Abstract
Key message
This study quantified genetic variation in root system architecture (root number, angle, length and dry mass) within a diversity panel of 1771 Ethiopian sorghum landraces and identified 22 genomic regions associated with the root variations.
Abstract
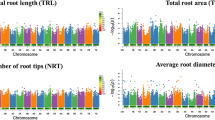
The root system architecture (RSA) of crop plants influences adaptation to water-limited conditions and determines the capacity of a plant to access soil water and nutrients. Four key root traits (number, angle, length and dry mass) were evaluated in a diversity panel of 1771 Ethiopian sorghum landraces using purpose-built root chambers. Significant genetic variation was observed in all studied root traits, with nodal root angle ranging from 16.4° to 26.6°, with a high repeatability of 78.9%. Genome wide association studies identified a total of 22 genomic regions associated with root traits which were distributed on all chromosomes except chromosome SBI-10. Among the 22 root genomic regions, 15 co-located with RSA trait QTL previously identified in sorghum, with the remaining seven representing novel RSA QTL. The majority (85.7%) of identified root angle QTL also co-localized with QTL previously identified for stay-green in sorghum. This suggests that the stay-green phenotype might be associated with root architecture that enhances water extraction during water stress conditions. The results open avenues for manipulating root phenotypes to improve productivity in abiotic stress environments via marker-assisted selection.





Similar content being viewed by others
Availability of data and material
All data are publicly accessible. Genotypic list with geographical information, SNP genotype and phenotypic data are available from Digital Repository and provided in Online Resource.
Code availability
Not applicable.
References
Ali M, Abbas A, Niaz S, Zulkiffal M, Ali S (2009) Morpho-physiological criteria for drought tolerance in sorghum (Sorghum bicolor) at seedling and post-anthesis stages. Int J Agric Biol 11(6):674–680
Ali ML, Luetchens J, Singh A, Shaver TM, Kruger GR, Lorenz AJ (2016) Greenhouse screening of maize genotypes for deep root mass and related root traits and their association with grain yield under water-deficit conditions in the field. Euphytica 207(1):79–94. https://doi.org/10.1007/s10681-015-1533-x
Atkinson JA, Rasmussen A, Traini R, Voß U, Sturrock C, Mooney SJ, Wells DM, Bennett MJ (2014) Branching out in roots: uncovering form, function, and regulation. Plant Physiol 166:538–550. https://doi.org/10.1104/pp.114.245423
Bekele WA, Fiedler K, Shiringani A, Schnaubelt D, Windpassinger S, Uptmoor R et al (2014) Unravelling the genetic complexity of sorghum seedling development under low-temperature conditions. Plant Cell Environ 37(3):707–723. https://doi.org/10.1111/pce.12189
Bernardino KC, Pastina MM, Menezes CB, de Sousa SM, Maciel LS, Carvalho G Jr et al (2019) The genetic architecture of phosphorus efficiency in sorghum involves pleiotropic QTL for root morphology and grain yield under low phosphorus availability in the soil. BMC Plant Biol 19(1):87. https://doi.org/10.1186/s12870-019-1689-y
Blum A, Arkin G, Jordan W (1977) Sorghum root morphogenesis and growth. I. Effect of maturity genes. Crop Sci 17(1):149–153. https://doi.org/10.2135/cropsci1977.0011183X001700010039x
Borrell AK, Hammer GL, Douglas AC (2000a) Does maintaining green leaf area in sorghum improve yield under drought? I. Leaf growth and senescence. Crop Sci 40(4):1026–1037. https://doi.org/10.2135/cropsci2000.4041026x
