Abstract
Introduction
Patients with diabetes mellitus and end-stage renal disease are at a high risk of developing coronary, cerebrovascular, and peripheral vascular diseases. This study aimed to characterize hypoglycemia and blood glucose fluctuations associated with maintenance hemodialysis in older adult patients with diabetes mellitus and end-stage renal disease using a continuous glucose monitoring system.
Methods
Seven patients were enrolled in this study, and 13 pairs of continuous glucose monitoring system data were collected. Each pair included data of 1 dialysis-on day and 1 dialysis-off day. Information on basic patient characteristics, including age, diabetes mellitus duration, hemodialysis duration, and proportions of hemoglobin A1c and glycated albumin, were collected. Differences in blood glucose fluctuation were compared between dialysis-on days and dialysis-off days.
Results
The mean blood glucose on dialysis-on days (6.96 ± 2.57 mmol/L) was significantly lower than that on dialysis-off days (7.68 ± 2.31 mmol/L; P < 0.05). In contrast, the following parameters had significantly higher values (all P < 0.05) on dialysis-on days compared to dialysis-off days: large amplitude of glycemic excursion level (5.82 ± 2.86 mmol/L versus 4.21 ± 1.71 mmol/L), large amplitude of glycemic excursion level from 8 a.m. to 2 p.m. (3.6 ± 1.74 mmol/L versus 2.8 ± 1.33 mmol/L), mean amplitude of glycemic excursion level (4.78 ± 1.68 mmol/L versus 3.89 ± 1.67 mmol/L), mean amplitude of glycemic excursion level from 8 a.m. to 2 p.m. (4.01 ± 1.03 mmol/L versus 3.12. ± 0.97 mmol/L), standard deviation of blood glucose (1.55 ± 0.89 mmol/L versus 1.03 ± 0.4 mmol/L), and time below a target glucose range of less than 3.9 mmol/L (8.27% versus 4.25%).
Conclusion
Fluctuations in blood glucose levels were larger on dialysis-on days, particularly from the start of hemodialysis to 2 h post-hemodialysis, than on dialysis-off days. Hypoglycemia, as indicated by the time below a target glucose range of less than 3.9 mmol/L, occurred more frequently on dialysis-on days than on dialysis-off days.
Graphical Abstract

Similar content being viewed by others
Avoid common mistakes on your manuscript.
Why carry out this study? |
While it is well established that dialysis can affect blood glucose fluctuations, few studies have evaluated this relationship in older adult patients with diabetes mellitus (DM) and end-stage renal disease (ESRD) on maintenance hemodialysis |
This study characterized hypoglycemia and blood glucose fluctuations in older adult patients with DM and ESRD on maintenance hemodialysis, with the aim of highlighting the need for clinicians to closely monitor blood glucose status in patients undergoing dialysis |
What was learned from the study? |
We observed that fluctuations in blood glucose levels were larger on dialysis-on days, particularly from the start of hemodialysis to 2 h post-hemodialysis, than on dialysis-off days; hypoglycemia occurred more frequently on dialysis-on days than on dialysis-off days |
Our results emphasize the importance of monitoring blood glucose in older adult patients with DM and ESRD on maintenance hemodialysis and provide an evidence base that will facilitate the development of future intervention studies |
Digital Features
This article is published with digital features, including a graphical abstract, to facilitate understanding of the article. To view digital features for this article go to https://doi.org/10.6084/m9.figshare.20014982.
Introduction
Patients with diabetes mellitus (DM) and end-stage renal disease (ESRD) are at a high risk of developing coronary, cerebrovascular, and peripheral vascular diseases. These complications are the leading causes of death among patients with ESRD [1, 2]. Hypoglycemia, hyperglycemia, and blood glucose fluctuations can accelerate the occurrence and development of these complications. Patients with DM and ESRD are not only insulin resistant but are also prone to hypoglycemia due to impaired renal gluconeogenesis, malnutrition, altered insulin pharmacokinetics, and hypoglycemic agent therapy [3, 4].
