylococcus aureus (MRSA) was suspected (previous history of nursing home or hemodialysis or positive cocci on gram staining). If piperacillin or ticarcillin had previously been used, carbapenem was indicated to limit the risk of inadequacy of our initial treatment. We then compared our local antibiotic strategy (guided by the EA) to the antibiotics that would have been prescribed if EA was not available (as recommended by the ATS guidelines).
Studied parameters
Upon ICU admission, we recorded demographic data and severity scores [16, 17], length of stay in hospital before admission to the ICU, length of mechanical ventilation before VAP and data for antibiotic therapy administered before VAP. On the day of inclusion (day of BAL), we recorded the clinical pulmonary infection score (CPIS) [18], the antibiotic therapy prescribed for the episode of VAP and the results of the last EA performed before the BAL. We assessed the concordance between EA and BAL. Antibiotics prescribed were compared to those that would have been prescribed if the ATS guidelines had been followed.
ATS guidelines stratify patients in two categories: early onset (before day 5 of ventilation) and/or without risk factors for multiresistant pathogens and late onset (after day 5 of ventilation) and/or risk factors for multiresistant pathogens. The antibiotic strategy is then divided into broad spectrum or limited spectrum antibiotics.
Endpoints
The primary endpoint was the concordance of the antibiotic therapy guided by the results of EA with an antibiotic therapy that would have been prescribed if the ATS guidelines had been applied. The antibiotic therapy was judged adequate if all species were sensitive to at least one of the antibiotics prescribed except for Pseudomonas aeruginosa, for which a double therapy was required. Positive concordance between EA and BAL was determined if the same species was identified as having exceeded the thresholds for positivity for both specimens and had the same antibiotic susceptibility patterns [12]. Finally, we compared the clinical outcomes between the three groups.
Statistical analysis
Data are reported using mean values ± SD and the number of cases in percentages. Three groups were analyzed. The first group was constituted by concordant EA and BAL. The second group was constituted by discordant EA and BAL and the third group was characterized by the absence of EA before the BAL. We considered only the first episode of VAP for the analysis. Continuous variables were compared using nonparametric tests of Mann–Withney, Kruskall–Wallis and Friedman. The χ 2 test was used to compare categorical variables. A P value < 0.05 was considered to be statistically significant. Tests were performed by Statview® 5.0 software (SAS Institute, USA).
Results
Patient population
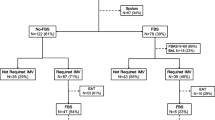
During the study period, 2,036 patients were admitted to our ICU. Sixty-two percent of them received mechanical ventilation for at least 48 h. During the study, 295 BAL were performed on 255 patients for VAP suspicion. Of 137 confirmed VAP cases, we included the 113 first episodes of VAP for the study.
The incidence of VAP was 11%. The main characteristics of the patients are reported in Table 1. There were no statistical differences between groups except for the length of ICU stay before VAP onset, which was shorter when EA was not performed than in the EA–BAL concordant group. Fifty patients developed early-onset pneumonia, whereas 63 patients developed late-onset pneumonia. Fifty-four patients (48%) were already treated by antibiotics upon admission to our ICU (Table 1). The most common pathogen was P. aeruginosa (35/113, 31%).