Borrell AK, Hammer GL, Henzell RG (2000b) Does maintaining green leaf area in sorghum improve yield under drought? II. Dry matter production and yield. Crop Sci 40(4):1037–1048. https://doi.org/10.2135/cropsci2000.4041037x
Borrell AK, Mullet JE, George-Jaeggli B, van Oosterom EJ, Hammer GL, Klein PE, Jordan DR (2014b) Drought adaptation of stay-green in sorghum associated with canopy development, leaf anatomy, root growth and water uptake. J Exp Bot 65(21):6251–6263. https://doi.org/10.1093/jxb/eru232
Borrell A, Oosterom EV, George-Jaeggli B, Rodriguez D, Eyre J, Jordan D et al (2021) Sorghum. In: Sadras VO, Calderini DF (eds) Chapter 5 in Crop physiology case histories for major crops. Academic Press, Elsevier, pp 197–214
Borrell A, Van Oosterom E, George-Jaeggli B, Vadez V, Singh V, Hammer G (2020) Physiology of growth, development and yield. Sorghum in the 21st century: food–fodder–feed–fuel for a rapidly changing world. Springer, pp 127–155. https://doi.org/10.1007/978-981-15-8249-3_6
Borrell AK, Oosterom EJ, Mullet JE, George-Jaeggli B, Jordan DR, Klein PE et al (2014a) Stay-green alleles individually enhance grain yield in sorghum under drought by modifying canopy development and water uptake patterns. New Phytol 203(3):817–830. https://doi.org/10.1111/nph.12869
Borrell AK, Wong A, George-Jaeggli B, van Oosterom EJ, Mace ES, Godwin ID et al (2022) Genetic modification of PIN genes induces causal mechanisms of stay-green drought adaptation phenotype. J Exp Bot. https://doi.org/10.1093/jxb/erac336
Browning BL, Zhou Y, Browning SR (2018) A one-penny imputed genome from next-generation reference panels. Am J Hum Genet 103(3):338–348. https://doi.org/10.1016/j.ajhg.2018.07.015
Chopra R, Burow G, Burke JJ, Gladman N, Xin Z (2017) Genome-wide association analysis of seedling traits in diverse Sorghum germplasm under thermal stress. BMC Plant Biol 17(1):12. https://doi.org/10.1186/s12870-016-0966-2
Christopher M, Chenu K, Jennings R, Fletcher S, Butler D, Borrell A et al (2018) QTL for stay-green traits in wheat in well-watered and water-limited environments. Field Crop Res 217:32–44. https://doi.org/10.1016/j.fcr.2017.11.003
Christopher JT, Manschadi AM, Hammer GL, Borrell AK (2008) Developmental and physiological traits associated with high yield and stay-green phenotype in wheat. Aust J Agric Res 59:354–364
Cullis BR, Smith AB, Coombes NE (2006) On the design of early generation variety trials with correlated data. J Agric Biol Environ Stat 11(4):381. https://doi.org/10.1198/108571106X154443
EIAR. (2014) Ethiopian strategy for sorghum.
Fakrudin B, Kavil S, Girma Y, Arun S, Dadakhalandar D, Gurusiddesh B et al (2013) Molecular mapping of genomic regions harbouring QTLs for root and yield traits in sorghum (Sorghum bicolor L. Moench). Physiol Mol Biol Plants 19(3):409–419. https://doi.org/10.1007/s12298-013-0188-0
Gilmour A, Cullis B, Harding S, Thompson R (2006) ASReml update: what’s new in Release 2.00. Hemel Hempstead, UK, VSN International Ltd
Haussmann B, Mahalakshmi V, Reddy B, Seetharama N, Hash C, Geiger H (2002) QTL mapping of stay-green in two sorghum recombinant inbred populations. Theor Appl Genet 106(1):133–142. https://doi.org/10.1007/s00122-002-1012-3
Huang M, Liu X, Zhou Y, Summers RM, Zhang Z (2018) BLINK: a package for the next level of genome-wide association studies with both individuals and markers in the millions. GigaScience 8(2):giy154. https://doi.org/10.1093/gigascience/giy154
Huang X, Zhao Y, Li C, Wang A, Zhao Q, Li W et al (2012) Genome-wide association study of flowering time and grain yield traits in a worldwide collection of rice germplasm. Nat Genet 44(1):32–39
