Abstract
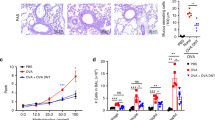
T cells expressing a type-2 T helper profile of cytokines (Th2 cells) have been demonstrated to play an important role in the initiation and progression of allergic asthma, and it is well known that Fas ligand (FasL) induces apoptosis when bound to its receptor, Fas. In the present study, we examined the possibility of modulating asthma manifestations by dendritic cells (DCs) genetically engineered to express FasL (DC-FasL), which could deliver a death signal to T cells in an antigen-specific manner. The delivery of DC-FasL into ovalbumin (OVA)-immunized allergic mice decreased the airway hyper-responsiveness (AHR). Moreover, we established a mouse model of airway inflammation by using an adoptive transfer of Th2 cells derived from ovalbumin T cell receptor transgenic mice to study the effect of DC-FasL on airway reactivity. The administration of DC-FasL in Th2-cell-induced allergic mice had significantly decreased AHR, airway inflammation, and IL-4, IL-5 and IL-13 production. Furthermore, the numbers of OVA-specific T cells were decreased in the lung of mice receiving DC-FasL. These results demonstrate that FasL-expressing dendritic cells might be applied for the modulation of allergic responses.






Similar content being viewed by others
Abbreviations
- FasL:
-
Fas ligand
- OVA:
-
Ovalbumin
- AHR:
-
Airway hypersensitivity
- DC:
-
Dendritic cell
- BMDC:
-
Bone-marrow-derived dendritic cell
- BALF:
-
Bronchoalveolar lavage fluid
- Penh:
-
Enhanced pulse
References
Herrick CA, Bottomly K (2003) To respond or not to respond: T cells in allergic asthma. Nat Rev Immunol 3:405–412
Leong KP, Huston DP (2001) Understanding the pathogenesis of allergic asthma using mouse models. Ann Allergy Asthma Immunol 87:96–109
Lee YL, Fu CL, Ye YL, Chiang BL (1999) Administration of interleukin-12 prevents mite Der p 1 allergen-IgE antibody production and airway eosinophil infiltration in an animal model of airway inflammation. Scand J Immunol 49:229–236
Chiang DJ, Ye YL, Chen WL, Lee YL, Hsu NY, Chiang BL (2003) Ribavirin or CpG DNA sequence-modulated dendritic cells decrease the IgE level and airway inflammation. Am J Respir Crit Care Med 168:575–580
Hamelmann E, Oshiba A, Loader J, Larsen GL, Gleich G, Lee J, Gelfand EW (1997) Antiinterleukin-5 antibody prevents airway hyperresponsiveness in a murine model of airway sensitization. Am J Respir Crit Care Med 155:819–825
Shardonofsky FR, Venzor J III, Barrios R, Leong KP, Huston DP (1999) Therapeutic efficacy of an anti-IL-5 monoclonal antibody delivered into the respiratory tract in a murine model of asthma. J Allergy Clin Immunol 104:215–221
Hansen G, Berry G, DeKruyff RH, Umetsu DT (1999) Allergen-specific Th1 cells fail to counterbalance Th2 cell-induced airway hyperreactivity but cause severe airway inflammation. J Clin Invest 103:175–183
Randolph DA, Stephens R, Carruthers CJ, Chaplin DD (1999) Cooperation between Th1 and Th2 cells in a murine model of eosinophilic airway inflammation. J Clin Invest 104:1021–1029
Randolph DA, Carruthers CJ, Szabo SJ, Murphy KM, Chaplin DD (1999) Modulation of airway inflammation by passive transfer of allergen-specific Th1 and Th2 cells in a mouse model of asthma. J Immunol 162:2375–2783
Takaoka A, Tanaka Y, Tsuji T, Jinushi T, Hoshino A, Asakura Y, Mita Y, Watanabe K, Nakaike S, Togashi Y, Koda T, Matsushima K, Nishimura T (2001) A critical role for mouse CXC chemokine(s) in pulmonary neutrophilia during Th type 1-dependent airway inflammation. J Immunol 167:2349–2353
Dhein J, Walczak H, Baumler C, Debatin KM, Krammer PH (1995) Autocrine T-cell suicide mediated by APO-1/(Fas/CD95). Nature 373:438–441