Concordance between BAL and endotracheal aspirations cultures
Among the 113 VAP episodes, EA was performed for 90 (79%) and not performed for 23. In those 23 cases, patients were not intubated when admitted in ICU. We did not perform a pre-BAL EA, because clinical suspicion of VAP was high and therefore BAL was the first sample performed, or simply because EA was forgotten. Sixty-six of the 90 patients (73%) were already treated by antibiotics when the EA was performed. We only considered EAs obtained within the 7 days preceding the BAL. Six EAs were culture-negative, one below the threshold of 104, and 83 were considered positive. Among the 90 EAs performed, 65 (72%) were concordant with the results of the BAL (concordant group). Twenty-five EAs (28%) were not concordant with the BAL and constituted the “discordant” group, described as follows: no growth on EA the day of the BAL in ten cases (i.e., delay between EA and BAL less than 2 days), negative in seven cases and another microorganism than BAL in eight cases. In the 25 discordant cases, the sensibility of EA to diagnose pathogens found by the BAL was 4/13 (31%) and specificity 4/16 (25%). The EA–BAL concordance rate was not effected by the time of onset of VAP [25/34 (74%) and 40/56 (71%) for early and late onset VAP], the existence of previous antibiotic therapy (81 vs. 77% with or without previous treatment) or the delay between EA and BAL (2 ± 2 days in the concordant group and 3 ± 2 days in the discordant group) (Table 1).
Adequacy of antibiotic therapy according to the strategy used
Antibiotic choice based on the available results of pre-BAL EA cultures was adequate in 77 of 90 patients (85%). In contrast, if the ATS guidelines had been used, the empiric antibiotic treatment would have been adequate in 64 patients (71%) (P = 0.04).
In 13 cases, our strategy allowed an adequate initial treatment, whereas the ATS guidelines did not (Table 2). Although EA and BAL results were discordant in four of those 13 patients (Table 2), the initial antibiotic therapy was adequate, but broad-spectrum antibiotics were not necessary (patients 2, 4 and 6) and treatment was de-escalated just after the BAL result.
In the absence of a pre-BAL EA, initial antibiotic therapies were adequate in only 15/23 cases (65%). The causes of nonadequacy were nonenzymatic beta-lactam P. aeruginosa resistance (two cases), although imipenem was ongoing for an abdominal infection; misclassed “early-onset VAP without any risk factors for multiresistant pathogens” because of occurrence before the fifth day of mechanical ventilation but more than 10 days after admission to the ward (three cases); MRSA without anti-gram positive antibiotic (one case); extended-spectrum beta-lactamase (ESBL)-related VAP with ongoing piperacillin–tazobactam (one case) and VAP related to cefepime-resistant P. aeruginosa, initially treated with this antibiotic (one case). Double antibiotic therapy was performed as frequently as possible with either fluoroquinolone or aminoglycoside. When both antibiotics were ineffective and P. aeruginosa was the pathogen (one case), aerosolized colimycine was added. Monotherapy on ESBL was prescribed once.
Table 3 reports the clinical outcome of the patients. No significant differences were observed between the groups for length of mechanical ventilation, length of ICU stay, nonpulmonary nosocomial infections and mortality.
Discussion
The main findings of the study are that (1) the results of the most recent performed EA were concordant with the BAL in 65 patients (72% of assessable cases) and discordant in 25 (28%); (2) our antibiotic strategy based on the pre-VAP EA was adequate in 85% of situations, significantly superior to that obtained with the ATS guidelines [1]; (3) when the clinicians did not have a pre-VAP EA to guide their treatment (23 cases, EA nonperformed), only 61% of overall antibiotic strategies were adequate; (4) there were no statistical differences in duration of mechanical ventilation, length of stay in ICU, extrapulmonary infections or mortality between patients whose treatment was based on a pre-BAL EA (concordant and discordant groups; local strategy) and those whose treatment was not EA-based (EA not performed). Few studies have reported the importance of routine colonization surveillance to predict the treatment or even the pathogens implicated in VAP.
Our study is in accordance with a study showing that tracheal colonization was an independent predictive factor of VAP in head trauma patients [19] and with a study showing a 83% concordance between BAL and the results of twice weekly EA [12], but not with those published by Hayon et al., who reported a poor correlation between routine multisite surveillance and pathogens diagnosed by the BAL [20]. More recently, Depuydt et al. [21] showed that thrice weekly EA surveillance allowed 69% of concordance between EA and blood culture in patients presenting bacteriemic nosocomial pneumonia. Our study is the first, to our knowledge, to evaluate the impact of once weekly EA to guide the initial treatment of VAP in 113 medical and surgical patients.