Hufnagel B, de Sousa SM, Assis L, Guimaraes CT, Leiser W, Azevedo GC et al (2014) Duplicate and conquer: multiple homologs of PHOSPHORUS-STARVATION TOLERANCE1 enhance phosphorus acquisition and sorghum performance on low-phosphorus soils. Plant Physiol 166(2):659–677. https://doi.org/10.1104/pp.114.243949
Hulluka M, Esele J (1992) Sorghum diseases in eastern Africa. Sorghum Millet Dis Second World Rev. 502(324):21
Jordan D, Hunt C, Cruickshank A, Borrell A, Henzell R (2012) The relationship between the stay-green trait and grain yield in elite sorghum hybrids grown in a range of environments. Crop Sci 52(3):1153–1161. https://doi.org/10.2135/cropsci2011.06.0326
Joshi DC, Singh V, Hunt C, Mace E, van Oosterom E, Sulman R et al (2017) Development of a phenotyping platform for high throughput screening of nodal root angle in sorghum. Plant Methods 13(1):56. https://doi.org/10.1186/s13007-017-0206-2
Kebede H, Subudhi P, Rosenow D, Nguyen H (2001) Quantitative trait loci influencing drought tolerance in grain sorghum (Sorghum bicolor L. Moench). Theor Appl Genet 103(2–3):266–276. https://doi.org/10.1007/s001220100541
Kramer PJ, Boyer JS (1995) Water relations of plants and soils. Academic press
Lafarge T, Broad I, Hammer G (2002) Tillering in grain sorghum over a wide range of population densities: identification of a common hierarchy for tiller emergence, leaf area development and fertility. Ann Bot 90(1):87–98. https://doi.org/10.1093/aob/mcf152
Li R, Han Y, Lv P, Du R, Liu G (2014) Molecular mapping of the brace root traits in sorghum (Sorghum bicolor L. Moench). Breed Sci 64(2):193–198. https://doi.org/10.1270/jsbbs.64.193
Li M-X, Yeung JM, Cherny SS, Sham PC (2012) Evaluating the effective numbers of independent tests and significant p-value thresholds in commercial genotyping arrays and public imputation reference datasets. Hum Genet 131(5):747–756. https://doi.org/10.1007/s00439-011-1118-2
Liedtke JD, Hunt CH, George-Jaeggli B, Laws K, Watson J, Potgieter AB, Cruickshank A, Jordan DR (2020) High-throughput phenotyping of dynamic canopy traits associated with stay-green in grain sorghum. Plant Phenomics. https://doi.org/10.34133/2020/4635153
Lobell DB, Roberts MJ, Schlenker W, Braun N, Little BB, Rejesus RM et al (2014) Greater sensitivity to drought accompanies maize yield increase in the US Midwest. Science 344(6183):516–519. https://doi.org/10.1126/science.1251423
Mace ES, Rami JF, Bouchet S et al (2009) A consensus genetic map of sorghum that integrates multiple component maps and high-throughput Diversity Array Technology (DArT) markers. BMC Plant Biol 9:13. https://doi.org/10.1186/1471-2229-9-13
Mace E, Singh V, Van Oosterom E, Hammer G, Hunt C, Jordan D (2012) QTL for nodal root angle in sorghum (Sorghum bicolor L. Moench) co-locate with QTL for traits associated with drought adaptation. Theor Appl Genet 124(1):97–109. https://doi.org/10.1007/s00122-011-1690-9
Mace E, Innes D, Hunt C, Wang X, Tao Y, Baxter J et al (2019) The Sorghum QTL Atlas: a powerful tool for trait dissection, comparative genomics and crop improvement. Theor Appl Genet 132(3):751–766. https://doi.org/10.1007/s00122-018-3212-5
Manschadi AM, Christopher J, deVoil P, Hammer GL (2006) The role of root architectural traits in adaptation of wheat to water-limited environments. Funct Plant Biol 33(9):823–837. https://doi.org/10.1071/FP06055
McCormick RF, Truong SK, Sreedasyam A, Jenkins J, Shu S, Sims D et al (2018) The Sorghum bicolor reference genome: improved assembly, gene annotations, a transcriptome atlas, and signatures of genome organization. Plant J 93(2):338–354. https://doi.org/10.1111/tpj.13781
