Abstract
Objective
To investigate whether electrical impedance tomography (EIT) is capable of monitoring regional lung recruitment and lung collapse during a positive end-expiratory pressure (PEEP) trial.
Design
Experimental animal study of acute lung injury.
Subject
Six pigs with saline-lavage-induced acute lung injury.
Interventions
An incremental and decremental PEEP trial at ten pressure levels was performed. Ventilatory, gas exchange, and hemodynamic parameters were automatically recorded. EIT and computed tomography (CT) scans of the same slice were simultaneously taken at each PEEP level.
Measurements and results
A significant correlation between EIT and CT analyses of end-expiratory gas volumes (r = 0.98 up to 0.99) and tidal volumes (r = 0.55 up to r = 0.88) could be demonstrated. Changes in global and regional tidal volumes and arterial oxygenation (PaO2/FiO2) demonstrated recruitment/derecruitment during the trial, but at different onsets. During the decremental trial, derecruitment first occurred in dependent lung areas. This was indicated by lowered regional tidal volumes measured in this area and by a decrease of PaO2/FiO2. At the same time, the global tidal volume still continued to increase, because the increase of ventilation of the non-dependent areas was higher than the loss in the dependent areas. This indicates that opposing regional changes might cancel each other out when combined in a global parameter.
Conclusions
EIT is suitable for monitoring the dynamic effects of PEEP variations on the regional change of tidal volume. It is superior to global ventilation parameters in assessing the beginning of alveolar recruitment and lung collapse.
Similar content being viewed by others
Introduction
In acute lung injury (ALI) and acute respiratory distress syndrome (ARDS), ventilation in general is inhomogeneous [1]; this leads to an increased ventilation–perfusion mismatch [2], an increased dead-space fraction [3], and possible ventilator-induced lung injury (VILI) [4, 5]. Global indices such as gas exchange parameters, pressure–volume curves, or pulmonary compliance are important parameters for assessing lung function during ventilation. However, they provide only limited information regarding regional dynamic lung mechanisms [6, 7].
Clinical management of ARDS can be successfully examined by computed tomography (CT) analysis, which allows an assessment of the pattern of opacities of the injured lungs [8–10]. However, CT scans expose the patient to radiation. In addition, CT diagnosis is a static method and not a bedside technique. By contrast, dynamic information regarding ventilation can be obtained non-invasively by using electrical impedance tomography (EIT) [11, 12]. Although it is currently not a routine clinical technique, EIT is capable of measuring global [11] and regional [13–15] ventilation. To date, only a few studies on the potential of EIT in ALI or ARDS are available, even though evidence is accumulating that EIT is useful in monitoring regional lung recruitment and derecruitment [16–18]. The value of bedside regional EIT measurements for determining the effect of positive end-expiratory pressure (PEEP) in comparison with traditional indirect methods, such as arterial oxygenation or global tidal volumes, is still unknown.
Thus, the aim of the present study was to evaluate EIT in terms of its potential to assess changes in regional and global tidal volumes in injured lungs by comparing it with simultaneously recorded CT scans taken at the end of inspiration and expiration during a PEEP trial. Additionally, we examined changes in regional EIT measurements and global ventilation parameters regarding their ability to detect the beginning of pulmonary recruitment and lung collapse. Results were presented partly during the 2006 ISICEM [19] and ESICM meeting [20].
Material and methods
Animal preparation
After approval by the local authority for animal research, six pigs (25–34 kg) were included in the study. Following premedication (10 mg/kg ketamine, 2 mg/kg xylazine, 15 μg/kg atropine), anesthesia was induced with etomidate (0.4 mg/kg). After endotracheal intubation (6.5-mm tube) anesthesia was maintained with propofol (8 mg/kg/h) and ketamine (10 mg/kg/h). Muscle paralysis was maintained with pancuronium (0.2 mg/kg, repetitive 0.1 mg/kg).
