Abstract
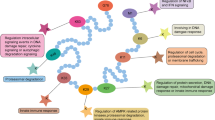
The 26S proteasome is the multi-protein protease that recognizes and degrades ubiquitinylated substrates targeted for destruction by the ubiquitin pathway. In addition to the well-documented subunit organization of the 26S holoenzyme, it is clear that a number of other proteins transiently associate with the 26S complex. These transiently associated proteins confer a number of different roles such as substrate presentation, cleavage of the multi-ubiquitin chain from the protein substrate and turnover of misfolded proteins. Such activities are essential for the 26S proteasome to efficiently fulfill its intracellular function in protein degradation.
Similar content being viewed by others
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hartmann-Petersen, R., Gordon, C. Ubiquitin-proteasome system. CMLS, Cell. Mol. Life Sci. 61, 1589–1595 (2004). https://doi.org/10.1007/s00018-004-4132-x
Issue Date:
DOI: https://doi.org/10.1007/s00018-004-4132-x