Our strategy allowed us to improve the adequacy of initial treatment compared to the treatment that would have been prescribed if the ATS guidelines had been followed in 13 patients (Table 2). Our results are in accordance with a previous study who reported the superiority of initial treatment guided by twice weekly EA over the ATS guidelines [1].
In our ICU, most patients were already on antibiotics and came from wards where they developed a VAP; they presented a high incidence of risk factors for multiresistant pathogens as defined by the ATS (Table 1). This particularity of some ICUs has been reported elsewhere [5, 22–24], and although it may not be applicable to all ICUs, it brings to light the importance of knowing local ecology of nosocomial infections. Because delays in the administration of adequate therapy have been associated with excess mortality from severe infections in the ICU, the prompt administration of empiric therapy for patients likely to have VAP is essential [25–27]. However, the extensive use of antibiotics, especially broad-spectrum antibiotics, is associated with the emergence of resistant pathogens [23]. In our unit, initial antibiotic therapy is given without delay after the BAL, but is oriented by the results of airway colonization diagnosed by the pre-BAL EA.
Although our strategy based on the colonization was more frequently adequate than the one recommended by the ATS (P < 0.05), the ATS-guideline strategy was more often appropriate than that prescribed when no EA was available. Among the 23 cases where the clinicians did not have a pre-BAL EA, the initial antibiotic therapy was not adequate in eight situations. In five of them, the cause of discrepancy was related to the nonadhesion to the international guidelines, a finding well described elsewhere [28].
Length of stay before ICU was longer for the patients of the EA nonperformed group than for those of concordant group (Table 3). Length of ICU stay and length of mechanical ventilation before VAP were shorter for the patients of the EA nonperformed group than for those of the concordant group. This was mainly due to the decision to bypass the EA and to perform a BAL in the first intention.
Limitations of the study
Some limitations of the present study must be noted. It is an epidemiological, retrospective analysis of a prospectively acquired database, which reflects the results of a single center.
In our study, we did not consider the impact of routine surveillance of nasal carriage or rectal screening, which could contribute to improve the proportion of initially adequate antibiotic therapy [21], but may result in an increase of broad-spectrum antibiotic use [20]. We did not consider EAs to be positive when below the threshold of 104 cfu/ml; however, only one EA below this threshold was actually positive, without any consequences on our results.
The overall mortality was nearly 30% and was not different between groups (Table 3). The primary endpoint of this retrospective study was to compare different antibiotic strategies and not to evaluate the mortality; the analysis of this small cohort may explain this absence of difference for this outcome.
Conclusion
This study showed that an initial antibiotic treatment for VAP guided by the results of weekly systematic pre-VAP EA allowed an adequate treatment in 85% of patients. Routine surveillance cultures may limit the inadequate prescription of broad spectrum antibiotics and could perhaps help to prescribe a better targeted antibiotic therapy in patients infected with multiresistant pathogens. However, the ATS guidelines [1] remain the gold standard to initiate antibiotic therapy for VAP when the clinicians do not have any data on airway colonization. The impact of this strategy “early and guided by lower respiratory tract colonization” on patient outcomes and resistance rates is still unknown and needs to be determined by a randomized clinical trial.
