Abstract
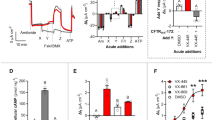
Mutations in the cystic fibrosis transmembrane conductance regulator (CFTR) gene cause the disease cystic fibrosis. We previously reported that gentamicin administration suppressed a CFTR premature stop mutation in a Cftr−/− mouse model carrying a human CFTR-G542X (hCFTR-G542X) transgene, resulting in the appearance of hCFTR protein and function. However, the high doses used in that study resulted in peak serum levels well beyond the levels typically administered to humans. To address this problem, we identified doses of both gentamicin and amikacin that resulted in peak serum levels within their accepted clinical ranges. We then asked whether these doses could suppress the hCFTR-G542X mutation in the Cftr−/− hCFTR-G542X mouse model. Our results indicate that low doses of each compound restored some hCFTR protein expression and function, as shown by immunofluorescence and short-circuit current measurements. However, we found that amikacin suppressed the hCFTR-G542X premature stop mutation more effectively than gentamicin when administered at these clinically relevant doses. Because amikacin is also less toxic than gentamicin, it may represent a superior choice for suppression therapy in patients that carry a premature stop mutation in the CFTR gene.




Similar content being viewed by others
References
Krawczak M, Ball EV, Fenton I, Stenson PD, Abeysinghe S, Thomas N, Cooper DN (2000) Human gene mutation database—a biomedical information and research resource. Hum Mutat 15:45–51
Fearon K, McClendon V, Bonetti B, Bedwell DM (1994) Premature translation termination mutations are efficiently suppressed in a highly conserved region of yeast Ste6p, a member of the ATP-binding cassette (ABC) transporter family. J Biol Chem 269:17802–17808
Bonetti B, Fu L, Moon J, Bedwell DM (1995) The efficiency of translation termination is determined by a synergistic interplay between upstream and downstream sequences in Saccharomyces cerevisiae. J Mol Biol 251:334–345
Manuvakhova M, Keeling K, Bedwell DM (2000) Aminoglycoside antibiotics mediate context-dependent suppression of termination codons in a mammalian translation system. RNA 6:1044–1055
Keeling KM, Bedwell DM (2002) Clinically relevant aminoglycosides can suppress disease-associated premature stop mutations in the IDUA and P53 cDNAs in a mammalian translation system. J Mol Med 80:367–376
Zsembery A, Jessner W, Sitter G, Spirli C, Strazzabosco M, Graf J (2002) Correction of CFTR malfunction and stimulation of Ca-activated Cl channels restore HCO3- secretion in cystic fibrosis bile ductular cells. Hepatology 35:95–104
Howard MT, Shirts BH, Petros LM, Flanigan KM, Gesteland RF, Atkins JF (2000) Sequence specificity of aminoglycoside-induced stop codon readthrough: potential implications for treatment of Duchenne muscular dystrophy. Ann Neurol 48:164–169
Howard M, Frizzell RA, Bedwell DM (1996) Aminoglycoside antibiotics restore CFTR function by overcoming premature stop mutations. Nat Med 2:467–469
Bedwell DM, Kaenjak A, Benos DJ, Bebok Z, Bubien JK, Hong J, Tousson A, Clancy JP, Sorscher EJ (1997) Suppression of a CFTR premature stop mutation in a bronchial epithelial cell line. Nat Med 3:1280–1284
Barton-Davis ER, Cordier L, Shoturma DI, Leland SE, Sweeney HL (1999) Aminoglycoside antibiotics restore dystrophin function to skeletal muscles of mdx mice. J Clin Invest 104:375–381
Sleat DE, Sohar I, Gin RM, Lobel P (2001) Aminoglycoside-mediated suppression of nonsense mutations in late infantile neuronal ceroid lipofuscinosis. Eur J Paediatr Neurol 5(Suppl A):57–62
Keeling KM, Brooks DA, Hopwood JJ, Li P, Thompson JN, Bedwell DM (2001) Gentamicin-mediated suppression of Hurler syndrome stop mutations restores a low level of alpha-l-iduronidase activity and reduces lysosomal glycosaminoglycan accumulation. Hum Mol Genet 10:291–299
