Abstract
Background
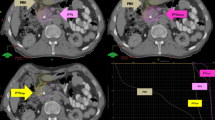
Stereotactic body radiation therapy (SBRT) represents a new treatment option for locally advanced pancreatic cancer (LAPC). An accurate treatment planning with risk-adapted dose prescription with adherence to specific dose constraints for organs at risk (OARs) and the use of daily cone beam CT (CBCT) for image guidance could allow an effective and safe treatment delivery. Here, feasibility and efficacy of SBRT in LAPC treated in our cancer care center are reported.
Patients and methods
33 unresectable LAPC patients underwent SBRT. In order to respect OAR dose constraints, a risk-adapted dose prescription strategy was adopted, choosing between the following schedules: 42 Gy or 45 Gy in 6 daily fractions with a biologically effective dose (BED) > 70 Gy10 or 36 Gy/6 fractions (estimating a BED 57.6 Gy10). SBRT was delivered with volumetric modulated arc technique (VMAT) and flattening filter-free (FFF) mode. Image guidance was performed by means of CBCT before every treatment session. The patients were evaluated at the end of treatment for acute toxicity and at 3, 6, and 12 months for late toxicity and treatment response.
Results
At the time of analysis, the median follow-up was 18 months (range 5–34 months). Prior to SBRT, 24 out of 33 patients received induction chemotherapy. Although all patients were previously judged as unresectable, 6 out of 33 (18%) underwent surgery after SBRT; all of them received a BED > 70 Gy10. One-year LC and OS were 81% and 75%, respectively. A total of 12 patients (37%) had an extra-pancreatic progression. No cases of ≥G3 acute or late toxicity were reported.
Conclusion
In our experience, risk-adapted dose prescription and image-guided SBRT represents an effective treatment option for LAPC patients.
Zusammenfassung
Hintergrund
Stellt die Körperstereotaxie (SBRT) eine neue Behandlungsoption für lokal fortgeschrittene Pankreaskarzinome (LAPC) dar. Eine präzise Bestrahlungsplanung mit risikoadaptierten Dosiskonzepten und Einhaltung spezifischer Toleranzdosisgrenzen für Risikoorgane (OARs) sowie die Anwendung von täglicher Image-Guidance mittels Cone-beam-Computertomographie (CBCT) könnte eine effektive und sichere Behandlungsoption darstellen. Wir berichten über die Anwendbarkeit und Effektivität von SBRT bei LAPC-Patienten, die in unserem Krebszentrum behandelt wurden.
Patienten und Methoden
33 Patienten mit nichtresektablem LAPC wurden stereotaxiert. Um OAR-Toleranzwerte einzuhalten, wurde eine risikoadaptierte Dosisverordnung durchgeführt und zwischen den folgenden Dosiskonzepten gewählt: 42 Gy oder 45 Gy in 6 täglichen Fraktionen mit einer biologisch äquivalenten Dosis (BED) > 70 Gy10 oder 36 Gy in 6 Fraktionen (BED 57,6 Gy10). Die SBRT wurde mit volumenmodulierter Radiotherapie (VMAT) ohne Ausgleichsfilter („flattening filter-free“, FFF) durchgeführt. Vor jeder Behandlungseinheit erfolgte eine Image-Guidance mittels CBCT. Das Ansprechen sowie die Akut- und Spätnebenwirkungen wurden am Ende der Therapie sowie nach 3, 6 und 12 Monaten ausgewertet.
Ergebnisse
Die mediane Nachbeobachtungszeit betrug 18 Monate (5–34 Monate). Vor der SBRT erhielten 24 von 33 Patienten eine Induktionschemotherapie. Obwohl alle Patienten zuvor als inoperabel eingestuft wurden, wurden 6 von 33 (18 %) nach SBRT operiert. Alle operierten Patienten hatten eine BED > 70 Gy10 erhalten. Lokoregionale Kontrolle (LC) und Gesamtüberleben (OS) nach einem Jahr waren 81 % und 75 %. Insgesamt erlitten 12 Patienten (37 %) eine distante Progression. Kein Patient entwickelte eine akute oder späte Grad-3-Toxizität.
Schlussfolgerung
Die bildgeführte SBRT mit risikoadaptierter Dosisverordnung stellt eine effektive Behandlungsoption für LAPC-Patienten dar.