Abbreviations
- VAP:
-
Ventilator-associated pneumonia
- EA:
-
Endotracheal aspiration
- BAL:
-
Broncho-alveolar lavage
- ICU:
-
Intensive care unit
- SAPS II:
-
Simplified acute physiology score II
- SOFA:
-
Sequential organ failure assessment score
- CPIS:
-
Clinical pulmonary infectious score
- ESBL:
-
Extended-spectrum beta-lactamase
- MRSA:
-
Methicillin-resistant Staphylococcus aureus
- MSSA:
-
Methicillin-sensible Staphylococcus aureus
- FiO2 :
-
Fraction of inspired oxygen
References
American Thoracic Society; Infectious Diseases Society of America (2005) Guidelines for the management of adults with hospital-acquired, ventilator-associated, and healthcare-associated pneumonia. Am J Respir Crit Care Med 171:388–416
Fagon JY, Chastre J, Hance AJ, Montravers P, Novara A, Gibert C (1993) Nosocomial pneumonia in ventilated patients: a cohort study evaluating attributable mortality and hospital stay. Am J Med 94:281–288
Papazian L, Bregeon F, Thirion X, Gregoire R, Saux P, Denis JP, Perin G, Charrel J, Dumon JF, Affray JP, Gouin F (1996) Effect of ventilator-associated pneumonia on mortality and morbidity. Am J Respir Crit Care Med 154:91–97
Valles J, Pobo A, Garcia-Esquirol O, Mariscal D, Real J, Fernandez R (2007) Excess ICU mortality attributable to ventilator-associated pneumonia: the role of early vs late onset. Intensive Care Med 33:1363–1368
Rello J, Ollendorf DA, Oster G, Vera-Llonch M, Bellm L, Redman R, Kollef MH (2002) Epidemiology and outcomes of ventilator-associated pneumonia in a large US database. Chest 122:2115–2121
Richards MJ, Edwards JR, Culver DH, Gaynes RP (1999) Nosocomial infections in medical intensive care units in the United States. National Nosocomial Infections Surveillance System. Crit Care Med 27:887–892
Warren DK, Shukla SJ, Olsen MA, Kollef MH, Hollenbeak CS, Cox MJ, Cohen MM, Fraser VJ (2003) Outcome and attributable cost of ventilator-associated pneumonia among intensive care unit patients in a suburban medical center. Crit Care Med 31:1312–1317
Dupont H, Mentec H, Sollet JP, Bleichner G (2001) Impact of appropriateness of initial antibiotic therapy on the outcome of ventilator-associated pneumonia. Intensive Care Med 27:355–362
Heyland DK, Cook DJ, Griffith L, Keenan SP, Brun-Buisson C (1999) The attributable morbidity and mortality of ventilator-associated pneumonia in the critically ill patient. The Canadian Critical Trials Group. Am J Respir Crit Care Med 159:1249–1256
Kollef MH (1999) The prevention of ventilator-associated pneumonia. N Engl J Med 340:627–634
Kollef MH, Fraser VJ (2001) Antibiotic resistance in the intensive care unit. Ann Intern Med 134:298–314
Michel F, Franceschini B, Berger P, Arnal JM, Gainnier M, Sainty JM, Papazian L (2005) Early antibiotic treatment for BAL-confirmed ventilator-associated pneumonia: a role for routine endotracheal aspirate cultures. Chest 127:589–597
Jung B, Jaber S, Perrigault P, Sebbane M, Verzilli D, Jeanpierre H, Eledjam J (2006) Weekly systematic endotracheal aspiration allows early and adequate antibiotic therapy for VAP. Intensive Care Med 32:S102
Papazian L, Martin C, Meric B, Dumon JF, Gouin F (1993) A reappraisal of blind bronchial sampling in the microbiologic diagnosis of nosocomial bronchopneumonia. A comparative study in ventilated patients. Chest 103:236–242
Souweine B, Veber B, Bedos JP, Gachot B, Dombret MC, Regnier B, Wolff M (1998) Diagnostic accuracy of protected specimen brush and bronchoalveolar lavage in nosocomial pneumonia: impact of previous antimicrobial treatments. Crit Care Med 26:236–244
Le Gall JR, Lemeshow S, Saulnier F (1993) A new simplified acute physiology score (SAPS II) based on a european/north American multicenter study. JAMA 270:2957–2963