Ju ST, Panka DJ, Cui H, Ettinger R, el Khatib M, Sherr DH, Stanger BZ, Marshak-Rothstein A (1995) Fas(CD95)/FasL interactions required for programmed cell death after T-cell activation. Nature 373:444–448
Lau HT, Yu M, Fontana A, Stoeckert CJ Jr (1996) Prevention of islet allograft rejection with engineered myoblasts expressing FasL in mice. Science 273:109–112
Arai H, Gordon D, Nabel EG, Nabel GJ (1997) Gene transfer of Fas ligand induces tumor regression in vivo. Proc Natl Acad Sci U S A 94:13862–13867
Ke B, Coito AJ, Kato H, Zhai Y, Wang T, Sawitzki B, Seu P, Busuttil RW, Kupiec-Weglinski JW (2000) Fas ligand gene transfer prolongs rat renal allograft survival and down-regulates anti-apoptotic Bag-1 in parallel with enhanced Th2-type cytokine expression. Transplantation 69:1690–1694
Batteux F, Lores P, Bucchini D, Chiocchia G (2000) Transgenic expression of Fas ligand on thyroid follicular cells prevents autoimmune thyroiditis. J Immunol 164:1681–1688
Zhu B, Luo L, Chen Y, Paty DW, Cynader MS (2002) Intrathecal Fas ligand infusion strengthens immunoprivilege of central nervous system and suppresses experimental autoimmune encephalomyelitis. J Immunol 169:1561–1569
Chuang YH, Fu CL, Lo YC, Chiang BL (2004) Adenovirus expressing Fas ligand gene decreases airway hyper-responsiveness and eosinophilia in a murine model of asthma. Gene Ther 11:1497–1505
Banchereau J, Briere F, Caux C, Davoust J, Lebecque S, Liu YJ, Pulendran B, Palucka K (2000) Immunobiology of dendritic cells. Annu Rev Immunol 18:767–811
Bell D, Young JW, Banchereau J (1999) Dendritic cells. Adv Immunol 72:255–324
Hackstein H, Morelli AE, Thomson AW (2001) Designer dendritic cells for tolerance induction: guided not misguided missiles. Trends Immunol 22:437–442
Suen JL, Chuang YH, Chiang BL (2002) In vivo tolerance breakdown with dendritic cells pulsed with U1A protein in non-autoimmune mice: the induction of a high level of autoantibodies but not renal pathological changes. Immunology 106:326–335
Lee YL, Ye YL, Yu CI, Wu YL, Lai YL, Ku PH, Hong RL, Chiang BL (2001) Construction of single-chain interleukin-12 DNA plasmid to treat airway hyperresponsiveness in an animal model of asthma. Hum Gene Ther 12:2065–2079
Adler A, Cieslewicz G, Irvin CG (2005) Unrestrained plethysmography is an unreliable measure of airway responsiveness in BALB/c and C57BL/6 mice. J Appl Physiol 97:286–292
Schwarze J, Hamelmann E, Gelfand EW (2005) Barometric whole body plethysmography in mice. J Appl Physiol 98:1955–1957
Matsue H, Matsue K, Kusuhara M, Kumamoto T, Okumura K, Yagita H, Takashima A (2001) Immunosuppressive properties of CD95L-transduced “killer” hybrids created by fusing donor- and recipient-derived dendritic cells. Blood 98:3465–3472
Min WP, Gorczynski R, Huang XY, Kushida M, Kim P, Obataki M, Lei J, Suri RM, Cattral MS (2000) Dendritic cells genetically engineered to express Fas ligand induce donor-specific hyporesponsiveness and prolong allograft survival. J Immunol 164:161–167
Kusuhara M, Matsue K, Edelbaum D, Loftus J, Takashima A, Matsue H (2002) Killing of naive T cells by CD95L-transfected dendritic cells (DC): in vivo study using killer DC–DC hybrids and CD4(+) T cells from DO11.10 mice. Eur J Immunol 32:1035–1043
Whartenby KA, Straley EE, Kim H, Racke F, Tanavde V, Gorski KS, Cheng L, Pardoll DM, Civin CI (2002) Transduction of donor hematopoietic stem-progenitor cells with Fas ligand enhanced short-term engraftment in a murine model of allogeneic bone marrow transplantation. Blood 100:3147–3154
Zhan HG, Mountz JD, Fleck M, Zhou T, Hsu HC (2002) Specific deletion of autoreactive T cells by adenovirus-transfected, Fas ligand-producing antigen-presenting cells. Immunol Res 26:235–246