All measurements were made using a medically approved Panel PC (POC-153; Advantech, Taiwan), an electrically controllable ventilator (Servo 300; Siemens-Elema, Sweden), an online blood gas analyzer (TrendCare Satellite; Diametrics Medical, UK), a monitor for hemodynamic parameters (Sirecust 1281; Siemens, Germany) and a capnograph (CO2SMO+; Respironics, USA). An EIT GoeMF II prototype system (EIT evaluation kit; Dräger Medical, Germany) with 16 electrodes (Ag/AgCl EEG-EMG electrodes, CDES002822; spes medica, Italy) at a juxtadiaphragmatic level and a reference electrode approximately 10 cm below the electrode plane was used. The frequency of the injected currents was 50 kHz at an amplitude of 5 mArms. A complete EIT measurement was made every 80 ms (12.5 frames/s) [21–23]. All parameters were continually recorded using the graphical development environment LabVIEW® (National Instruments, USA).
The animals were initially ventilated in a volume-controlled mode [tidal volume (VT) 8 ml/kg, PEEP 5 cmH2O, I:E 1:1, respiration rate 16–20 breaths/min and inspiratory fraction of oxygen (FiO2) 0.5] in supine position. Volume deficits were compensated by using 8 ml/kg isotonic or 6% hydroxyethyl starch solution. Infusion of isotonic solution was continued at a rate of 5 ml/kg/h, and the intravascular volume was adapted to adjust the central venous pressure to ≥ 12 mmHg.
Experimental protocol
After baseline measurements at a PEEP of 10 cmH2O [pressure-controlled ventilation (PCV), driving pressure 8 cmH2O, respiratory rate 25 breaths/min, I:E 1:1, FiO2 = 0.5] had been performed, ALI was induced by repetitive lung lavage with warmed saline (8–12 l). During the procedure, the animals were ventilated with a PEEP of 5 cmH2O and a FiO2 of 1.0. The lavage was repeated until the PaO2 decreased to 100 mmHg for at least 1 h [24].
Thereafter, an automatic stepwise incremental and decremental PEEP trial of respective 2 min duration at ten pressure levels (i. e. 20 min overall) was performed (PEEP = 10, 15, 20, 25, 30, 25, 20, 15, 10, 5 cmH2O) (Fig. 1). During the maneuver, the animals were ventilated with a FiO2 of 1.0 using the same ventilation mode and parameters as during the baseline measurements. The body temperature of the animals was held constant by body surface warming.
Example of an original EIT curve during the PEEP trial: the curve represents the level of measured sum of relative impedance change (rel. ΔZ) and its tidal change (Tidal Variation, TV). At the end of each PEEP level, two triggered dynamic CT scans were performed simultaneously with the EIT measurement at end-inspiration and end-expiration
EIT off-line data analysis
We used a prototype software (Dräger Medical, Germany) with a modified back-projection algorithm [23]. Additionally, all data were low-pass filtered with a cut-off frequency of 55 beats/min in order to reduce cardiac-related oscillations caused by the heartbeats.
The calculated images (32 × 32 color-scaled matrix) are relative EIT scans referenced to the steady-state end-expiratory values after lavage as a baseline vector. Therefore, only the relative impedance change (rel. ΔZ) was measured. The difference between rel. ΔZ at the end of inspiration and at the end of expiration is defined as tidal variation (TV) (Fig. 1).
To compare TV with the CT-based tidal change of gas volume (ΔGV) of all pigs, TV was normalized with VT given by the ventilator. Due to high correlation coefficients per animal of the corresponding values a linear regression model was fitted ([TV, VT]), and parameters were estimated using an ordinary least-squares procedure. Then, the estimated linear relationship was used to predict future respond values (VT) based on the independent variable (TV).
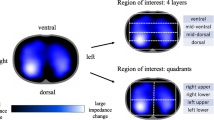
Regional rel. ΔZ (ΔZreg) and TV (TVreg) were calculated by offline analysis in four regions of interest (ROIs; size 32 × 5 pixels), which were adjusted to the asymmetric localization of the lung within the chest of the pig (Fig. 2).
