Abstract
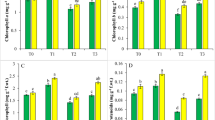
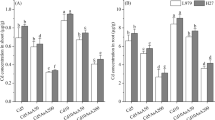
Wheat (Triticum aestivum L.) has relatively high tolerance to cadmium (Cd), but the underlying mechanisms are poorly understood. Growth and physiological parameters of wheat exposed to different Cd concentrations (0, 0.5, 5 and 50 µM) were characterized. The fresh weight, leaf chlorophyll and carotenoid concentrations and photosynthesis parameters did not differ among Cd treatments, suggesting relatively high Cd tolerance in wheat. However, the soluble sugar concentrations increased with the increasing Cd concentration and the soluble protein concentrations decreased in both shoots and roots, suggesting that the Cd application promoted nitrogen metabolism over carbon metabolism. In addition, the higher concentrations of MDA, GSH and AsA and activities of antioxidant enzymes (SOD, POD, and CAT) were observed in leaves and roots in the Cd50 treatment. Our results reveal that wheat can tolerate Cd by enhancing the antioxidant enzymes activities and increasing the concentration of ascorbate and glutathione.




Similar content being viewed by others
References
Aebi H (1984) Catalase in vitro. Methods Enzymol 105:121–126. https://doi.org/10.1016/S0076-6879(84)05016-3
Anderson ME (1985) Determination of glutathione and glutathione disulfide in biological samples. Methods Enzymol 113(4):548–555. https://doi.org/10.1016/S0076-6879(85)13073-9
Beauchamp C, Fridovich I (1971) Superoxide dismutase: improved assays and an assay applicable to acrylamide gels. Anal Biochem 44(1):276–287. https://doi.org/10.1016/0003-2697(71)90370-8
Burzyński M, Kłobus G (2004) Changes of photosynthetic parameters in cucumber leaves under Cu, Cd, and Pb stress. Photosynthetica 42(4):505–510. https://doi.org/10.1007/S11099-005-0005-2
Ci D, Jiang D, Dai T, Jing Q, Cao W (2009) Effects of cadmium on plant growth and physiological traits in contrast wheat recombinant inbred lines differing in cadmium tolerance. Chemosphere 77(11):1620–1625. https://doi.org/10.1016/j.chemosphere.2009.08.062
Costa G, Spitz E (1997) Influence of cadmium on soluble carbohydrates, free amino acids, protein content of in vitro cultured Lupinus albus. Plant Sci 128(2):131–140. https://doi.org/10.1016/S0168-9452(97)00148-9
Dalcorso G, Manara A, Furini A (2013) An overview of heavy metal challenge in plants: from roots to shoots. Metallomics 5(9):1117–1132. https://doi.org/10.1039/c3mt00038a
Fridovich I (1989) Superoxide dismutases. An adaptation to a paramagnetic gas. J Biol Chem 264(14):7761–7764
Gallego SM, Pena LB, Barcia RA, Azpilicueta CE, Iannone MF, Rosales EP, Zawoznik MS, Groppa MD, Benavides MP (2012) Unravelling cadmium toxicity and tolerance in plants: insight into regulatory mechanisms. Environ Exp Bot 83:33–46. https://doi.org/10.1016/j.envexpbot.2012.04.006
Gutiérrez RA, Lejay LV, Dean A, Chiaromonte F, Shasha DE, Coruzzi GM (2007) Qualitative network models and genome-wide expression data define carbon/nitrogen-responsive molecular machines in Arabidopsis. Genome Biol 8(1):R7. https://doi.org/10.1186/gb-2007-8-1-r7
Jin CX, Zhou QX, Fan J (2010) Effects of chlorimuron-ethyl and cadimum on biomass growth and cadimum accumulation of wheat in the phaiozem area, Northeast China. Bull Environ Contam Toxicol 84:395–400. https://doi.org/10.1007/s00128-009-9635-6
Lin R, Wang X, Luo Y, Du W, Guo H, Yin D (2007) Effects of soil cadmium on growth, oxidative stress and antioxidant system in wheat seedlings (Triticum aestivum L.). Chemosphere 69(1):89–98. https://doi.org/10.1016/j.chemosphere.2007.04.041
Morita S, Tasaka M, Fujisawa H, Ushimaru T, Tsuji H (1994) A cDNA clone encoding a rice catalase isozyme. Plant Physiol 105(3):1015–1016. https://doi.org/10.1104/pp.105.3.1015
Naeem A, Saifullah, Rehman MZ, Akhtar T, Ok YS, Rengel Z (2016) Genetic variation in cadmium accumulation and tolerance among wheat cultivars at the seedling stage. Commun Soil Sci Plant Anal 47(5):554–562. https://doi.org/10.1080/00103624.2016.1141918
