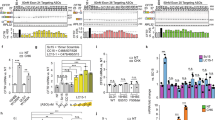
Abstract
Certain aminoglycosides are capable of inducing “translational readthrough” of premature termination codons (PTCs). However, toxicity and relative lack of efficacy deter treatment with clinically available aminoglycosides for genetic diseases caused by PTCs, including cystic fibrosis (CF). Using a structure-based approach, the novel aminoglycoside NB54 was developed that exhibits reduced toxicity and enhanced suppression of PTCs in cell-based reporter assays relative to gentamicin. We examined whether NB54 administration rescued CFTR protein and function in clinically relevant CF models. In a fluorescence-based halide efflux assay, NB54 partially restored halide efflux in a CF bronchial epithelial cell line (CFTR genotype W1282X/F508del), but not in a CF epithelial cell line lacking a PTC (F508del/F508del). In polarized airway epithelial cells expressing either a CFTR-W1282X or -G542X cDNA, treatment with NB54 increased stimulated short-circuit current (I SC) with greater efficiency than gentamicin. NB54 and gentamicin induced comparable increases in forskolin-stimulated I SC in primary airway epithelial cells derived from a G542X/F508del CF donor. Systemic administration of NB54 to Cftr−/− mice expressing a human CFTR-G542X transgene restored 15–17% of the average stimulated transepithelial chloride currents observed in wild-type (Cftr+/+) mice, comparable to gentamicin. NB54 exhibited reduced cellular toxicity in vitro and was tolerated at higher concentrations than gentamicin in vivo. These results provide evidence that synthetic aminoglycosides are capable of PTC suppression in relevant human CF cells and a CF animal model and support further development of these compounds as a treatment modality for genetic diseases caused by PTCs.







Similar content being viewed by others
Abbreviations
- AHB:
-
(S)-4-Amino-2 hydroxybutanoyl
- CF:
-
Cystic fibrosis
- CFTR:
-
Cystic fibrosis transmembrane conductance regulator
- I SC :
-
Short-circuit current
- PTC:
-
Premature termination codons
- SPQ:
-
6-Methoxy-N-(3-sulfopropyl)-quinolinium
References
Rowe SM, Miller S, Sorscher EJ (2005) Cystic fibrosis. N Engl J Med 352:1992–2001
Anonymous (1993) Correlation between genotype and phenotype in patients with cystic fibrosis. The Cystic Fibrosis Genotype-Phenotype Consortium. N Engl J Med 329:1308–1313
Shoshani T, Kerem E, Szeinberg A, Augarten A, Yahav Y, Cohen D, Rivlin J, Tal A, Kerem B (1994) Similar levels of mRNA from the W1282X and the delta F508 cystic fibrosis alleles, in nasal epithelial cells. J Clin Invest 93:1502–1507
Kerem E (2004) Pharmacologic therapy for stop mutations: how much CFTR activity is enough? Curr Opin Pulm Med 10:547–552
Keeling KM, Brooks DA, Hopwood JJ, Li P, Thompson JN, Bedwell DM (2001) Gentamicin-mediated suppression of Hurler syndrome stop mutations restores a low level of alpha-l-iduronidase activity and reduces lysosomal glycosaminoglycan accumulation. Hum Mol Genet 10:291–299
Howard M, Frizzell RA, Bedwell DM (1996) Aminoglycoside antibiotics restore CFTR function by overcoming premature stop mutations. Nat Med 2:467–469
Bedwell DM, Kaenjak A, Benos DJ, Bebok Z, Bubien JK, Hong J, Tousson A, Clancy JP, Sorscher EJ (1997) Suppression of a CFTR premature stop mutation in a bronchial epithelial cell line. Nat Med 3:1280–1284
Tok JB, Bi L (2003) Aminoglycoside and its derivatives as ligands to target the ribosome. Curr Top Med Chem 3:1001–1019