Regions of interest (ROIs) in computed tomography (CT) and functional EIT (fEIT): Each pixel of the fEIT image is gray-tone-encoded. Black pixels describe areas with no ventilation, and white pixels indicate areas with high ventilation. In CT scans, the ventral-dorsal height of each lung was divided into four equal intervals, indicating the four ROIs
For the evaluation of the regional and global ventilation distribution, functional EIT (fEIT) images were used [25]. After normalization with VT, the normalized fEIT images at the end of each PEEP step were subtracted from those at the end of the previous PEEP step (resulting in nine differential fEIT images, dfEIT). Consequently, these dfEIT images qualitatively depicted regional changes of VT as ΔVT (i. e. an increase, a decrease, or no change) when the PEEP level was elevated or reduced. In order to quantify these changes for each dfEIT image, all pixels with positive values and all pixels with negative values were summed separately and displayed by a color scale. Thus were described the tidal volume gain (TVG) and loss (TVL) for the pulmonary cross-section and their dependent/non-dependent lung areas. The advantage of splitting ΔVT into TVG and TVL lies in the possibility of detecting inhomogeneous changes of regional tidal volumes.
CT scanning and analysis
Simultaneously to the EIT measurements, a CT scanner (Toshiba Aquilion 4; Toshiba, Japan) was automatically turned on at the end of each PEEP step to obtain reference images in the electrode plane (Fig. 1). End-inspiratory and end-expiratory CT scans were taken, triggered using the standard ECG input port of the scanner. The trigger impulses were generated by analyzing the flow signal of the respirator. Exposures were taken at 120 kV and 100 mA (collimation 2 mm, 512 × 512 matrix) and were reconstructed with a high-resolution reconstruction algorithm. The ventral–dorsal height of each lung was manually divided into four equal intervals (Fig. 2), and the mean lung densities in Hounsfield units (HU) at the end of expiration and inspiration were obtained for the whole slice and for all four ROIs. The global and regional CT gas volumes (GV; GVreg) and tidal volumes (ΔGV; ΔGVreg) were calculated [26]; delta-CT scans were obtained by means of subtraction.
Statistics
Data were analyzed by means ± SD, median, and range with SPSS® 14.0 (SPSS, USA). For changes during the PEEP trial, means were analyzed, and if a normal distribution was unlikely and errors from multiple testing had to be avoided, we used the Friedman analysis and, in the case of significance (p < 0.05), the Wilcoxon–Wilcox test (p < 0.05, two sided). The data obtained by linear regression analysis and the calculation of the correlation coefficient presented the relationship between EIT and CT measurements. To analyze the nonlinear relationship between gas/tidal volumes and time, orthogonal polynomial trend analysis was performed. Then, parameters up to degree two were compared by a Wilcoxon test for two dependent samples. Differences between the parameters were defined as statistically significant with p < 0.05.
Results
Global parameters
Ventilatory, gas exchange, and hemodynamic parameters are shown in Table 1. With regard to the PEEP trial, a significant depression of the mean arterial pressure occurred (from 113.3 ± 17.2 to 88.4 ± 18.1 mmHg). During the incremental part, PaO2 increased from 78 ± 43.6 to 551.7 ± 70.8 mmHg. At the same time, VT increased strikingly (from 59.3 ± 78.7 to 265.6 ± 57.9 ml). During the decremental part, PaO2 started to drop again as soon as the PEEP level was reduced to 10 cmH2O (375 ± 146.6 mmHg). VT, however, did not decrease until the PEEP level was reduced to 5 cmH2O (Fig. 3). Compared to the beginning of the trial, VT (from 99.3 ± 45.7 to 189.4 ± 48.8 ml) as well as static respiratory compliance (Crs) (from 11.1 ± 7.5 to 20.6 ± 11.9 ml/cmH2O) were still significantly higher at the end of the trial.