Hein LK, Bawden M, Muller VJ, Sillence D, Hopwood JJ, Brooks DA (2004) Alpha-l-iduronidase premature stop codons and potential read-through in mucopolysaccharidosis type I patients. J Mol Biol 338:453–462
Sossi V, Giuli A, Vitali T, Tiziano F, Mirabella M, Antonelli A, Neri G, Brahe C (2001) Premature termination mutations in exon 3 of the SMN1 gene are associated with exon skipping and a relatively mild SMA phenotype. Eur J Hum Genet 9:113–120
Schulz A, Sangkuhl K, Lennert T, Wigger M, Price DA, Nuuja A, Gruters A, Schultz G, Schoneberg T (2002) Aminoglycoside pretreatment partially restores the function of truncated V(2) vasopressin receptors found in patients with nephrogenic diabetes insipidus. J Clin Endocrinol Metab 87:5247–5257
Helip-Wooley A, Park MA, Lemons RM, Thoene JG (2002) Expression of CTNS alleles: subcellular localization and aminoglycoside correction in vitro. Mol Genet Metab 75:128–133
Wolstencroft EC, Mattis V, Bajer AA, Young PJ, Lorson CL (2005) A non-sequence-specific requirement for SMN protein activity: the role of aminoglycosides in inducing elevated SMN protein levels. Hum Mol Genet 14:1199–1210
Lai CH, Chun HH, Nahas SA, Mitui M, Gamo KM, Du L, Gatti RA (2004) Correction of ATM gene function by aminoglycoside-induced read-through of premature termination codons. Proc Natl Acad Sci USA 101:15676–15681
Aguiari G, Banzi M, Gessi S, Cai Y, Zeggio E, Manzati E, Piva R, Lambertini E, Ferrari L, Peters DJ, Lanza F, Harris PC, Borea PA, Somlo S, Del Senno L (2004) Deficiency of polycystin-2 reduces Ca2+ channel activity and cell proliferation in ADPKD lymphoblastoid cells. FASEB J 18:884–886
Du M, Jones JR, Lanier J, Keeling KM, Lindsey JR, Tousson A, Bebok Z, Whitsett JA, Dey CR, Colledge WH, Evans MJ, Sorscher EJ, Bedwell DM (2002) Aminoglycoside suppression of a premature stop mutation in a Cftr−/− mouse carrying a human CFTR-G542X transgene. J Mol Med 80:595–604
Politano L, Nigro G, Nigro V, Piluso G, Papparella S, Paciello O, Comi LI (2003) Gentamicin administration in Duchenne patients with premature stop codon. Preliminary results. Acta Myol 22:15–21
Wilschanski M, Yahav Y, Yaacov Y, Blau H, Bentur L, Rivlin J, Aviram M, Bdolah-Abram T, Bebok Z, Shushi L, Kerem B, Kerem E (2003) Gentamicin-induced correction of CFTR function in patients with cystic fibrosis and CFTR stop mutations. N Engl J Med 349:1433–1441
Wilschanski M, Famini C, Blau H, Rivlin J, Augarten A, Avital A, Kerem B, Kerem E (2000) A pilot study of the effect of gentamicin on nasal potential difference measurements in cystic fibrosis patients carrying stop mutations. Am J Respir Crit Care Med 161:860–865
Clancy JP, Bebok Z, Ruiz F, King C, Jones J, Walker L, Greer H, Hong J, Wing L, Macaluso M, Lyrene R, Sorscher EJ, Bedwell DM (2001) Evidence that systemic gentamicin suppresses premature stop mutations in patients with cystic fibrosis. Am J Respir Crit Care Med 163:1683–1692
Snouwaert JN, Brigman KK, Latour AM, Malouf NN, Boucher RC, Smithies O, Koller BH (1992) An animal model for cystic fibrosis made by gene targeting. Science 257:1083–1088
Koerner TJ, Hill JE, Myers AM, Tzagoloff A (1991) High-expression vectors with multiple cloning sites for construction of trpE fusion genes: pATH vectors. Methods Enzymol 194:477–490
Dawson DC (1977) Na and Cl transport across the isolated turtle colon: parallel pathways for transmural ion movement. J Membr Biol 37:213–233
Arky R (1996) Physicians’ desk reference. Medical economics, Montvale, NJ
Beauchamp D, Labrecque G (2001) Aminoglycoside nephrotoxicity: do time and frequency of administration matter? Curr Opin Crit Care 7:401–408
Nagai J, Takano M (2004) Molecular aspects of renal handling of aminoglycosides and strategies for preventing the nephrotoxicity. Drug Metab Pharmacokinet 19:159–170
Mingeot-Leclercq MP, Tulkens PM (1999) Aminoglycosides: nephrotoxicity. Antimicrob Agents Chemother 43:1003–1012
