Abstract
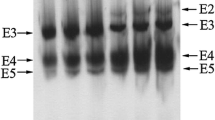
This study characterized esterase activity in Cerastoderma edule tissues using different substrates and specific inhibitors and identified the tissue distribution of esterases in this species. Synthetic thiocholines and thioacetate esters and specific inhibitors (eserine, BW284C51 and iso-OMPA) were used to identify and quantify cholines and carboxyl esterases. The results demonstrated the presence of a non-specific propionyl thiocholine (PrSCh)-cleaving cholinesterase (ChE) and a large amount of carboxylesterases (CaE). For further studies using C. edule esterases as biomarkers, our results suggest that the adductor muscle, with PrSCh (5 mM) as substrate should be used to analyze ChE, and for CaE analyses, phenyl thioacetate should be used in digestive gland extracts (PSA, 5 mM).



Similar content being viewed by others
References
Baudrimont M, de Montaudouin X, Palvadeau A (2003) Bivalve vulnerability is enhanced by parasites through the deficit of metallothionein synthesis: a field monitoring on cockles (Cerastoderma edule). J Phys IV France 107:131–134
Bolton-Warberg M, Coen L, Weinstein J (2007) Acute toxicity and acetylcholinesterase inhibition in grass shrimp (Palaemonetes pugio) and oysters (Crassostrea virginica) exposed to the organophosphate dichlorvos: laboratory and field studies. Arch Environ Contam Toxicol 52:207–216
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Cheung VV, Depledge MH, Jha AN (2006) An evaluation of the relative sensitivity of two marine bivalve mollusc species using the Comet assay. Mar Environ Res 62:S301–S305
Corsi I, Pastore AM, Lodde A, Palmerini E, Castagnolo L, Focardi S (2007) Potential role of cholinesterases in the invasive capacity of the freshwater bivalve, Anodonta woodiana (Bivalvia: Unionacea): A comparative study with the indigenous species of the genus, Anodonta sp. Comp Biochem Physiol C Toxicol Pharmacol 145:413–419
Diaz S, Cao A, Villalba A, Carballal MJ (2010) Expression of mutant protein p53 and Hsp70 and Hsp90 chaperones in cockles Cerastoderma edule affected by neoplasia. Dis Aquat Organ 90:215–222
Ellman GL, Courtney KD, Andres V Jr, Featherstone RM (1961) A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem Pharmacol 7:88–90
Escartín E, Porte C (1997) The use of cholinesterase and carboxylesterase activities from Mytilus galloprovincialis in pollution monitoring. Environ Toxicol Chem 16:2090–2095
Galloway TS, Millward N, Browne MA, Depledge MH (2002) Rapid assessment of organophosphorous/carbamate exposure in the bivalve mollusc Mytilus edulis using combined esterase activities as biomarkers. Aquat Toxicol 61:169–180
Guilhermino L, Lopes MC, Carvalho AP, Soares AMVM (1996) Acetylcholinesterase activity in Juveniles of Daphnia magna Straus. Bull Environ Contam Toxicol 57:979–985
Kozlovskaya VI, Mayer FL, Menzikova OV, Chuylo GM (1993) Cholinesterases of aquatic animals. In: Ware GW (ed) Reviews of environmental contamination and toxicology. Springer, New York, p 117
Lagerspetz KYH, Impivaara H, Senius K (1970) Acetylcholine in the thermal resistance acclimation of the ciliary activity in the gills of anodonta. Comp Gen Pharmacol 1:236–240
Laguerre C, Sanchez-Hernandez JC, Köhler HR, Triebskorn R, Capowiez Y, Rault M, Mazzia C (2009) B-type esterases in the snail Xeropicta derbentina: an enzymological analysis to evaluate their use as biomarkers of pesticide exposure. Environ Pollut 157:199–207
Machreki-Ajmi M, Hamza-Chaffai A (2008) Assessment of sediment/water contamination by in vivo transplantation of the cockles Cerastoderma glaucum from a non contaminated to a contaminated area by cadmium. Ecotoxicology 17:802–810
Meng W, Liu L (2010) On approaches of estuarine ecosystems health studies. Estuar Coast Shelf Sci 86:313–316
Monserrat JM, Bianchini A, Bainy ACD (2002) Kinetic and toxicological characteristics of acetylcholinesterase from the gills of oysters (Crassostrea rhizophorae) and other aquatic species. Mar Environ Res 54:781–785
Monteiro M, Quintaneiro C, Morgado F, Soares AMVM, Guilhermino L (2005) Characterization of the cholinesterases present in head tissues of the estuarine fish Pomatoschistus microps: application to biomonitoring. Ecotoxicol Environ Saf 62:341–347
Mora P, Michel X, Narbonne J-F (1999) Cholinesterase activity as potential biomarker in two bivalves. Environ Toxicol Phar 7:253–260
Moreira SM, Coimbra J, Guilhermino L (2001) Acetylcholinesterase of Mytilus galloprovincialis LmK. hemolymph: a suitable environmental biomarker. Bull Environ Contam Toxicol 67:470–475
Paul-Pont I, Gonzalez P, Baudrimont M, Nili H, de Montaudouin X (2010) Short-term metallothionein inductions in the edible cockle Cerastoderma edule after cadmium or mercury exposure: discrepancy between mRNA and protein responses. Aquat Toxicol 97:260–267
Solé M, Shaw J, Frickers P, Readman J, Hutchinson T (2010) Effects on feeding rate and biomarker responses of marine mussels experimentally exposed to propranolol and acetaminophen. Anal Bioanal Chem 396:649–656
Stefano B, Ilaria C, Silvano F (2008) Cholinesterase activities in the scallop Pecten jacobaeus: characterization and effects of exposure to aquatic contaminants. Sci Total Environ 392:99–109
Valbonesi P, Sartor G, Fabbri E (2003) Characterization of cholinesterase activity in three bivalves inhabiting the North Adriatic sea and their possible use as sentinel organisms for biosurveillance programmes. Sci Total Environ 312:79–88
Wheelock CE, Phillips BM, Anderson BS, Miller JL, Miller MJ, Hammock BD (2008) Applications of carboxylesterase activity in environmental monitoring and toxicity identification evaluations (TIEs). In: Whitacre DM (ed) Reviews of environmental contamination and toxicology, vol 195. Springer, New York, p 117
Acknowledgments
This study was supported by Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES/Brazil) in international cooperation with Fundação de Ciência e Tecnologia (FCT/Portugal) (process 240/09) and the post-doctoral fellowship of M.S. Monteiro (SFRH/BPD/45911/2008). This publication has been partially financially supported by the Atlantic Area Operational Programme, through the European Regional Development Fund (ERDF), Project PORTONOVO (ref 2009-1/119). Technicians of Biology Department of University of Aveiro, Aldiro Pereira and Rui Marques assisted with organism collection. The English was edited by American Journal Experts (1875-CE76-75F9-2EAB-B015).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nilin, J., Monteiro, M., Domingues, I. et al. Bivalve Esterases as Biomarker: Identification and Characterization in European Cockles (Cerastoderma edule). Bull Environ Contam Toxicol 88, 707–711 (2012). https://doi.org/10.1007/s00128-012-0521-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00128-012-0521-2