Course of tidal change of gas volume (ΔGV), normalized tidal variation (TV*), tidal volume VT (ventilator), and oxygenation (PaO2/FiO2) at baseline and during the PEEP trial: the dynamic behavior of VT and PaO2/FiO2 is displayed by using continuous lines (median). The box plots show the median, upper, and lower quartiles of the PaO2/FiO2 and VT values at the end of each PEEP step. The whiskers indicate the range of minimum and maximum (n = 6). * (PaO2/FiO2, VT), † (TV*), § (ΔGV); p < 0.05 is considered a statistically significant increase between initial and subsequent PEEP levels
Comparison of EIT and CT scans
A significant correlation was found between rel. ΔZ and GV at the end of inspiration (r = 0.75, p < 0.01) and expiration (r = 0.78, p < 0.01) when combining the data of all animals. Comparing end-expiratory rel. ΔZ and GV of each animal individually showed much higher linear correlations (from r = 0.98 to r = 0.99, p < 0.01). Furthermore, TV and CT-based tidal volumes (ΔGV) showed a linear correlation (from r = 0.55 to r = 0.88, p < 0.01). Trend analysis of both methods regarding increase and decrease of global tidal volumes showed no significant differences. The linear trend varied in ROI 1 and 2 (p = 0.028) and the quadratic trend varied in ROI 3 (p = 0.046), but they were indiscriminative in ROI 4.
Global and regional changes of ventilation in EIT and CT scans
During the PEEP trial, EIT and CT were able to monitor global and regional changes in tidal volumes. Like VT, the values for TV* and ΔGV improved significantly at the decremental part of the PEEP trial when compared with the initial PEEP level (Fig. 3).
The impact of PEEP changes on the regional tidal volumes was greater in the dependent lung areas than in the non-dependent lung areas. Compared with the initial measurements, ΔGV followed the significant changes of the EIT values but without significance in most of the pressure levels (Fig. 4).
Regional tidal volume change during the PEEP trial (ROI 1–4): box plots representing the regional tidal change of gas volumes ΔGVreg and regional tidal variation TVreg during the PEEP trial. Impedance data were not normalized. The box plots show the median, upper, and lower quartiles, whereas the whiskers indicate the minimum and maximum (n = 6). § (ΔGVreg), † (TVreg) p < 0.05 is considered a statistically significant increase between initial and subsequent PEEP levels
Distribution of regional lung recruitment and derecruitment
Figure 5 exemplarily shows the change of regional lung recruitment and derecruitment in one pig during the PEEP trial. The increase of tidal volume (EIT) in the incremental part of the PEEP trial started at between 10 and 15 cmH2O in all animals and was mainly found in the dependent part of the lung (TVGdependent 17.8 ± 13.3, TVGnon-dependent 5.6 ± 3.6; p < 0.05). Derecruitment of the lung started when reducing PEEP from 15 to 10 cmH2O also in the dependent parts (TVLdependent 15.9 ± 8.7, TVLnon-dependent 0.9 ± 0.5; p < 0.05), although the global tidal volume (TVG 35.6 ± 15.3) was still increasing. This coincided with the drop in oxygenation at this PEEP level (Fig. 3).
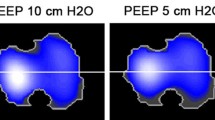
Visualization of regional changes of tidal volume: exemplary CT scans (left) and fixed-scaled functional EIT images (fEIT) (middle) from one of the six pigs. In the fEIT images, blue areas symbolize no ventilation, and red areas high ventilation. Normalization of the fEIT images with the corresponding VT allows the calculation of differential fEIT images (dfEIT) (right) by means of subtraction. dfEIT images enable a qualitative and quantitative assessment of regional change of VT when altering the PEEP level. Of particular interest were the dfEIT images that contained areas of both increase and decrease of VT (PEEP level 15 to 20 cmH2O and 15 to 10 cmH2O). During these steps the hidden individual starting points of regional recruitment and lung collapse can be seen
Discussion
The aim of our study was to assess global and regional lung recruitment and lung collapse with EIT as well as CT images during an incremental and decremental PEEP trial in experimental ALI.
Evaluation of EIT vs CT data
The present study demonstrates that EIT is able to describe global and regional changes in gas volumes and tidal volumes of an intrathoracic slice during a PEEP trial in surfactant-deficient lungs. In agreement with other studies [11, 14], the gas and tidal volumes derived from CT scans show a high linear correlation with corresponding values in the EIT (rel. ΔZ, TV). Because EIT measurements do not represent absolute values and depend on different baseline references, amplification factors, and individual anatomies [27], the linear regression lines between, for example, rel. ΔZ and GV have different slopes and axis intercepts for each animal, even though exactly the same variation of PEEP was used. Therefore, the correlation coefficient is lower when combining the data of all animals (r = 0.78) than the intra-individual correlation of each animal (from r = 0.98 to r = 0.99).