Bonetti B, Fu L, Moon J, Bedwell DM (1995) The efficiency of translation termination is determined by a synergistic interplay between upstream and downstream sequences in Saccharomyces cerevisiae. J Mol Biol 251:334–345
Barton-Davis ER, Cordier L, Shoturma DI, Leland SE, Sweeney HL (1999) Aminoglycoside antibiotics restore dystrophin function to skeletal muscles of mdx mice. J Clin Invest 104:375–381
Manuvakhova M, Keeling K, Bedwell DM (2000) Aminoglycoside antibiotics mediate context-dependent suppression of termination codons in a mammalian translation system. RNA 6:1044–1055
Clancy JP, Bebok Z, Ruiz F, King C, Jones J, Walker L, Greer H, Hong J, Wing L, Macaluso M et al (2001) Evidence that systemic gentamicin suppresses premature stop mutations in patients with cystic fibrosis. Am J Respir Crit Care Med 163:1683–1692
Sermet-Gaudelus I, Renouil M, Fajac A, Bidou L, Parbaille B, Pierrot S, Davy N, Bismuth E, Reinert P, Lenoir G et al (2007) In vitro prediction of stop-codon suppression by intravenous gentamicin in patients with cystic fibrosis: a pilot study. BMC Med 5:5–14
Wilschanski M, Famini C, Blau H, Rivlin J, Augarten A, Avital A, Kerem B, Kerem E (2000) A pilot study of the effect of gentamicin on nasal potential difference measurements in cystic fibrosis patients carrying stop mutations. Am J Respir Crit Care Med 161:860–865
Wilschanski M, Yahav Y, Yaacov Y, Blau H, Bentur L, Rivlin J, Aviram M, Bdolah-Abram T, Bebok Z, Shushi L et al (2003) Gentamicin-induced correction of CFTR function in patients with cystic fibrosis and CFTR stop mutations. N Engl J Med 349:1433–1441
Clancy JP, Rowe SM, Bebok Z, Aitken ML, Gibson R, Zeitlin P, Berclaz P, Moss R, Knowles MR, Oster RA et al (2007) No detectable improvements in cystic fibrosis transmembrane conductance regulator by nasal aminoglycosides in patients with cystic fibrosis with stop mutations. Am J Respir Cell Mol Biol 37:57–66
Du M, Liu X, Welch EM, Hirawat S, Peltz SW, Bedwell DM (2008) PTC124 is an orally bioavailable compound that promotes suppression of the human CFTR-G542X nonsense allele in a CF mouse model. Proc Natl Acad Sci U S A 105:2064–2069
Kerem E, Hirawat S, Armoni S, Yaakov Y, Shoseyov D, Cohen M, Nissim-Rafinia M, Blau H, Rivlin J, Aviram M et al (2008) Effectiveness of PTC124 treatment of cystic fibrosis caused by nonsense mutations: a prospective phase II trial. Lancet 372:719–727
Nudelman I, Rebibo-Sabbah A, Cherniavsky M, Belakhov V, Hainrichson M, Chen F, Schacht J, Pilch DS, Ben-Yosef T, Baasov T (2009) Development of novel aminoglycoside (NB54) with reduced toxicity and enhanced suppression of disease-causing premature stop mutations. J Med Chem 52:2836–2845
Nudelman I, Rebibo-Sabbah A, Shallom-Shezifi D, Hainrichson M, Stahl I, Ben-Yosef T, Baasov T (2006) Redesign of aminoglycosides for treatment of human genetic diseases caused by premature stop mutations. Bioorg Med Chem Lett 16:6310–6315
Rebibo-Sabbah A, Nudelman I, Ahmed ZM, Baasov T, Ben-Yosef T (2007) In vitro and ex vivo suppression by aminoglycosides of PCDH15 nonsense mutations underlying type 1 Usher syndrome. Hum Genet 122:373–381
Wu X, Wakefield JK, Liu H, Xiao H, Kralovics R, Prchal JT, Kappes JC (2000) Development of a novel trans-lentiviral vector that affords predictable safety. Mol Ther 2:47–55
Rowe SM, Varga K, Rab A, Bebok Z, Byram K, Li Y, Sorscher EJ, Clancy JP (2007) Restoration of W1282X CFTR activity by enhanced expression. Am J Respir Cell Mol Biol 37:347–356
