Abstract
Aims/hypothesis
The aim of this study was to test the hypothesis that intensive glycaemic control (INT) and higher plasma C-peptide levels in patients with poorly controlled diabetes would be associated with better eye outcomes.
Methods
The incidence and progression of diabetic retinopathy (DR) was assessed by grading seven-field stereoscopic fundus photographs at baseline and 5 years later in 858 of 1,791 participants in the Veterans Affairs Diabetes Trial (VADT).
Results
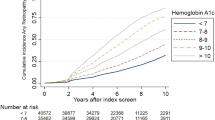
After adjustment for all covariates, risk of progression (but not incidence) of DR increased by 30% for each 1% increase in baseline HbA1c (OR 1.3; 95% CI 1.123, 1.503; p = 0.0004). Neither assignment to INT nor age was independently associated with DR in the entire cohort. However, INT showed a biphasic interaction with age. The incidence of DR was decreased in INT participants ≤55 years of age (OR 0.49; 95% CI 0.24, 1.0) but increased in those ≥70 years old (OR 2.88; 95% CI 1.0, 8.24) (p = 0.0043). The incidence of DR was reduced by 67.2% with each 1 pmol/ml increment in baseline C-peptide (OR 0.328; 95% CI 0.155, 0.7; p = 0.0037). Baseline C-peptide was also an independent inverse risk factor for the progression of DR, with a reduction of 47% with each 1 pmol/ml increase in C-peptide (OR 0.53; 95% CI 0.305, 0.921; p = 0.0244).
Conclusions/interpretation
Poor glucose control at baseline was associated with an increased risk of progression of DR. INT was associated with a decreased incidence of DR in younger patients but with an increased risk of DR in older patients. Higher C-peptide at baseline was associated with reduced incidence and progression of DR.

Similar content being viewed by others
Abbreviations
- ACCORD:
-
Action to Control Cardiovascular Risk in Diabetes
- ADVANCE:
-
Action in Diabetes and Vascular Disease: Preterax and Diamicron Modified Release Controlled Evaluation
- DR:
-
Diabetic retinopathy
- eGFR:
-
Estimated GFR
- ETDRS:
-
Early Treatment Diabetic Retinopathy Study
- HDL-C:
-
HDL cholesterol
- INT:
-
Intensive glycaemic control
- LDL-C:
-
LDL cholesterol
- PAI-1:
-
Plasminogen activator inhibitor-1
- STD:
-
Standard glycaemic therapy
- TC:
-
Total cholesterol
- UKPDS:
-
UK Prospective Diabetic Study
- VADT:
-
Veterans Affairs Diabetes Trial
References
U.S. Department of Health and Human Services, Centers for Disease Control and Prevention (2011) National estimates and general information on diabetes and pre-diabetes in the U.S. U.S. Department of Health and Human Services, Centers for Disease Control and Prevention, Atlanta, GA
Kostev K, Rathmann W (2013) Diabetic retinopathy at diagnosis of type 2 diabetes in the UK: a database analysis. Diabetologia 56:109–111
Ko F, Vitale S, Chou CF, Cotch MF, Saaddine J, Friedman DS (2012) Prevalence of nonrefractive visual impairment in US adults and associated risk factors, 1999–2002 and 2005–2008. JAMA 308:2361–2368
Fong DS, Aiello L, Gardner TW et al (2003) Diabetic retinopathy. Diabetes Care 26:226–229
Herman WH (2013) The economic costs of diabetes: is it time for a new treatment paradigm? Diabetes Care 36:775–776
The Diabetes Control and Complications Trial Research Group (1993) The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. N Engl J Med 329:977–986
UK Prospective Diabetes Study (UKPDS) Group (1998) Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS33). Lancet 352:837–853
Duckworth W, Abraira C, Moritz T et al (2009) Glucose control and vascular complications in veterans with type 2 diabetes. N Engl J Med 360:129–130
Abraira C, Duckworth W, McCarren M et al (2003) Design of the cooperative study on glycemic control and complication in diabetes mellitus type 2: Veterans Affairs Diabetes Trial (VADT). J Diabetes Complicat 7:314–322
Faber OK, Binder C, Markussen J et al (1978) Characterization of seven C-peptide antisera. Diabetes 27(Suppl l):170–177
Emanuele N, Moritz T, Klein R, The VADT Study Group (2009) Ethnicity, race, and clinically significant macular edema in the Veterans Affairs Diabetes Trail (VADT). Diabetes Res Clin Pract 86:104–110
Emanuele N, Klein R, Mortiz M et al (2009) Comparison of dilated fundus examination by ophthalmologists, with 7-field stereo photographs in the VADT. J Diabetes Complicat 23:323–329
Chew EY, Bethesda MD, Ambrosius WT et al (2010) The ACCORD Study Group and ACCORD Eye Study Group. Effects of medical therapies on retinopathy progression in type 2 diabetes. N Engl J Med 363:233–244
Patel A, MacMahon SD, Chalmers J et al (2008) The Action in Diabetes and Vascular Disease: Preterax and Diamicron Modified Release Controlled Evaluation (ADVANCE) Collaborative Group. Intensive blood glucose control and vascular outcomes in patients with type 2 diabetes. N Engl J Med 358:2560–2572