Menamo T, Kassahun B, Borrell A, Jordan D, Tao Y, Hunt C et al (2021) Genetic diversity of Ethiopian sorghum reveals signatures of climatic adaptation. Theor Appl Genet 134(2):731–742. https://doi.org/10.1007/s00122-020-03727-5
Moghimi N, Desai JS, Bheemanahalli R, Impa SM, Vennapusa AR, Sebela D et al (2019) New candidate loci and marker genes on chromosome 7 for improved chilling tolerance in sorghum. J Exp Bot 50(12):3357–3371. https://doi.org/10.1093/jxb/erz143
Morris GP, Ramu P, Deshpande SP, Hash CT, Shah T, Upadhyaya HD et al (2013) Population genomic and genome-wide association studies of agroclimatic traits in sorghum. Proc Natl Acad Sci 110(2):453–458
Parra-Londono S, Kavka M, Samans B, Snowdon R, Wieckhorst S, Uptmoor R (2018) Sorghum root-system classification in contrasting P environments reveals three main rooting types and root-architecture-related marker–trait associations. Ann Bot 121(2):267–280. https://doi.org/10.1093/aob/mcx157
Reddy NRR, Ragimasalawada M, Sabbavarapu MM, Nadoor S, Patil JV (2014) Detection and validation of stay-green QTL in post-rainy sorghum involving widely adapted cultivar, M35–1 and a popular stay-green genotype B35. BMC Genomics 15(1):909. https://doi.org/10.1186/1471-2164-15-909
Rich SM, Christopher J, Richards R, Watt M (2020) Root phenotypes of young wheat plants grown in controlled environments show inconsistent correlation with mature root traits in the field. J Exp Bot 71(16):4751–4762
Rich SM, Watt M (2013) Soil conditions and cereal root system architecture: review and considerations for linking Darwin and Weaver. J Exp Bot 64(5):1193–1208
Richard CA, Hickey LT, Fletcher S et al (2015) High-throughput phenotyping of seminal root traits in wheat. Plant Methods 11:13. https://doi.org/10.1186/s13007-015-0055-9
Richard C, Christopher JT, Chenu K, Borrell AK, Christopher MJ, Hickey L (2018) Selection in early generations to shift allele frequency for seminal root angle in wheat the plant. Genome. https://doi.org/10.3835/plantgenome2017.08.0071
Robinson H, Hickey L, Richard C, Mace E, Kelly A, Borrell A, Franckowiak J, Fox G (2016) Genomic regions influencing seminal root traits in Barley. Plant Genome. https://doi.org/10.3835/plantgenome2015.03.0012
Robinson H, Kandic V, Christopher J, Fox G, Kelly A, Borrell A et al (2018) Root traits for yield improvement in barley. GRDC Update Papers
Sabadin P, Malosetti M, Boer M, Tardin F, Santos F, Guimaraes C et al (2012) Studying the genetic basis of drought tolerance in sorghum by managed stress trials and adjustments for phenological and plant height differences. Theor Appl Genet 124(8):1389–1402. https://doi.org/10.1007/s00122-012-1795-9
Sanguineti M, Li S, Maccaferri M, Corneti S, Rotondo F, Chiari T et al (2007) Genetic dissection of seminal root architecture in elite durum wheat germplasm. Ann Appl Biol 151(3):291–305. https://doi.org/10.1111/j.1744-7348.2007.00198.x
Singh V, Hammer G, Van Oosterom E (2008) Variability in structure and function of sorghum root systems. Global Issues, Paddock Action. In: Proceedings of the 14th Australian society of agronomy conference
Singh V, Oosterom E, Jordan D, Messina C, Cooper M, Hammer G (2010) Morphological and architectural development of root systems in sorghum and maize. Plant and Soil. 333:287–299. https://doi.org/10.1007/s11104-010-0343-0
Singh V, van Oosterom EJ, Jordan DR, Hunt CH, Hammer GL (2011) Genetic variability and control of nodal root angle in sorghum. Crop Sci. https://doi.org/10.2135/cropsci2011.01.0038
Singh V, van Oosterom EJ, Jordan DR, Hammer GL (2012) Genetic control of nodal root angle in sorghum and its implications on water extraction. Eur J Agron 42:3–10. https://doi.org/10.1016/j.eja.2012.04.006
