Abstract
Objective
To compare two solutions for fluid resuscitation in post-coronary artery bypass grafting (CABG) surgery patients: Ringer’s lactate (RL) versus a new solution containing half-molar sodium-lactate (HL).
Design
Prospective randomized open label study.
Setting
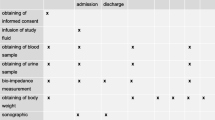
The first 12 h post-CABG surgery in an intensive care unit (ICU).
Patients
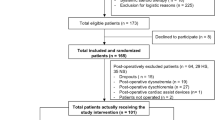
There were 230 patients enrolled in the study: 208 were analyzed, with 109 from the HL group and 99 from the RL group.
Interventions
Patients received over the first 12 h post-CABG 10 ml kg BW−1 HL solution in the HL group versus 30 ml kg BW−1 of RL solution in the RL group.
Measurements and results
Hemodynamic status, body fluid balance and inotrope utilization were compared in the two groups. Post-operative cardiac index increase was significantly higher in HL than in RL (P = 0.02), while mean arterial pressure and other hemodynamic parameters were comparable together with urinary output, indicating similar tissue perfusion in both the groups despite a much lower fluid infusion in the HL group. Therefore, a significant negative fluid balance was achieved in the HL but not in the RL group (−790 ± 71 vs. +43 ± 115 mL 12 h−1, P < 0.0001 for HL and RL, respectively). None of the enrolled patients exhibited side effects related to the treatment.
Conclusion
Half-molar lactate solution is effective for fluid resuscitation in post-CABG patients. Compared to Ringer’s Lactate, its use results in a significantly higher cardiac index with less volume being infused, resulting in a very negative post-operative body fluid balance.


Similar content being viewed by others
References
Boldt J (2003) New light on intravascular volume replacement regimens: what did we learn from the past three years? Anesth Analg 97:1595–1604
Utley JR (1990) Pathophysiology of cardiopulmonary bypass: current issues. J Card Surg 5:177–189
Koller ME, Bert J, Segadal L, Reed RK (1992) Estimation of total body fluid shifts between plasma and interstitium in man during extracorporeal circulation. Acta Anaesthesiol Scand 36:255–259
Stone JG, Hoar PF, Khambatta HJ (1983) Influence of volume loading on intraoperative hemodynamics and perioperative fluid retention in patients with valvular regurgitation undergoing prosthetic replacement. Am J Cardiol 52:530–533
Cross JS, Gruber DP, Burchard KW, Singh AK, Moran JM, Gann DS (1989) Hypertonic saline fluid therapy following surgery: a prospective study. J Trauma 29:817–825 Discussion 825–816
Schierhout G, Roberts I (1998) Fluid resuscitation with colloid or crystalloid solutions in critically ill patients: a systematic review of randomised trials. BMJ 316:961–964
Webb AR (1999) Crystalloid or colloid for resuscitation. Are we any the wiser? Crit Care 3:R25–R28
Boldt J (2005) Volume therapy in cardiac surgery. Ann Card Anaesth 8:104–116
Moon PF, Hollyfield-Gilbert MA, Myers TL, Kramer GC (1994) Effects of isotonic crystalloid resuscitation on fluid compartments in hemorrhaged rats. Shock 2:355–361
Steele A, Gowrishankar M, Abrahamson S, Mazer CD, Feldman RD, Halperin ML (1997) Postoperative hyponatremia despite near-isotonic saline infusion: a phenomenon of desalination. Ann Intern Med 126:20–25
Cross JS, Gruber DP, Gann DS, Singh AK, Moran JM, Burchard KW (1989) Hypertonic saline attenuates the hormonal response to injury. Ann Surg 209:684–691 Discussions 691–682
Velasco IT, Pontieri V, Rocha e Silva M, Lopes OU Jr (1980) Hyperosmotic NaCl and severe hemorrhagic shock. Am J Physiol 239:H664–H673
Vassar MJ, Perry CA, Holcroft JW (1990) Analysis of potential risks associated with 7.5% sodium chloride resuscitation of traumatic shock. Arch Surg 125:1309–1315