Beulens JWJ, Patel A, Vingerling JR et al (2009) Effects of blood pressure lowering and intensive glucose control on the incidence and progression of retinopathy in patients with type 2 diabetes mellitus: a randomized controlled trial. Diabetologia 52:2027–2036
The Diabetes Control and Complications Trial Research Group (1995) The relationship of glycemic exposure (HbA1c) to the risk of development and progression of retinopathy in the Diabetes Control and Complication Trial. Diabetes 44:968–983
Shichiri M, Kishikawa H, Ohkubo Y, Wake N, Mogensen CE, Pass P (2000) Long term results of the Kumamoto Study on optimal diabetes control in type 2 diabetic patients. Diabetes Care 23:B21–B29
Stratton IM, Kohner EM, Aldington SJ et al (2001) UKPDS 50: risk factors for incidence and progression of retinopathy in type II diabetes over 6 years from diagnosis. Diabetologia 44:156–163
Ohkubo Y, Kishikawa H, Araki E et al (1995) Intensive insulin therapy prevents the progression of diabetic microvascular complications in Japanese patients with non-insulin-dependent diabetes mellitus: a randomized prospective 6-year study. Diabetes Res Clin Pract 28:103–117
Kohner EM, Aldington SJ, Stratton IM et al (1998) United Kingdom Prospective Diabetes Study, 30: diabetic retinopathy at diagnosis of non-insulin-dependent diabetes mellitus and associated risk factors. Arch Ophthalmol 116:297–303
Porta M, Sjoelie AK, Chaturvedi N et al (2001) Risk factors for progression to proliferative diabetic retinopathy in the EURODIAB Prospective Complications Study. Diabetologia 44:2203–2209
Klein R, Klein BE, Moss SE, Davis MD, DeMets DL (1984) The Wisconsin Epidemiologic Study of diabetic retinopathy. III. Prevalence and risk of diabetic retinopathy when age at diagnosis is 30 or more years. Arch Ophthalmol 102:527–532
Suzuki K, Watanabe K, Motegi T, Kajinuma H (1989) High prevalence of proliferative retinopathy in diabetic patients with low pancreatic B cell capacity. Diabetes Res Clin Pract 6:45–53
Steffes MW, Sibley S, Jackson M, Thomas W (2003) Beta-cell function and the development of diabetes-related complication in the Diabetes Control and Complications Trial. Diabetes Care 26:832–836
Kim BY, Jung CH, Mok JO, Kang SK, Kim CH (2012) Association between serum C-peptide levels and chronic microvascular complications in Korean type 2 diabetic patients. Acta Diabetol 49:9–15
Inukai T, Matsutomo R, Tayama K, Aso Y, Takemora Y (1999) Relation between the serum level of C-peptide and risk factors for coronary heart disease and diabetic microangiopathic patients with type 2 diabetes mellitus. Exp Clin Endocrinol Diabetes 107:40–45
Cifarelli V, Geng X, Styche A, Lakomy R, Trucco M, Luppi P (2011) C-peptide reduces high-glucose-induced apoptosis of endothelial cells and decreases NAD(P)H-oxidase reactive oxygen species generation in human aortic endothelial cells. Diabetologia 54:2702–2712
Funding
The study was sponsored by the Cooperative Studies Program of the Department of Veterans Affairs Office of Research and Development. There was also generous support from the National Eye Institute. Medications and financial support were provided by Sanofi-Aventis, GlaxoSmithKline, Novo Nordisk, Roche, Kos Pharmaceuticals, Merck and Amylin. These companies had no role in the design of the study, in the accrual or analysis of the data, or in the preparation or approval of the manuscript.
Duality of interest
The authors declare that there is no duality of interest associated with this manuscript.
Contribution statement
All authors vouch for the accuracy and completeness of the data and the analysis. NA is the guarantor of this work and, as such, had full access to the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. NA, LA, NVE, PR, RK and GDB contributed to the conception and design of the data analysis for this paper. They all were involved in drafting and revising the manuscript critically for important intellectual content, and all gave final approval of the version to be published. In addition, GDB performed the statistical analysis and provided the figures and tables.
Author information
Authors and Affiliations
Consortia
Corresponding author
Rights and permissions
About this article
Cite this article
Azad, N., Agrawal, L., Emanuele, N.V. et al. Association of blood glucose control and pancreatic reserve with diabetic retinopathy in the Veterans Affairs Diabetes Trial (VADT). Diabetologia 57, 1124–1131 (2014). https://doi.org/10.1007/s00125-014-3199-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00125-014-3199-7