Rowe SM, Pyle LC, Jurkevante A, Varga K, Collawn J, Sloane PA, Woodworth B, Mazur M, Fulton J, Fan L et al (2010) DeltaF508 CFTR processing correction and activity in polarized airway and non-airway cell monolayers. Pulm Pharmacol Ther 23:268–278
Van Goor F, Hadida S, Grootenhuis PD, Burton B, Cao D, Neuberger T, Turnbull A, Singh A, Joubran J, Hazlewood A et al (2009) Rescue of CF airway epithelial cell function in vitro by a CFTR potentiator, VX-770. Proc Natl Acad Sci U S A 106:18825–18830
Clancy JP, Hong JS, Bebok Z, King SA, Demolombe S, Bedwell DM, Sorscher EJ (1998) Cystic fibrosis transmembrane conductance regulator (CFTR) nucleotide-binding domain 1 (NBD-1) and CFTR truncated within NBD-1 target to the epithelial plasma membrane and increase anion permeability. Biochemistry 37:15222–15230
Cobb BR, Ruiz F, King CM, Fortenberry J, Greer H, Kovacs T, Sorscher EJ, Clancy JP (2002) A(2) adenosine receptors regulate CFTR through PKA and PLA(2). Am J Physiol Lung Cell Mol Physiol 282:L12–L25
Sermet-Gaudelus I, Vallee B, Urbin I, Torossi T, Marianovski R, Fajac A, Feuillet MN, Bresson JL, Lenoir G, Bernaudin JF, Edelman A (2002) Normal function of the cystic fibrosis conductance regulator protein can be associated with homozygous (delta)F508 mutation. Pediatr Res 52:628–635
Jurkuvenaite A, Varga K, Nowotarski K, Kirk KL, Sorscher EJ, Li Y, Clancy JP, Bebok Z, Collawn JF (2006) Mutations in the amino terminus of the cystic fibrosis transmembrane conductance regulator enhance endocytosis. J Biol Chem 281:3329–3334
Ratcliff R, Evans MJ, Cuthbert AW, MacVinish LJ, Foster D, Anderson JR, Colledge WH (1993) Production of a severe cystic fibrosis mutation in mice by gene targeting. Nat Genet 4:35–41
Du M, Jones JR, Lanier J, Keeling KM, Lindsey RJ, Tousson A, Bebok Z, Whitsett JA, Dey CR, Colledge WH et al (2002) Aminoglycoside suppression of a premature stop mutation in a Cftr−/− mouse carrying a human CFTR-G542X transgene. J Mol Med 80:595–604
Du M, Keeling KM, Fan L, Liu X, Kovacs T, Sorscher E, Bedwell DM (2006) Clinical doses of amikacin provide more effective suppression of the human CFTR-G542X stop mutation than gentamicin in a transgenic CF mouse model. J Mol Med 84:573–582
Du M, Keeling KM, Fan L, Liu X, Bedwell DM (2009) Poly-l-aspartic acid enhances and prolongs gentamicin-mediated suppression of the CFTR-G542X mutation in a cystic fibrosis mouse model. J Biol Chem 284:6885–6892
Varga K, Jurkuvenaite A, Wakefield J, Hong JS, Guimbellot JS, Venglarik CJ, Niraj A, Mazur M, Sorscher EJ, Collawn JF, Bebok Z (2004) Efficient intracellular processing of the endogenous cystic fibrosis transmembrane conductance regulator in epithelial cell lines. J Biol Chem 279:22578–22584
Pyle LC, Fulton JC, Sloane PA, Backer K, Mazur M, Prasain J, Barnes S, Clancy JP, Rowe SM (2009) Activation of the cystic fibrosis transmembrane conductance regulator by the flavonoid quercetin: potential use as a biomarker of deltaF508 cystic fibrosis transmembrane conductance regulator rescue. Am J Respir Cell Mol Biol 43:607–616
Wilschanski M, Dupuis A, Ellis L, Jarvi K, Zielenski J, Tullis E, Martin S, Corey M, Tsui LC, Durie P (2006) Mutations in the cystic fibrosis transmembrane regulator gene and in vivo transepithelial potentials. Am J Respir Crit Care Med 174:787–794