Srinivas G, Satish K, Madhusudhana R, Reddy RN, Mohan SM, Seetharama N (2009) Identification of quantitative trait loci for agronomically important traits and their association with genic-microsatellite markers in sorghum. Theor Appl Genet 118(8):1439–1454. https://doi.org/10.1007/s00122-009-0993-6
Tao Y, Trusov Y, Zhao X, Wang X, Cruickshank AW, Hunt C et al (2021) Manipulating assimilate availability provides insight into the genes controlling grain size in sorghum. Plant J. https://doi.org/10.1111/tpj.15437
Tao Y, Zhao X, Wang X, Hathorn A, Hunt C, Cruickshank AW et al (2020) Large-scale GWAS in sorghum reveals common genetic control of grain size among cereals. Plant Biotechnol J 18(4):1093–1105. https://doi.org/10.1111/pbi.13284
Trachsel S, Kaeppler SM, Brown KM, Lynch JP (2011) Shovelomics: high throughput phenotyping of maize (Zea mays L.) root architecture in the field. Plant Soil 341(1–2):75–87. https://doi.org/10.1007/s11104-010-0623-8
Uga Y, Sugimoto K, Ogawa S, Rane J, Ishitani M, Hara N et al (2013) Control of root system architecture by DEEPER ROOTING 1 increases rice yield under drought conditions. Nat Genet 45(9):1097. https://doi.org/10.1038/ng.2725
Van-Raden PM (2008) Efficient methods to compute genomic predictions. J Dairy Sci 91(11):4414–4423. https://doi.org/10.3168/jds.2007-0980
Voss-Fels KP, Robinson H, Mudge SR, Richard C, Newman S, Wittkop B et al (2018) VERNALIZATION1 modulates root system architecture in wheat and barley. Mol Plant 11(1):226–229. https://doi.org/10.1016/j.molp.2017.10.005
Wang H, Chen G, Zhang H, Liu B, Yang Y, Qin L et al (2014) Identification of QTLs for salt tolerance at germination and seedling stage of Sorghum bicolor L. Moench Euphytica 196(1):117–127. https://doi.org/10.1007/s10681-013-1019-7
Wei T, Simko V, Levy M, Xie Y, Jin Y, Zemla J (2017) Package ‘corrplot’: visualization of a correlation matrix (Version 0.84). Stat 56:316–324
Williams JL, Sherman JD, Lamb P, Cook J, Lachowiec JA, Bourgault M (2022) Relationships between roots, the stay-green phenotype, and agronomic performance in barley and wheat grown in semi-arid conditions. Plant Phenome J 5(1):e220050. https://doi.org/10.1002/ppj2.20050
Zheng Z, Hey S, Jubery T, Liu H, Yang Y, Coffey L et al (2020) Shared genetic control of root system architecture between Zea mays and Sorghum bicolor. Plant Physiol 182(2):977–991. https://doi.org/10.1104/pp.19.00752
Acknowledgements
We thank the Ethiopian Biodiversity Institute (EBI), and EIAR’s Melkassa Agricultural Research Center for providing us with the sorghum landraces and passport data. This study was supported by the Bill and Melinda Gates Foundation PEARL (Program for Emerging Agricultural Leaders) Program.
Funding
This study was funded by The Bill and Melinda Gates Foundation (BMGF).
Author information
Authors and Affiliations
Contributions
KB, AB, DJ and EM conceived and designed the experiments. TM and KB performed the experiments. TM, EM, CH and YT analysed the data. TM, KB, DJ, EM, AB and YT wrote the paper and editing.
Corresponding author
Ethics declarations
Conflict of interest
Authors have declared that no competing interests exist.
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Communicated by Hai-Chun Jing.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Menamo, T., Borrell, A.K., Mace, E. et al. Genetic dissection of root architecture in Ethiopian sorghum landraces. Theor Appl Genet 136, 209 (2023). https://doi.org/10.1007/s00122-023-04457-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00122-023-04457-0