Zhang HG, Yang P, Xie J, Liu Z, Liu D, Xiu L, Zhou T, Wang Y, Hsu HC, Mountz JD (2002) Depletion of collagen II-reactive T cells and blocking of B cell activation prevents collagen II-induced arthritis in DBA/1j mice. J Immunol 168:4164–4172
Zhang HG, Liu D, Heike Y, Yang P, Wang Z, Wang X, Curiel DT, Zhou T, Mountz JD (1998) Induction of specific T-cell tolerance by adenovirus-transfected, Fas ligand-producing antigen presenting cells. Nat Biotechnol 16:1045–1049
Zhang HG, Su X, Liu D, Liu W, Yang P, Wang Z, Edwards CK, Bluethmann H, Mountz JD, Zhou T (1999) Induction of specific T cell tolerance by Fas ligand-expressing antigen-presenting cells. J Immunol 162:1423–1430
Matsue H, Matsue K, Walters M, Okumura K, Yagita H, Takashima A (1999) Induction of antigen-specific immunosuppression by CD95L cDNA-transfected ‘killer’ dendritic cells. Nat Med 5:930–937
Kim SH, Kim S, Oligino TJ, Robbins PD (2002) Effective treatment of established mouse collagen-induced arthritis by systemic administration of dendritic cells genetically modified to express FasL. Mol Ther 6:584–590
Zhang HG, Fleck M, Kern ER, Liu D, Wang Y, Hsu HC, Yang P, Wang Z, Curiel DT, Zhou T, Mountz JD (2000) Antigen presenting cells expressing Fas ligand down-modulate chronic inflammatory disease in Fas ligand-deficient mice. J Clin Invest 105:813–821
Wolfe T, Asseman C, Hughes A, Matsue H, Takashima A, von Herrath MG (2002) Reduction of antiviral CD8 lymphocytes in vivo with dendritic cells expressing Fas ligand-increased survival of viral (lymphocytic choriomeningitis virus) central nervous system infection. J Immunol 169:4867–4872
Buonocore S, Flamand V, Claessen N, Heeringa P, Goldman M, Floquin S (2004) Dendritic cells overexpressing Fas-ligand induce pulmonary vasculitis in mice. Clin Exp Immunol 137:74–80
Lambrecht BN, De Veerman M, Coyle AJ, Gutierrez-Ramos JC, Thielemans K, Pauwels RA (2000) Myeloid dendritic cells induce Th2 responses to inhaled antigen, leading to eosinophilic airway inflammation. J Clin Invest 106:551–559
Lambrecht BN, Pauwels RA, Fazekas De St GB (2000) Induction of rapid T cell activation, division, and recirculation by intratracheal injection of dendritic cells in a TCR transgenic model. J Immunol 164:2937–2946
van Rijt LS, Prins JB, Leenen PJ, Thielemans K, de Vries VC, Hoogsteden HC, Lambrecht BN (2002) Allergen-induced accumulation of airway dendritic cells is supported by an increase in CD31(hi)Ly-6C(neg) bone marrow precursors in a mouse model of asthma. Blood 100:3663–3671
Graffi SJ, Dekan G, Stingl G, Epstein MM (2002) Systemic administration of antigen-pulsed dendritic cells induces experimental allergic asthma in mice upon aerosol antigen rechallenge. Clin Immunol 103:176–184
Akbari O, Freeman GJ, Meyer EH, Greenfield EA, Chang TT, Sharpe AH, Berry G, DeKruyff RH, Umetsu DT (2002) Antigen-specific regulatory T cells develop via the ICOS-ICOS-ligand pathway and inhibit allergen-induced airway hyperreactivity. Nat Med 8:1024–1032
Akbari O, DeKruyff RH, Umetsu DT (2001) Pulmonary dendritic cells producing IL-10 mediate tolerance induced by respiratory exposure to antigen. Nat Immunol 2:725–731
Ye YL, Lee YL, Chuang ZJ, Lai HJ, Chen CC, Tao MH, Chiang BL (2004) Dendritic cells modulated by cytokine-expressing adenoviruses alleviate eosinophilia and airway hyperresponsiveness in an animal model of asthma. J Allergy Clin Immunol 114:88–96
Acknowledgement
This work was supported by a grant from National Science Council of the Republic of China.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chuang, YH., Suen, JL. & Chiang, BL. Fas-ligand-expressing adenovirus-transfected dendritic cells decrease allergen-specific T cells and airway inflammation in a murine model of asthma. J Mol Med 84, 595–603 (2006). https://doi.org/10.1007/s00109-006-0047-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00109-006-0047-3