Abstract
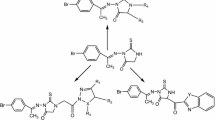
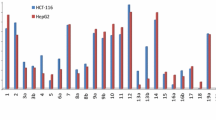
A novel series of hydroxy and non-hydroxy long chain substituted 1,3,4-oxadiazole moiety bearing 2-methyl-1H-benzimidazoles 15(a–d) have been synthesized from cyclization reaction of 2-(2-methyl-1H-benzimidazol-1-yl) acetohydrazide (13) with different unsaturated hydroxy and non-hydroxy fatty esters in the presence of phosphorus oxychloride and product obtained in appreciable yield. Compound (13) was synthesized from the corresponding ethyl (2-methyl-1H-benzimidazol-1-yl) acetate (12) and 2-methyl-1H-benzimidazole (11). All the synthesized compounds were characterized with the help of IR, 1H NMR, 13C NMR, and mass spectral studies. These compounds were tested for cytotoxic (or antiproliferative) activity against normal human blood cells (PBMCs), Hep3B (human hepatocellular carcinoma), MCF 7 (human breast adenocarcinoma), and HeLa (human cervical carcinoma) cell lines.


Similar content being viewed by others
Abbreviations
- SAR studies:
-
Structure activity relationship studies
References
Ansari KF, Lal C (2009a) Synthesis, physiochemical properties and antimicrobial activity of some new benzimidazole derivatives. Eur J Med Chem 44:4028–4033
Ansari KF, Lal C (2009b) Synthesis and evaluation of some new benzimidazole derivatives as potential antimicrobial agents. Eur J Med Chem 44:2294–2299
Bauer AW, Kirby WMM, Sherris JC, Truch M (1966) Antibiotic susceptibility testing by a standardized single disk method. Am J Clin Pathol 45:493–496
Bhat KS, Karthikeyan MS, Holla BS, Shetty NS (2004) Synthesis of some new fluorine containing 1,3,4-oxadiazole derivatives as potential antibacterial and anticancer agents. Indian J Chem 43B:1765–1769
Bin H, Prabha MV, Sharon FC, Christine NH, Peter HA, Albert MC, Ahmad AA, David SR, Jame PS (1998) Synthesis of 2-piperidinecarboxylic acid derivatives as potential anticonvulsants. Eur J Med Chem 33:23–31
Boiani M, Gonzalez M (2005) Imidazole and benzimidazole derivatives as chemotherapeutic agents. Mini-Rev Med Chem 5:409–424
Boström J, Hogner A, Llinàs A, Wellner E, Plowright AT (2012) Oxadiazoles in medicinal chemistry. J Med Chem 55:1817–1830
Chakravarty PK, Strelitz RA, Chen TB, Chang RSL, Lotti VJ, Zingaro GJ, Schorn TW, Kivlighn SD, Siegl PKS, Patchett AA, Greenlee WJ (1994) Quinazolinione biphenyl acylsulphonamides: a potent new class of angiotensin-II receptor antagonists. Bioorg Med Chem Lett 4:75–80
Chen Q, Zhu XL, Jiang LL, Liu ZM, Yang GF (2008) Synthesis, antifungal activity and CoMFA analysis of novel 1,2,4-triazolo[1,5-a]pyrimidine derivatives. Eur J Med Chem 43:595–603
Goodfriend TL, Elliott ME, Catt KJ (1996) Angiotensin receptors and their antagonists. N Engl J Med 334:1649–1654
Gunstone FD (1954) Fatty acids. Part II. The nature of the oxygenated acid present in Vernonia Anthelmintica (Willd.) seed oil. J Chem Soc. doi:10.1039/JR9540001611
Guo XZ, Shi L, Wang R, Liu XX, Li BG, Lu XX (2008) Synthesis and biological activities of novel nonpeptide angiotensin II receptor antagonists based on benzimidazole derivatives bearing a heterocyclic ring. Bioorg Med Chem 16:10301–10310