Reed RL 2nd, Johnston TD, Chen Y, Fischer RP (1991) Hypertonic saline alters plasma clotting times and platelet aggregation. J Trauma 31:8–14
Huang PP, Stucky FS, Dimick AR, Treat RC, Bessey PQ, Rue LW (1995) Hypertonic sodium resuscitation is associated with renal failure and death. Ann Surg 221:543–554 (Discussion 554–547)
Mustafa I, Leverve XM (2002) Metabolic and hemodynamic effects of hypertonic solutions: sodium-lactate versus sodium chloride infusion in postoperative patients. Shock 18:306–310
Deitch EA, Shi HP, Feketeova E, Hauser CJ, Xu DZ (2003) Hypertonic saline resuscitation limits neutrophil activation after trauma-hemorrhagic shock. Shock 19:328–333
Bakker J, Gris P, Coffernils M, Kahn RJ, Vincent JL (1996) Serial blood lactate levels can predict the development of multiple organ failure following septic shock. Am J Surg 171:221–226
Munoz R, Laussen PC, Palacio G, Zienko L, Piercey G, Wessel DL (2000) Changes in whole blood lactate levels during cardiopulmonary bypass for surgery for congenital cardiac disease: an early indicator of morbidity and mortality. J Thorac Cardiovasc Surg 119:155–162
Chiolero RL, Revelly JP, Leverve X, Gersbach P, Cayeux MC, Berger MM, Tappy L (2000) Effects of cardiogenic shock on lactate and glucose metabolism after heart surgery. Crit Care Med 28:3784–3791
Leverve XM (1999) Energy metabolism in critically ill patients: lactate is a major oxidizable substrate. Curr Opin Clin Nutr Metab Care 2:165–169
Leverve X, Mustafa I, Péronnet F (1998) Pivotal role of lactate in aerobic metabolism. In: Yearbook of intensive care and emergency medicine. J. V. Springer, Berlin, pp 588–596
Mustafa I, Roth H, Hanafiah A, Hakim T, Anwar M, Siregar E, Leverve XM (2003) Effect of cardiopulmonary bypass on lactate metabolism. Intensive Care Med 29:1279–1285
Revelly JP, Tappy L, Martinez A, Bollmann M, Cayeux MC, Berger MM, Chiolero RL (2005) Lactate and glucose metabolism in severe sepsis and cardiogenic shock. Crit Care Med 33:2235–2240
King P, Kong MF, Parkin H, MacDonald IA, Barber C, Tattersall RB (1998) Intravenous lactate prevents cerebral dysfunction during hypoglycaemia in insulin-dependent diabetes mellitus. Clin Sci (Lond) 94:157–163
Maran A, Crepaldi C, Trupiani S, Lucca T, Jori E, Macdonald IA, Tiengo A, Avogaro A, Del Prato S (2000) Brain function rescue effect of lactate following hypoglycaemia is not an adaptation process in both normal and type I diabetic subjects. Diabetologia 43:733–741
Maran A, Cranston I, Lomas J, Macdonald I, Amiel SA (1994) Protection by lactate of cerebral function during hypoglycaemia. Lancet 343:16–20
Schurr A (2006) Lactate: the ultimate cerebral oxidative energy substrate? J Cereb Blood Flow Metab 26:142–152
Schurr A, Payne RS, Miller JJ, Rigor BM (1997) Brain lactate is an obligatory aerobic energy substrate for functional recovery after hypoxia: further in vitro validation. J Neurochem 69:423–426
Rice AC, Zsoldos R, Chen T, Wilson MS, Alessandri B, Hamm RJ, Bullock MR (2002) Lactate administration attenuates cognitive deficits following traumatic brain injury. Brain Res 928:156–159
Philp A, Macdonald AL, Watt PW (2005) Lactate—a signal coordinating cell and systemic function. J Exp Biol 208:4561–4575
Schurr A (2002) Lactate, glucose and energy metabolism in the ischemic brain (Review). Int J Mol Med 10:131–136
Schurr A, Payne RS, Miller JJ, Rigor BM (1997) Brain lactate, not glucose, fuels the recovery of synaptic function from hypoxia upon reoxygenation: an in vitro study. Brain Res 744:105–111
Barbee RW, Kline JA, Watts JA (2000) Depletion of lactate by dichloroacetate reduces cardiac efficiency after hemorrhagic shock. Shock 14:208–214