Similar content being viewed by others
Abbreviations
- BED:
-
Biologically effective dose
- CT:
-
Computed tomography
- CTV:
-
Clinical target volume
- CR:
-
Complete response
- Dx:
-
Dose per x cc
- GTV:
-
Gross tumor volume
- IMRT:
-
Intensity-modulated radiotherapy
- IGRT:
-
Image-guided radiotherapy
- LAPC:
-
Locally advanced pancreatic cancer
- LC:
-
Local control
- Linac:
-
Linear accelerator
- OS:
-
Overall survival
- OARs:
-
Organs at risk
- PD:
-
Progressive disease
- PERCIST:
-
PET Response Criteria in Solid Tumors
- PET:
-
Positron-emission tomography
- PR:
-
Partial response
- PTV:
-
Planning target volume
- RT:
-
Radiotherapy
- RECIST:
-
Response Evaluation Criteria in Solid Tumors
- SBRT:
-
Stereotactic body radiotherapy
- SD:
-
Stable disease
- Vx:
-
Volume receiving x Gy
References
Gillen S, Schuster T, Meyer Zum Büschenfelde C et al (2010) Preoperative/neoadjuvant therapy in pancreatic cancer: a systematic review and meta-analysis of response and resection percentages. Plos Med 7(4):e1000267
Andriulli A, Festa V, Botteri E et al (2012) Neoadjuvant/Preoperative gemcitabine for patients with localized pancreatic cancer: a meta-analysis of prospective studies. Ann Surg Oncol 19(5):1644–1662
De Bari B, Porta L, Mazzola R et al (2016) Hypofractionated radiotherapy in pancreatic cancer: lessons from the past in the era of stereotactic body radiation therapy. Crit Rev Oncol Hematol 103:49–61
Mahadevan A, Miksad R, Goldstein M et al (2011) Induction gemcitabine and stereotactic body radiotherapy for locally advanced nonmetastatic pancreas cancer. Int J Radiat Oncol Biol Phys 81(4):e615–22
Alongi F, Mazzola R, Ricchetti F et al (2015) Volumetric-modulated arc therapy with vaginal cuff simultaneous integrated boost as an alternative to brachytherapy in adjuvant irradiation for endometrial cancer: a prospective study. Anticancer Res 35(4):2149–2155
Ferrera G, Mortellaro G, Mannino M et al (2015) Moderate hypofractionation and simultaneous integrated boost by helical tomotherapy in prostate cancer: monoinstitutional report of acute tolerability assessment with different toxicity scales. Radiol Med 120(12):1170–1176
Mazzola R, Ferrera G, Alongi F et al (2015) Organ sparing and clinical outcome with step-and-shoot IMRT for head and neck cancer: a mono-institutional experience. Radiol Med 120(8):753–758
NCCN Clinical Practice Guidelines in Oncology (2017) Pancreatic adenocarcinoma. Version 2. http://www.nccn.org/professionals/physician_gls/PDF/pancreatic.pdf. Accessed 1 May 2017
Comito T, Cozzi L, Clerici E et al (2017) Can Stereotactic body radiation therapy be a viable and efficient therapeutic option for unresectable locally advanced pancreatic adenocarcinoma? Results of a phase 2 study. Technol Cancer Res Treat 16(3):295–301
Comito T, Cozzi L, Zerbi A et al (2017) Clinical results of stereotactic body radiotherapy (SBRT) in the treatment of isolated local recurrence of pancreatic cancer after R0 surgery: a retrospective study. Eur J Surg Oncol 43(4):735–742
Schwartz LH, Litière S, de Vries E et al (2016) RECIST 1.1-Update and clarification: From the RECIST committee. Eur J Cancer 62:132–137
Wahl RL, Jacene H, Kasamon Y et al (2009) From RECIST to PERCIST: Evolving considerations for PET response criteria in solid tumors. J Nucl Med 50(suppl 1):122S–150S
Expert Panel on Radiation Oncology-Gastrointestinal, Small W Jr, Hayes JP, Suh WW et al (2016) ACR appropriateness criteria® borderline and unresectable pancreas cancer. Oncology 30(7):619–24, 627, 632
Schellenberg D, Goodman KA, Lee F et al (2008) Gemcitabine chemotherapy and single-fraction stereotactic body radiotherapy for locally advanced pancreatic cancer. Int J Radiat Oncol Biol Phys 72(3):678–686
Koong AC, Le QT, Ho A et al (2004) Phase I study of stereotactic radiosurgery in patients with locally advanced pancreatic cancer. Int J Radiat Oncol Biol Phys 58(4):1017–1021
Koong AC, Christofferson E, Le QT et al (2005) Phase II study to assess the efficacy of conventionally fractionated radiotherapy followed by a stereotactic radiosurgery boost in patients with locally advanced pancreatic cancer. Int J Radiat Oncol Biol Phys 63(2):320–323
Shen ZT, Wu XH, Li B et al (2010) Preliminary efficacy of CyberKnife radiosurgery for locally advanced pancreatic cancer. Chin J Cancer 29(9):802–809
Herman JM, Chang DT, Goodman KA et al (2015) Phase 2 multi-institutional trial evaluating gemcitabine and stereotactic body radiotherapy for patients with locally advanced unresectable pancreatic adenocarcinoma. Cancer 121(7):1128–1137
Toesca DAS, Pollom EL et al (2017) Assessing local progression after stereotactic body radiation therapy for unresectable pancreatic adenocarcinoma: CT versus PET. Pract Radiat Oncol. https://doi.org/10.1016/j.prro.2016.09.002
Brunner TB, Nestle U, Adebahr S et al (2016) Simultaneous integrated protection: a new concept for high-precision radiation therapy. Strahlenther Onkol 192(12):886–894
Panje C, Andratschke N, Brunner TB et al (2016) Stereotactic body radiotherapy for renal cell cancer and pancreatic cancer: literature review and practice recommendations of the DEGRO Working Group on Stereotactic Radiotherapy. Strahlenther Onkol 192(12):875–885
Chang DT, Schellenberg D, Shen J et al (2009) Stereotactic radiotherapy for unresectable adenocarcinoma of the pancreas. Cancer 115:665–672
Goyal K, Einstein D, Ibarra RA et al (2012) Stereotactic body radiation therapy for nonresectable tumors of the pancreas. J Surg Res 174(2):319–325
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
R. Mazzola, S. Fersino, D. Aiello, F. Gregucci, U. Tebano, S. Corradini, G. Di Paola, M. Cirillo, L. Tondulli, G. Ruffo, R. Ruggieri, and F. Alongi declare that they have no competing interests.
Ethical standards
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Mazzola, R., Fersino, S., Aiello, D. et al. Linac-based stereotactic body radiation therapy for unresectable locally advanced pancreatic cancer: risk-adapted dose prescription and image-guided delivery. Strahlenther Onkol 194, 835–842 (2018). https://doi.org/10.1007/s00066-018-1306-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00066-018-1306-2