Hazarika J, Kataky JC (1998) Synthesis and biological screening of some 2,5-disubstituted-1,3,4-oxadiazoles. Indian J Heterocycl Chem 8:83–84
Holla BS, Prasanna CS, Poojary B, Rao KS, Shridhara K, Bhat UG (2004) Synthesis and insecticidal activity of some 1,3,4-oxadiazoles derived from 2-chloro-pyridine-5-acetic acid. Indian J Chem 43B:864–869
Hori A, Imaeda Y, Kubo K, Kusaka M (2002) Novel benzimidazole derivatives selectively inhibit endothelial cell growth and suppress angiogenesis in vitro and in vivo. Cancer Lett 183:53–60
Husain A, Ajmal M (2009) Synthesis of novel 1,3,4-oxadiazole derivatives and their biological properties. Acta Pharm 59:223–233
Jha KK, Kumar Y, Shaharyar M, Singhal S (2010) Microwave assisted synthesis, characterization & biological evaluation of benzimidazole substituted 1,3,4-oxadiazole. Int J ChemTech Res 2:716–727
Jiang LL, Tan Y, Zhu XL, Wang ZF, Zuo Y, Chen Q, Xi Z, Yang GF (2010) Design, synthesis, and 3D-QSAR analysis of novel 1,3,4-oxadiazol-2(3H)-ones as protoporphyrinogen oxidase inhibitors. J Agric Food Chem 58:2643–2651
Khan MSY, Akhtar M (2003) Synthesis of some new 2,5-disubstituted 1,3,4-oxadiazole derivatives and their biological activity. Indian J Chem 42B:900–904
Khan MSY, Chawla G, Mueed MA (2004) Synthesis and biological activity of some isoniazid based 1,3,4-oxadiazole derivatives. Indian J Chem 43B:1302–1305
Kiselyov AS, Semenova MN, Chernyshova NB, Leitao A, Samet AV, Kislyi KA, Raihstat MM, Oprea T, Lemcke H, Lantow M, Weiss DG, Ikizalp NN, Kuznetsov SA, Semenov VV (2010) Novel derivatives of 1, 3, 4-oxadiazoles are potent mitostatic agents featuring strong microtubule depolymerizing activity in the sea urchin embryo and cell culture assays. Eur J Med Chem 45:1683–1697
Kumar D, Jacob MR, Reynolds MB, Kerwin SM (2002) Synthesis and evaluation of anticancer benzoxazoles and benzimidazoles related to UK-1. Bioorg Med Chem 10:3997–4004
Kumar D, Patel G, Vijayakrishnan L, Dastidar SG, Ray A (2012) Design and synthesis of 3,5-disubstituted-1,2,4-oxadiazoles as potent inhibitors of phosphodiesterase4b2. Chem Biol Drug Des 79:810–818
Lam KW, Syahida A, Haq ZU, Rahman MBA, Lajis NH (2010) Synthesis and biological activity of oxadiazole and triazolothiadiazole derivatives as tyrosinase inhibitors. Bioorg Med Chem Lett 20:3755–3759
Li Z, Wang X (2003) Syntheses and biological activities of 2-(3-methylbenzoylamino)-5-aryloxymethyl-1,3,4-oxadiazoles. Indian J Chem 42B:941–944
Moglilaiah K, Vidya K (2006) Synthesis and antibacterial activity of 1,3,4-oxadiazolyl-1,8-naphthyridines. Indian J Chem 45B:1905–1906
Nagaraj Chaluvaraju KC, Niranjan MS, Kiran S (2011) 1,3,4-Oxadiazole: a potent drug candidate with various pharmacological activities. Int J Pharm Pharm Sci 3:9–16
Oliveira CS, Lira BF, Filho JMB, Lorenzo JGF, Filho PFA (2012) Synthetic approaches and pharmacological activity of 1,3,4-oxadiazoles: a review of the literature from 2000–2012. Molecules 17:10192–10231