This individual correlation of EIT and CT monitoring with regard to the end-expiratory air content during the PEEP trial carried out here is nearly identical with the correlation calculated by Victorino et al. [14] (r = 0.96) for ARDS patients without concurrently recorded CT and EIT. Their measurements were nevertheless performed during a slow inflation maneuver over 80 s and not during continuous ventilation over a longer period. In our study, all the measurements were performed simultaneously during the variation of PEEP in PCV, a setting close to clinical reality with regard to PEEP titration.
A major new approach in the present study is the comparison of global and regional tidal volumes in EIT and simultaneous CT scans. This was achieved by electronically synchronizing the CT measurement with the end of inspiration and expiration. The correlations that we obtained are not as high as those for the intrathoracic air content of the transverse thoracic plane, probably because the tidal volume changes are small compared with the gas volume content itself, which reduces the signal-to-noise ratio mainly in the delta-CT scans. Noise is introduced, for instance, by the non-transparent electrodes used and by not removing all the pulmonary vessels and bronchi from the manually defined ROIs. This problem occurred especially in ROI 3. One other reason for the lower correlation between both imaging systems is that the transverse thoracic volume measured by EIT is larger than the detected volume with the CT scan [28]. Furthermore, a precise definition of the ROI examined is difficult, because tomograms derived by EIT show no anatomical landmarks. Nevertheless, our data showed only minor differences in trend analysis between tidal volume alteration derived from EIT and CT during the PEEP trial.
Detection of recruitment and lung collapse
The present study shows that analysis of EIT measurements, which generally have a low spatial resolution, permits assessment of the regional changes of VT during the PEEP maneuver, not only qualitatively, but to a certain degree also quantitatively. During the maneuver, an effect of gravity occurs regarding the onset and the degree of increase or decrease of the regional tidal volumes in the four ROIs. Because of the distribution of atelectasis in the lavage lung model, the calculation of the regional VT derived from EIT and CT show the highest increase in the dependent ROIs. This result is in accordance with the common understanding of inhomogeneous gravity-dependent tidal volume distributions in ALI and ARDS [26, 29]. Kunst et al. [30] and Hinz et al. [31] studied this phenomenon by using EIT and demonstrated that the regional lower inflection point increases in the ventral–dorsal direction. To quantitatively investigate these changes in the regional distribution of tidal volumes after a recruitment maneuver, Kunst et al. [16] calculated the “impedance ratio”, i. e. the ratio between TV of the dependent and non-dependent lung area. Although these authors only obtained relative values for the regional changes of tidal volumes, and not the absolute values we have, the results show the same tendency.
With conventional techniques, assessment of the beginning of the opening and collapsing of lung units is difficult during a PEEP trial at the bedside. In our study, we focused on the measurement of oxygenation, VT, and regional EIT analysis. The data concerning oxygenation and global VT obtained in the present study show opposing tendencies with respect to the ability to assess the beginning of recruitment and lung collapse during the PEEP trial. For instance, after reducing PEEP from 15 to 10 cmH2O, PaO2 decreases, whereas VT increases (Fig. 3). By contrast, during a rise in PEEP from 20 to 25 cmH2O, PaO2 continues to increase, whereas global VT decreases. This opposing tendency of PaO2 and VT is a consequence of a changing pulmonary shunt fraction, mainly in the dependent well-perfused lung areas, and of the overinflation of normally ventilated areas attributable to the PEEP variation [1].
