Abstract
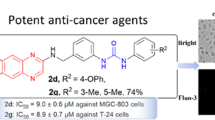
Eleven diarylurea derivatives (1a–1k) bearing N-methylpiperazinyl moiety were synthesized by the reaction of isocyanate with arylamine. The structures of 1a–1k were characterized by 1H-NMR and MS. The cytotoxic activities of 1a–1k were evaluated via MTT method against human lung adenocarcinoma epithelial cell line A549 and human prostate carcinoma cell line PC3. Compounds 1d and 1k displayed potent cytotoxic activity. The results suggested that the activities are considerably related to the substituent group at another phenyl ring.



Similar content being viewed by others
References
Anandan SK, Gless RD (2010) Exploration of secondary and tertiary pharmacophores in unsymmetrical N, N-diaryl urea inhibitors of soluble epoxide hydrolase. Bioorg Med Chem Lett 20:2740–2744
Anandan SK, Webb HK, Chen D, Wang YX, Aavula BR, Cases S, Cheng Y, Do ZN, Mehra U, Tran V, Vincelette J, Waszczuk J, White K, Kenneth RW, Zhang LN, Paul DJ, Bruce DH, Dinesh VP, Randall WD, Euan M, Sabry J, Gless R (2011) 1-(1-Acetyl-piperidin-4-yl)-3-adamantan-1-yl-urea (AR9281) as a potent, selective, and orally available soluble epoxide hydrolase inhibitor with efficacy in rodent models of hypertension and dysglycemia. Bioorg Med Chem Lett 21:983–988
Beaver DJ, Roman DP, Stoffel PJ (1957) The preparation and bacteriostatic activity of substituted ureas. J Am Chem Soc 79:1236–1245
Caputo R, Calabro ML, Micale N, Schimmer AD, Ali M, Zappala M, Grasso S, Med Chem Res (2012) Synthesis of benzothiazole derivatives and their biological evaluation as anticancer agents. Med Chem Res 21:2644–2651
Cheng Y, Paz K (2008) Tandutinib, an oral, small molecule inhibitor of FLT3 for the treatment of AML andother cancer indications. Idrugs 11:46–56
Dehnhardt CM, Venkatesan AM, Delos SE (2009) Lead optimization of N-3-Substituted 7-morpholinotriazolopyrimidines as dual phosphoinositide 3-kinase/mammalian target of rapamycin inhibitors: discovery of PKI-402. J Med Chem 53:798–810
Denoyelle S, Chen T, Chen L, Wang YB, Klosi E, José AH, Bertal HA, Michael C (2012) In vitro inhibition of translation initiation by N, N-diarylureas-potential anti-cancer agents. Bioorg Med Chem Lett 22:402–409
Hawes EM, Shetty HU, Cooper JK, Rauw G, McKay G, Midha KK (1984) Radioimmunoassay for psychotropic drugs III: synthesis and properties of haptens for trifluoperazine and fluphenazine. J Pharm Sci 73:247–250
Hayakawa I, Atarashi S, Yokohama S, Imamura M, Sakano K, Furukawa M (1986) Synthesis and antibacterial activities of optically active ofloxacin. Antimicrob Agents Chemother 29:163–164
Ji ZQ, Asma AA, Daniel HA, Jennifer JB, Peter FB, George AC, Gilbert D, Keith BG, Guo J, Christopher MH, Li JL, Patrick AM (2008) 3-Amino-benzo[d]isoxazoles as novel multitargeted inhibitors of receptor tyrosine kinases. J Med Chem 51:1231–1241
Lion CJ, Matthews CS, Wells G, Bradshaw TD, Stevens MFG, Westwell AD (2006) Antitumour properties of fluorinated benzothiazole-substituted hydroxycyclohexa-2, 5-dienones. Bioorg Med Chem Lett 16:5005–5008
Liu L, Cao YC, Chen C, Zhang XM, McNabola A, Wilkie D, Wilhelm S, Lynch M, Carter C (2006) Sorafenib Blocks the RAF/MEK/ERK Pathway, Inhibits Tumor Angiogenesis, and Induces Tumor Cell Apoptosis in Hepatocellular Carcinoma Model PLC/PRF/5. Cancer Res 66:11851–11858