Patrao P, Khader AMA, Kalluraya B, Vinayachandra (2013) Synthesis of new 5-naphthyl substituted 1,3,4-oxadiazole derivatives and their antioxidant activity. Der Pharm Chem 5:24–32
Preston PN (1974) Synthesis, reactions, and spectroscopic properties of benzimidazoles. Chem Rev 74:279–314
Preston PN (Ed.) (1981) Benzimidazoles and congeneric tricyclic compounds. Interscience Publication, John Wiley & Sons, New York
Rashid M, Husain A, Mishra R, Ahmed N, Ahmed NS (2012) 1-(1H-Benzimidazol-2-yl)-3-[5-(trichloromethyl)-1,3,4-oxadiazol-2-yl]propan-1-one. doi:10.3390/M766
Rathee PS, Dhankar R, Bhardwaj S, Gupta M, Kumar R (2011) Synthesis and antimicrobial studies of novel benzimidazole derivatives. J Appl Pharma Sci 01:127–130
Ravindra KC, Vagdevi HM, Vaidya VP, Padmashal B (2006) Synthesis, antimicrobial and antiinflammatory activities of 1,3,4-oxadiazoles linked to naphtho[2,1-b]furan. Indian J Chem 45B:2506–2511
Sharma S, Gangal S, Rauf A (2009) An efficient, one-pot synthesis of novel 3,5- disubstituted-1,2,4-oxadiazoles from long-chain carboxylic acid derivatives. Acta Chim Sol 56:369–372
Somani RR, Shirodkar PY (2009) Oxadiazoles: a biologically important heterocycle. Der Pharm Chem 1:130–140
Vasoya SL, Patel MR, Dobaria SV, Joshi HS (2005) Facile synthesis of some new azetidinones and acetyl oxadiazoles bearing benzo[b]thiophene nucleus as a potent biological active agent. Indian J Chem 44B:405–409
Xia G, Youa X, Liua L, Liu H, Wang J, Shi Y, Li P, Xiong B, Liu X, Shena J (2013) Design, synthesis and SAR of piperidyl-oxadiazoles as 11β-hydroxysteroid dehydrogenase 1 inhibitors. Eur J Med Chem 62:1–10
Yeap SK, Alitheen NB, Ali AM, Omar AR, Raha AR, Suraini AA, Muhajir AH (2007) Effect of Rhaphidophora korthalsii methanol extract on human peripheral blood mononuclear cell (PBMC) proliferation and cytolytic activity toward HepG2. J Ethnopharmacol 114:406–411
Zhang MZ, Mulholland N, Beattie D, Irwin D, Gu YC, Chen Q, Yang GF, Clough J (2013) Synthesis and antifungal activity of 3-(1,3,4-oxadiazol-5-yl)-indoles and 3-(1,3,4- oxadiazol-5-yl)methyl-indoles. Eur J Med Chem 63:22–32
Zuo Y, Yang SG, Jiang LL, Hao GF, Wang ZF, Wu QY, Xi Z, Yang GF (2012) Quantitative structure-activity relationships of 1,3,4-thiadiazol-2(3H)-ones and 1,3,4-oxadiazol-2(3H)-ones as human protoporphyrinogen oxidase inhibitors. Bioorg Med Chem 20:296–304
Acknowledgments
The authors are thankful to the Department of Chemistry, Aligarh Muslim University, Aligarh for providing the necessary research facility. Two of the authors namely (HV)1 and (AA)1 are thankful to DST and CSIR New Delhi for the award of SRF. We are also grateful to the SAIF, Punjab University, Chandigarh for providing spectral technique. In part the research is also supported by UGC-SAP (DRS-I).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Varshney, H., Ahmad, A., Rauf, A. et al. Multistep synthesis of 1-[{(5-alkenyl/hydroxyalkenylsubstituted)-1,3,4-oxadiazol-2-yl}-methyl]-2-methyl-1H-benzimidazole series and in vitro anticancer screening, SAR studies. Med Chem Res 24, 944–953 (2015). https://doi.org/10.1007/s00044-014-1162-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-014-1162-2