The EIT technique offers the advantage of dynamically studying regional changes of lung mechanics [17]. dfEIT scans show that dependent and non-dependent lung areas differ in their responses during the PEEP trial (Fig. 5). For the assessment of lung recruitment and collapse, it seems to be reasonable to evaluate the PEEP-induced changes of the dependent lung areas in the fEIT images. These areas experience the most distinct dynamic changes, so that results are easily obtainable. Thus, in dependent lung units, recruitment can be identified at the first PEEP elevation (between 10 and 15 cmH2O). The beginning of regional lung collapse is detected during the decremental phase, when PEEP changes from 15 to 10 cmH2O, which corresponds well to the decreasing PaO2. These observations show that the EIT technique has a higher degree of sensitivity than the global ventilation parameters (i. e., Crs or VT) in detecting the onset of regional alveolar recruitment or lung collapse. However, these issues might differ in other lung models and have to be confirmed in clinical studies before transferring them into routine practice.
Conclusion
The change in global and regional tidal volume attributable to an incremental and decremental PEEP trial has been demonstrated by analysis of EIT. The initiation of regional recruitment and the start of lung collapse can be displayed more advantageously in regional EIT measurements than by extraction from global ventilation parameters. EIT can thus be used as a tool to adapt the PEEP level to the individual degree of lung injury in order to maintain recruitment.
References
Gattinoni L, Pesenti A, Bombino M, Baglioni S, Rivolta M, Rossi F, Rossi G, Fumagalli R, Marcolin R, Mascheroni D, Torressin A (1988) Relationships between lung computed tomographic density, gas exchange, and PEEP in acute respiratory failure. Anesthesiology 69:824–832
Sinclair SE, Albert RK (1997) Altering ventilation–perfusion relationships in ventilated patients with acute lung injury. Intensive Care Med 23:942–950
Nuckton TJ, Alonso JA, Kallet RH, Daniel BM, Pittet JF, Eisner MD, Matthay MA (2002) Pulmonary dead-space fraction as a risk factor for death in the acute respiratory distress syndrome. N Engl J Med 346:1281–1286
Tremblay LN, Slutsky AS (2006) Ventilator-induced lung injury: from the bench to the bedside. Intensive Care Med 32:24–33
Dreyfuss D, Saumon G (1998) Ventilator-induced lung injury: lessons from experimental studies. Am J Respir Crit Care Med 157:294–323
Hickling KG (1998) The pressure-volume curve is greatly modified by recruitment. A mathematical model of ARDS lungs. Am J Respir Crit Care Med 158:194–202
Stenqvist O (2003) Practical assessment of respiratory mechanics. Br J Anaesth 91:92–105
Gattinoni L, Caironi P, Pelosi P, Goodman LR (2001) What has computed tomography taught us about the acute respiratory distress syndrome? Am J Respir Crit Care Med 164:1701–1711
Puybasset L, Cluzel P, Gusman P, Grenier P, Preteux F, Rouby JJ (2000) Regional distribution of gas and tissue in acute respiratory distress syndrome. I. Consequences for lung morphology. CT Scan ARDS Study Group. Intensive Care Med 26:857–869
Malbouisson LM, Muller JC, Constantin JM, Lu Q, Puybasset L, Rouby JJ (2001) Computed tomography assessment of positive end-expiratory pressure-induced alveolar recruitment in patients with acute respiratory distress syndrome. Am J Respir Crit Care Med 163:1444–1450
Frerichs I, Hinz J, Herrmann P, Weisser G, Hahn G, Dudykevych T, Quintel M, Hellige G (2002) Detection of local lung air content by electrical impedance tomography compared with electron beam CT. J Appl Physiol 93:660–666
Frerichs I, Dargaville PA, Dudykevych T, Rimensberger PC (2003) Electrical impedance tomography: a method for monitoring regional lung aeration and tidal volume distribution? Intensive Care Med 29:2312–2316
Frerichs I, Schiffmann H, Hahn G, Hellige G (2001) Non-invasive radiation-free monitoring of regional lung ventilation in critically ill infants. Intensive Care Med 27:1385–1394