Madapa S, Tusi Z, Mishra A, Srivastava K, Pandey SK, Tripathi R, Puri SK, Batra S (2009) Search for new pharmacophores for antimalarial activity. Part II: synthesis and antimalarial activity of new 6-ureido-4-anilinoquinazolines. Bioorg Med Chem 17:222–234
Marvania B, Lee PC, Chaniyara R, Dong HJ, Suman S, Kakadiya R, Chou TC, Lee TC, Shah A, Su TL (2011) Design, synthesis and antitumor evaluation of phenyl N-mustard-quinazoline conjugates. Bioorg Med Chem 19:1987–1998
Ogita H, Isobe Y, Takaku H, Sekine R, Goto Y, Misawa S, Hayashi H (2002) Synthesis and structure–activity relationship of diarylamide derivatives as selective inhibitors of the proliferation of human endothelial cells. Bioorg Med Chem 10:3473–3480
Pacchiano F, Carta F, Vullo D (2011) Inhibition of β-carbonic anhydrases with ureido-substituted benzenesulfonamides. Bioorg Med Chem Lett 21:102–105
Pan WT, Miao HQ, Xu YJ, Navarro EC, Tonra JR, Erik C, Armin L, Paul K, Alexander SK, Wai CW, Liu H (2006) 1-[4-(1H-Benzoimidazol-2-yl)-phenyl]-3-[4-(1H-benzoimidazol-2-yl)-phenyl]urea derivatives as small molecule heparanase inhibitors. Bioorg Med Chem Lett 16:409–412
Philip MG, Bernard M (1971) Morphogenesis derivatives on vaccinia virus and other rifamycin similar effect of rifampin. J Virol 8:225–231
Receveur JM, Emelie B, Trond U, Paul BL, Pia K (2004) 4-Acylamino- and 4-ureidobenzamides as melanin-concentrating hormone (MCH) receptor 1 antagonists. Bioorg Med Chem Lett 14:5075–5080
Sonmez F, Sevmezler S, Atahan A, Ceylan M, Demir D, Gencer N, Arslan O, Kucukislamoglu M (2011) Evaluation of new chalcone derivatives as polyphenol oxidase inhibitors. Bioorg Med Chem Lett 21:7479–7482
Venkatesan AM, Chen Z, Santos OD (2010a) PKI-179: an orally efficacious dual phosphatidylinositol-3-kinase (PI3 K)/mammalian target of rapamycin (mTOR) inhibitor. Bioorg Med Chem Lett 20:5869–5873
Venkatesan AM, Dehnhardt CM, Delos Santos E (2010b) Bis(morpholino-1,3,5-triazine) derivatives: potent adenosine 5′-triphosphate competitive phosphatidylinositol-3-kinase/mammalian target of rapamycin inhibitors: discovery of compound 26 (PKI-587), a highly efficacious dual inhibitor. J Med Chem 53:2636–2645
Witherington J, Blaney EL, Bordas V, Elliott RL, Gaiba A, Garton N, Green PM, Naylor A, Smith DG, Spalding DJ, Takl AK, Ward RW (2006) Pyridone derivatives as potent, orally bioavailable VLA-4 integrin antagonists. Bioorg Med Chem Lett 16:5538–5541
Yoshida M, Hayakawa I, Hayashi N, Agatsuma T, Oda Y, Tanzawa F, Iwasaki S, Koyama K, Furukawa H, Kurakatad S, Suganob Y (2005) Synthesis and biological evaluation of benzothiazole derivatives as potent antitumor agents. Bioorg Med Chem Lett 15:3328–3332
Yu B, Tang LD, Li YL, Song SH, Ji XL, Lin MS, Wu CF (2012) Design, synthesis and antitumor activity of 4-aminoquinazoline derivatives targeting VEGFR-2 tyrosine kinase. Bioorg Med Chem Lett 22:110–114
Acknowledgments
Financial support from The National Natural Science Foundation of China (Grant No. 21072156) is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Xuan, W., Ding, W., Hui, Hx. et al. Synthesis and cytotoxic activity of diaryl urea derivatives with a 4-methylpiperazinylcarbonyl moiety. Med Chem Res 22, 3857–3862 (2013). https://doi.org/10.1007/s00044-012-0398-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-012-0398-y