Kline JA, Thornton LR, Lopaschuk GD, Barbee RW, Watts JA (2000) Lactate improves cardiac efficiency after hemorrhagic shock. Shock 14:215–221
Levy B, Mansart A, Montemont C, Gibot S, Mallie JP, Regnault V, Lecompte T, Lacolley P (2007) Myocardial lactate deprivation is associated with decreased cardiovascular performance, decreased myocardial energetics, and early death in endotoxic shock. Intensive Care Med 33:495–502
Chiolero R, Tappy L, Gillet M, Revelly JP, Roth H, Cayeux C, Schneiter P, Leverve X (1999) Effect of major hepatectomy on glucose and lactate metabolism. Ann Surg 229:505–513
Connor H, Woods HF, Ledingham JG, Murray JD (1982) A model of L(+)-lactate metabolism in normal man. Ann Nutr Metab 26:254–263
Lobo DN, Dube MG, Neal KR, Allison SP, Rowlands BJ (2002) Peri-operative fluid and electrolyte management: a survey of consultant surgeons in the UK. Ann R Coll Surg Engl 84:156–160
Holte K, Sharrock NE, Kehlet H (2002) Pathophysiology and clinical implications of perioperative fluid excess. Br J Anaesth 89:622–632
Jarvela K, Kaukinen S (2002) Hypertonic saline (7.5%) decreases perioperative weight gain following cardiac surgery. J Cardiothorac Vasc Anesth 16:43–46
Jarvela K, Koskinen M, Kaukinen S, Koobi T (2001) Effects of hypertonic saline (7.5%) on extracellular fluid volumes compared with normal saline (0.9%) and 6% hydroxyethyl starch after aortocoronary bypass graft surgery. J Cardiothorac Vasc Anesth 15:210–215
Acknowledgments
This study was sponsored by Innogene Kalbiotech, Pte. Ltd., 24 Raffles Place 27 – 06 Clifford Centre, Singapore 048621. The half-molar sodium-lactate containing solution is patented (WO 2004/096204 -11/11/04, Gazette 2004/46) and registered (Totilac™). We are grateful to Mr. Gareth Butt for his English corrections to this paper.
Disclosure of interest
Professor Xavier Leverve is member of the Innogene International Scientific Board Advisors.
Author information
Authors and Affiliations
Corresponding author
Additional information
This clinical research work was performed at the Intensive Care Unit, Harapan Kita National Cardiovascular Center, Jakarta, Indonesia.
The present research work was the last clinical study carried out by Doctor Iqbal Mustafa, Head of the intensive care unit of Harapan Kita National Cardiovascular Center, shortly before he passed away so prematurely. This paper is dedicated to his memory and we hereby acknowledge the leading part he played in its realization.
This article is discussed in the editorial available at: doi:10.1007/s00134-008-1166-9.
Electronic supplementary material
Below is the link to the electronic supplementary material.
134_2008_1165_MOESM1_ESM.ppt
Fig. 1bis (electronic supplementary material). Effect of RL versus HL on heart rate, cardiac filling pressures, mean pulmonary artery pressure and vascular resistance. Open symbols: RL, closed symbols: HL. Panel A: Heart Rate (B/min); Panel B: Central Venous Pressure (CVP), mm Hg; panel C: Mean Pulmonary Artery Pressure (MPAP), mm Hg; Panel D: Pulmonary Artery Occluded Pressure (PAOP), mm Hg; panel E: Pulmonary Vascular Resistance Index (PVRI), dyne.s.cm−5; panel DF: Systemic Vascular Resistance Index (SVRI), dyne.s.cm−5. Results are expressed as means±sem; statistical comparisons with ANOVA for repeated measures: NS for all parameters, PVRI excepted: p=0.0367 (post hoc analysis: 2h p=0.004; 3h p=0.023; 5h p=0.011) (PPT 271 kb)
Rights and permissions
About this article
Cite this article
Leverve, X.M., Boon, C., Hakim, T. et al. Half-molar sodium-lactate solution has a beneficial effect in patients after coronary artery bypass grafting. Intensive Care Med 34, 1796–1803 (2008). https://doi.org/10.1007/s00134-008-1165-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00134-008-1165-x