Laurent G, Carlier MB, Rollman B, Van Hoof F, Tulkens P (1982) Mechanism of aminoglycoside-induced lysosomal phospholipidosis: in vitro and in vivo studies with gentamicin and amikacin. Biochem Pharmacol 31:3861–3870
De Broe ME, Paulus GJ, Verpooten GA, Roels F, Buyssens N, Wedeen R, Van Hoof F, Tulkens PM (1984) Early effects of gentamicin, tobramycin, and amikacin on the human kidney. Kidney Int 25:643–652
Carlier MB, Laurent G, Claes PJ, Vanderhaeghe HJ, Tulkens PM (1983) Inhibition of lysosomal phospholipases by aminoglycoside antibiotics: in vitro comparative studies. Antimicrob Agents Chemother 23:440–449
Lynch SR, Puglisi JD (2001) Structure of a eukaryotic decoding region A-site RNA. J Mol Biol 306:1023–1035
Yoshizawa S, Fourmy D, Puglisi JD (1998) Structural origins of gentamicin antibiotic action. EMBO J 17:6437–6448
Kohlhepp SJ, Loveless MO, Kohnen PW, Houghton DC, Bennett WM, Gilbert DN (1984) Nephrotoxicity of the constituents of the gentamicin complex. J Infect Dis 149:605–614
Kobayashi M, Umemura M, Sone M, Nakashima T (2003) Differing effects on the inner ear of three gentamicin compounds: GM-C1, -C2 and -C1a. Acta Otolaryngol 123:916–922
Isoherranen N, Lavy E, Soback S (2000) Pharmacokinetics of gentamicin C(1), C(1a), and C(2) in beagles after a single intravenous dose. Antimicrob Agents Chemother 44:1443–1447
Bartal C, Danon A, Schlaeffer F, Reisenberg K, Alkan M, Smoliakov R, Sidi A, Almog Y (2003) Pharmacokinetic dosing of aminoglycosides: a controlled trial. Am J Med 114:194–198
Mazzon E, Britti D, De Sarro A, Caputi AP, Cuzzocrea S (2001) Effect of N-acetylcysteine on gentamicin-mediated nephropathy in rats. Eur J Pharmacol 424:75–83
Kawamoto K, Sha SH, Minoda R, Izumikawa M, Kuriyama H, Schacht J, Raphael Y (2004) Antioxidant gene therapy can protect hearing and hair cells from ototoxicity. Mol Ther 9:173–181
Nakashima T, Teranishi M, Hibi T, Kobayashi M, Umemura M (2000) Vestibular and cochlear toxicity of aminoglycosides–a review. Acta Otolaryngol 120:904–911
Sener G, Sehirli AO, Altunbas HZ, Ersoy Y, Paskaloglu K, Arbak S, Ayanoglu-Dulger G (2002) Melatonin protects against gentamicin-induced nephrotoxicity in rats. J Pineal Res 32:231–236
Beauchamp D, Laurent G, Maldague P, Abid S, Kishore BK, Tulkens PM (1990) Protection against gentamicin-induced early renal alterations (phospholipidosis and increased DNA synthesis) by coadministration of poly-l-aspartic acid. J Pharmacol Exp Ther 255:858–866
Gilbert DN, Wood CA, Kohlhepp SJ, Kohnen PW, Houghton DC, Finkbeiner HC, Lindsley J, Bennett WM (1989) Polyaspartic acid prevents experimental aminoglycoside nephrotoxicity. J Infect Dis 159:945–953
Thibault N, Grenier L, Simard M, Bergeron MG, Beauchamp D (1994) Attenuation by daptomycin of gentamicin-induced experimental nephrotoxicity. Antimicrob Agents Chemother 38:1027–1035
Thibault N, Grenier L, Simard M, Bergeron MG, Beauchamp D (1995) Protection against gentamicin nephrotoxicity by daptomycin in nephrectomized rats. Life Sci 56:1877–1887
Ramalho AS, Beck S, Meyer M, Penque D, Cutting GR, Amaral MD (2002) Five percent of normal cystic fibrosis transmembrane conductance regulator mRNA ameliorates the severity of pulmonary disease in cystic fibrosis. Am J Respir Cell Mol Biol 27:619–627
Kerem E (2004) Pharmacologic therapy for stop mutations: how much CFTR activity is enough? Curr Opin Pulm Med 10:547–552
Acknowledgements
The authors wish to thank Lee Sweeney and Erik Schwiebert for helpful discussions. This work was supported by NIH grant P50 DK 53090.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Du, M., Keeling, K.M., Fan, L. et al. Clinical doses of amikacin provide more effective suppression of the human CFTR-G542X stop mutation than gentamicin in a transgenic CF mouse model. J Mol Med 84, 573–582 (2006). https://doi.org/10.1007/s00109-006-0045-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00109-006-0045-5