Vincent JL, Moreno R, Takala J, Willatts S, De Mendonca A, Bruining H, Reinhart CK, Suter PM, Thijs LG (1996) The SOFA (Sepsis-related Organ Failure Assessment) score to describe organ dysfunction/failure. On behalf of the Working Group on Sepsis-Related Problems of the European Society of Intensive Care Medicine. Intensive Care Med 22:707–710
Pugin J, Auckenthaler R, Mili N, Janssens JP, Lew PD, Suter PM (1991) Diagnosis of ventilator-associated pneumonia by bacteriologic analysis of bronchoscopic and nonbronchoscopic “blind” bronchoalveolar lavage fluid. Am Rev Respir Dis 143:1121–1129
Sirvent JM, Torres A, Vidaur L, Armengol J, de Batlle J, Bonet A (2000) Tracheal colonisation within 24 h of intubation in patients with head trauma: risk factor for developing early-onset ventilator-associated pneumonia. Intensive Care Med 26:1369–1372
Hayon J, Figliolini C, Combes A, Trouillet JL, Kassis N, Dombret MC, Gibert C, Chastre J (2002) Role of serial routine microbiologic culture results in the initial management of ventilator-associated pneumonia. Am J Respir Crit Care Med 165:41–46
Depuydt P, Benoit D, Vogelaers D, Decruyenaere J, Vandijck D, Claeys G, Verschraegen G, Blot S (2008) Systematic surveillance cultures as a toolto predict involvement of multidrug antibiotic resistant bacteria in ventilator-associated pneumonia. Intensive Care Med 34(4):675–682
Chastre J (2005) Antibiotic prescribing for ventilator-associated pneumonia: get it right from the beginning but be able to rapidly deescalate. Intensive Care Med 31:1463–1465
Giantsou E, Liratzopoulos N, Efraimidou E, Panopoulou M, Alepopoulou E, Kartali-Ktenidou S, Minopoulos GI, Zakynthinos S, Manolas KI (2005) Both early-onset and late-onset ventilator-associated pneumonia are caused mainly by potentially multiresistant bacteria. Intensive Care Med 31:1488–1494
Rello J, Diaz E, Roque M, Valles J (1999) Risk factors for developing pneumonia within 48 hours of intubation. Am J Respir Crit Care Med 159:1742–1746
Combes A, Luyt CE, Fagon JY, Wolff M, Trouillet JL, Chastre J (2006) Impact of piperacillin resistance on the outcome of Pseudomonas ventilator-associated pneumonia. Intensive Care Med 32:1970–1978
Ibrahim EH, Sherman G, Ward S, Fraser VJ, Kollef MH (2000) The influence of inadequate antimicrobial treatment of bloodstream infections on patient outcomes in the ICU setting. Chest 118:146–155
Leone M, Bourgoin A, Cambon S, Dubuc M, Albanese J, Martin C (2003) Empirical antimicrobial therapy of septic shock patients: adequacy and impact on the outcome. Crit Care Med 31:462–467
Young MP, Manning HL, Wilson DL, Mette SA, Riker RR, Leiter JC, Liu SK, Bates JT, Parsons PE (2004) Ventilation of patients with acute lung injury and acute respiratory distress syndrome: has new evidence changed clinical practice? Crit Care Med 32:1260–1265
Acknowledgments
The authors are grateful to Patrick McSweeny for his English editing.
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is discussed in the editorial available at: doi:10.1007/s00134-008-1249-7.
Presented in part at the 2006 Annual Meeting of European Society of Intensive Care Medicine (Barcelona, Spain, 24–27 September 2006).
All authors declare the absence of any involvement in any organization with a direct financial interest in the subject of the manuscript.
Rights and permissions
About this article
Cite this article
Jung, B., Sebbane, M., Chanques, G. et al. Previous endotracheal aspirate allows guiding the initial treatment of ventilator-associated pneumonia. Intensive Care Med 35, 101–107 (2009). https://doi.org/10.1007/s00134-008-1248-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00134-008-1248-8