Qin S, Sun X, Hu C, Tan Q, Zhao X, Xin J, Wen X (2017a) Effect of NO3 −:NH4 + ratios on growth, root morphology and leaf metabolism of oilseed rape (Brassica napus L.) seedlings. Acta Physiol Plant 39(9):198. https://doi.org/10.1007/s11738-017-2491-9
Qin S, Sun X, Hu C, Tan Q, Zhao X, Xu S (2017b) Effects of tungsten on uptake, transport and subcellular distribution of molybdenum in oilseed rape at two different molybdenum levels. Plant Sci 256:87–93. https://doi.org/10.1016/j.plantsci.2016.12.009
Rizwan M, Ali S, Abbas T, Zia-ur-Rehman M, Hannan F, Keller C, Al-Wabel MI, Ok YS (2016a) Cadmium minimization in wheat: a critical review. Ecotoxicol Environ Saf 130:43–53. https://doi.org/10.1016/j.ecoenv.2016.04.001
Rizwan M, Meunier JD, Davidian JC, Pokrovsky OS, Bovet N, Keller C (2016b) Silicon alleviates Cd stress of wheat seedlings (Triticum turgidum L. cv. Claudio) grown in hydroponics. Environ Sci Pollut Res 23(2):1414–1427. https://doi.org/10.1007/s11356-015-5351-4
Shah K, Kumar RG, Verma S, Dubey RS (2001) Effect of cadmium on lipid peroxidation, superoxide anion generation and activities of antioxidant enzymes in growing rice seedlings. Plant Sci 161(6):1135–1144. https://doi.org/10.1016/S0168-9452(01)00517-9
Singh S, Parihar P, Singh R, Singh VP, Prasad SM (2016) Heavy metal tolerance in plants: role of transcriptomics, proteomics, metabolomics, and ionomics. Front Plant Sci 6:1143. https://doi.org/10.3389/fpls.2015.01143
Sun RL, Zhou QX, Sun FH, Jin CX (2007) Antioxidative defense and proline/phytochelatin accumulation in a newly discovered Cd-hyperaccumulator, Solanum nigrum L. Environ Exp Bot 60(3):468–476. https://doi.org/10.1016/j.envexpbot.2007.01.004
Thounaojam TC, Panda P, Mazumdar P, Kumar D, Sharma GD, Sahoo L, Sanjib P (2012) Excess copper induced oxidative stress and response of antioxidants in rice. Plant Physiol Biochem 53(4):33–39. https://doi.org/10.1016/j.plaphy.2012.01.006
Wang F, Chen F, Cai Y, Zhang G, Wu F (2011) Modulation of exogenous glutathione in ultrastructure and photosynthetic performance against Cd stress in the two barley genotypes differing in Cd tolerance. Biol Trace Elem Res 144:1275–1288. https://doi.org/10.1007/s12011-011-9121-y
Willekens H, Chamnongpol S, Davey M, Schraudner M, Langebartels C, Van MM, Inzé D, Van WC (1997) Catalase is a sink for H2O2 and is indispensable for stress defence in C3 plants. Embo J 16(16):4806–4816. https://doi.org/10.1093/emboj/16.16.4806
Wu Z, Zhao X, Sun X, Tan Q, Tang Y, Nie Z, Qu C, Chen Z, Hu C (2015) Antioxidant enzyme systems and the ascorbate-glutathione cycle as contributing factors to cadmium accumulation and tolerance in two oilseed rape cultivars (Brassica napus L.) under moderate cadmium stress. Chemosphere 138:526–536. https://doi.org/10.1016/j.chemosphere.2015.06.080
Xin J, Zhao X, Tan Q, Sun X, Wen X, Qin S, Hu C (2016) The effects of cadmium exposure on cadmium fractionation and enzyme activities in the rhizosphere of two radish cultivars (Raphanus sativus L.). Bull Environ Contam Toxicol 98(2):290–295. https://doi.org/10.1007/s00128-016-1998-x
Zhang M, Hu C, Zhao X, Tan Q, Sun X, Cao A, Cui M, Zhang Y (2012) Molybdenum improves antioxidant and osmotic-adjustment ability against salt stress in Chinese cabbage (Brassica campestris L. ssp. Pekinensis). Plant Soil 355:375–383. https://doi.org/10.1007/s11104-011-1109-z
Acknowledgements
We thank Prof. Zed Rengel (University of Western Australia, Australia) for critical reviewing and revision of the manuscript. This work was financially supported by the Doctoral Scientific Research Foundation of Henan Agricultural University (Grant No. 30500592), National Key R&D Program of China (Grant No. 2017YFD0201700), and Henan Province Science and Technology Research Project (Grant No. 172102110060).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Qin, S., Liu, H., Nie, Z. et al. AsA–GSH Cycle and Antioxidant Enzymes Play Important Roles in Cd Tolerance of Wheat. Bull Environ Contam Toxicol 101, 684–690 (2018). https://doi.org/10.1007/s00128-018-2471-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00128-018-2471-9