Bijvelds MJ, Bot AG, Escher JC, De Jonge HR (2009) Activation of intestinal Cl− secretion by lubiprostone requires the cystic fibrosis transmembrane conductance regulator. Gastroenterology 137:976–985
Rowe SM, Clancy JP (2009) Pharmaceuticals targeting nonsense mutations in genetic diseases: progress in development. BioDrugs 23:165–174
Keeling K, Bedwell DM (2005) Pharmacological suppression of premature stop mutations that cause genetic diseases. Current Pharmacogenomics 3:259–269
Keeling K, Bedwell DM (2010) Recoding therapies for genetic diseases. In: Gesteland R, Atkins J (eds) Recoding: expansion of decoding rules enriches gene expression. Springer, New York, pp 123–146
Malik V, Rodino-Klapac LR, Viollet L, Wall C, King W, Al-Dahhak R, Lewis S, Shilling CJ, Kota J, Serrano-Munuera C et al (2010) Gentamicin-induced readthrough of stop codons in Duchenne muscular dystrophy. Ann Neurol 67:771–780
Mingeot-Leclercq MP, Tulkens PM (1999) Aminoglycosides: nephrotoxicity. Antimicrob Agents Chemother 43:1003–1012
Hutchin T, Cortopassi G (1994) Proposed molecular and cellular mechanism for aminoglycoside ototoxicity. Antimicrob Agents Chemother 38:2517–2520
Rowe SM, Clancy JP, Boyle M, Van Goor F, Ordonez C, Dong Q, Campbell P, Ashlock M, Accurso F (2010) Parallel effects of VX-770 on transepithelial potential difference in vitro and in vivo. J Cyst Fibros 9:S20
Moestrup SK, Cui S, Vorum H, Bregengard C, Bjorn SE, Norris K, Gliemann J, Christensen EI (1995) Evidence that epithelial glycoprotein 330/megalin mediates uptake of polybasic drugs. J Clin Invest 96:1404–1413
Acknowledgments
The authors thank Kim Keeling for critically reading the manuscript. This study was supported by US National Institute of Health grants 1K23DK075788-01 and 1R03DK084110-01 (to SMR), 1P30DK072482-01A1 and Cystic Fibrosis Foundation grant R464-CR02 (to EJS), US-Israel Binational Science Foundation grant 2006/301 and Mitchel Fund 2012386 (to TB), and Horowitz Fund (grant no. 2010826 to TB and DMB). VB acknowledges the financial support by the Center of Absorption in Science, the Ministry of Immigration Absorption, and the Ministry of Science and Technology, Israel (Kamea Program).
Disclosure statement
SMR serves on the advisory board for PTC Therapeutics, Inc. DMB serves as a consultant for PTC Therapeutics, Inc.
Author information
Authors and Affiliations
Corresponding author
Additional information
This research was funded by the US National Institute of Health grants 1K23DK075788-01 and 1R03DK084110-01 (to SMR), 1P30DK072482-01A1 and Cystic Fibrosis Foundation grant R464-CR02 (to EJS), US-Israel Binational Science Foundation grant 2006/301 and Mitchel Fund 2012386 (to TB), and Horowitz Fund (grant no. 2010826 to TB and DMB). VB acknowledges the financial support by the Center of Absorption in Science, the Ministry of Immigration Absorption, and the Ministry of Science and Technology, Israel (Kamea Program). We are grateful to Cheryl Owens for assistance with preparation of the manuscript.
Electronic supplementary materials
Below is the link to the electronic supplementary material.
ESM 1
PDF 46 kb
Rights and permissions
About this article
Cite this article
Rowe, S.M., Sloane, P., Tang, L.P. et al. Suppression of CFTR premature termination codons and rescue of CFTR protein and function by the synthetic aminoglycoside NB54. J Mol Med 89, 1149–1161 (2011). https://doi.org/10.1007/s00109-011-0787-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00109-011-0787-6