Victorino JA, Borges JB, Okamoto VN, Matos GF, Tucci MR, Caramez MP, Tanaka H, Sipmann FS, Santos DC, Barbas CS, Carvalho CR, Amato MB (2004) Imbalances in regional lung ventilation: a validation study on electrical impedance tomography. Am J Respir Crit Care Med 169:791–800
Frerichs I, Braun P, Dudykevych T, Hahn G, Genee D, Hellige G (2004) Distribution of ventilation in young and elderly adults determined by electrical impedance tomography. Respir Physiol Neurobiol 143:63–75
Kunst PW, Vazquez de Anda G, Bohm SH, Faes TJ, Lachmann B, Postmus PE, de Vries PM (2000) Monitoring of recruitment and derecruitment by electrical impedance tomography in a model of acute lung injury. Crit Care Med 28:3891–3895
Odenstedt H, Lindgren S, Olegard C, Erlandsson K, Lethvall S, Aneman A, Stenqvist O, Lundin S (2005) Slow moderate pressure recruitment maneuver minimizes negative circulatory and lung mechanic side effects: evaluation of recruitment maneuvers using electric impedance tomography. Intensive Care Med 31:1706–1714
Frerichs I, Dargaville PA, van Genderingen HR, Morel DR, Rimensberger PC (2006) Lung volume recruitment after surfactant administration modifies spatial distribution of ventilation. Am J Respir Crit Care Med 174:772–779
Meier T, Luepschen H, Karsten J, Leibecke T, Großherr M, Leonhardt S (2006) Comparison of regional lung recruitment in electrical impedance tomograms and CT scans in experimental acute lung injury. Critical Care [Suppl] 10(1):P7
Meier T, Luepschen H, Karsten J, Großherr M, Leibecke T, Gehring H, Leonhardt S (2006) Impact of different PEEP levels on regional compliance measured by electrical impedance tomography. Intensive Care Med [Suppl] 32(13):S221
Brown BH (2003) Electrical impedance tomography (EIT) a review. J Med Eng Technol 27:97–108
Brown BH, Barber DC (1987) Electrical impedance tomography; the construction and application to physiological measurement of electrical impedance images. Med Prog Technol 13:69–75
Barber DC (1989) A review of image reconstruction techniques for electrical impedance tomography. Med Phys 16:162–169
Lachmann B, Robertson B, Vogel J (1980) In vivo lung lavage as an experimental model of the respiratory distress syndrome. Acta Anaesthesiol Scand 24:231–236
Hahn G, Sipinkova I, Baisch F, Hellige G (1995) Changes in the thoracic impedance distribution under different ventilatory conditions. Physiol Meas [Suppl] 16:A161–A173
Gattinoni L, Pelosi P, Crotti S, Valenza F (1995) Effects of positive end-expiratory pressure on regional distribution of tidal volume and recruitment in adult respiratory distress syndrome. Am J Respir Crit Care Med 151:1807–1814
Wolf GK, Arnold JH (2005) Noninvasive assessment of lung volume: respiratory inductance plethysmography and electrical impedance tomography. Crit Care Med [Suppl] 33:S163–S169
Adler A, Amyot R, Guardo R, Bates JH, Berthiaume Y (1997) Monitoring changes in lung air and liquid volumes with electrical impedance tomography. J Appl Physiol 83:1762–1767
Luecke T, Meinhardt JP, Herrmann P, Weiss A, Quintel M, Pelosi P (2006) Oleic acid vs saline solution lung lavage-induced acute lung injury: effects on lung morphology, pressure–volume relationships, and response to positive end-expiratory pressure. Chest 130:392–401
Kunst PW, Bohm SH, Vazquez de Anda G, Amato MB, Lachmann B, Postmus PE, de Vries PM (2000) Regional pressure volume curves by electrical impedance tomography in a model of acute lung injury. Crit Care Med 28:178–183
Hinz J, Moerer O, Neumann P, Dudykevych T, Frerichs I, Hellige G, Quintel M (2006) Regional pulmonary pressure volume curves in mechanically ventilated patients with acute respiratory failure measured by electrical impedance tomography. Acta Anaesthesiol Scand 50:331–339
Acknowledgements
The support of Eckhard Teschner, Dräger Medical AG, is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Meier, T., Luepschen, H., Karsten, J. et al. Assessment of regional lung recruitment and derecruitment during a PEEP trial based on electrical impedance tomography. Intensive Care Med 34, 543–550 (2008). https://doi.org/10.1007/s00134-007-0786-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00134-007-0786-9