While the risk of hypoglycemia can be reduced by using a glucose-added dialysis fluid [5, 6], the prediction of glycemic patterns remains challenging. Therefore, it is difficult to control blood glucose levels without increasing the risk of hypoglycemia and large variations in blood glucose levels. The degree of blood glucose fluctuations in patients with DM and ESRD on maintenance hemodialysis is controversial. Some studies have reported fluctuations during dialysis-on days [7,8,9,10,11,12,13], while other studies have not observed significant fluctuations [14, 15].
A continuous glucose monitoring system (CGMS) enables the direct tracking of short-term (intra- and interday) glucose variability and hyper- and hypoglycemia [16, 17]. In 2019, the Advanced Technologies & Treatments for Diabetes Conference [18] released an updated consensus statement that aimed to refine core metrics for the assessment of glycemic control. It included three key CGMS-derived measurements: (i) the percentage of readings and time per day within a target glucose range of 3.9–10 mmol/L (time in range [TIR]3.9–10); (ii) the percentage of readings and time above a target glucose range of 13.8 mmol/L (time above range [TAR]>13.8); and (iii) the percentage of readings and time below a target glucose range of 3.9 mmol/L (time below range [TBR]<3.9). The 2020 Kidney Disease Improving Global Outcomes (KDIGO) guidelines state that the CGMS may be advantageous for the self-management of diabetes in individuals with chronic kidney disease [19]. As the CGMS measures blood glucose levels 288 times daily for up to 7 days, it provides a comprehensive assessment of blood glucose fluctuations on both dialysis-on days and dialysis-off days.
There is currently a lack of data on blood glucose fluctuations in advanced aging patients with DM and ESRD on maintenance hemodialysis. Therefore, in this study, we used the CGMS to compare glycemic variability and hypoglycemia incidence between dialysis-on and dialysis-off days in older adult patients with DM and ESRD on maintenance hemodialysis.
Methods
This study included patients with type 2 DM and ESRD who were hospitalized and undergoing maintenance hemodialysis in the Geriatric Nephrology Department of the People’s Liberation Army General Hospital (PLAGH). Patients were included if they were at least 65 years of age and underwent stable and regular hemodialysis (three times a week, hemodialysis duration of at least 3 months). The exclusion criteria comprised hormone administration in the previous 6 months, acute complications of diabetes, and acute infection. Data on the following patient demographic and clinical characteristics were collected: age, DM duration, hemodialysis duration, and proportions of hemoglobin A1c (HbA1c) and glycated albumin. The CGMS (iPro 2, Medtronic Inc. Minneapolis, Minnesota, USA) was applied on the afternoon before the day of dialysis. It provided 288 blood glucose level measurements daily and remained in place for 1–2 complete dialysis-on days and dialysis-off days. Trained nurses calibrated the CGMS by measuring capillary blood glucose four times a day (before three meals and bedtime) and recorded the time and number of meals, snacks, medications, and exercise. The three meals were provided at approximately 7:15 a.m., 11:15 a.m., and 5:15 p.m.; however, lunch on a dialysis day was provided at approximately 12:30 p.m. The standard amount of calories provided was 25–30 kcal/kg. Carbohydrates accounted for 55–60% of the calories and were divided among the three meals according to the following ratios: breakfast (1/5), lunch (2/5), and dinner (2/5). During dialysis (from 10 to 11:30 a.m.), extra meals (e.g., two pieces of chocolate and 150 mL of nutrient solution) were consumed according to the patient’s habits and preferences. All data were downloaded using CARELINK PRO (Medtronic Inc. Minneapolis, Minnesota, USA). Hypoglycemia referred to blood glucose concentrations below 2.8 mmol/L. Individuals with diabetes were diagnosed with hypoglycemia when the blood glucose level was below 3.9 mmol/L.
The dialysate was a bicarbonate concentrate that contained glucose (5 mmol/L), as well as sodium (138 mmol/L), potassium (2 mmol/L), calcium (1.5 mmol/L), magnesium (0.5 mmol/L), chloride (109 mmol/L), acetate (3 mmol/L), and bicarbonate (32 mmol/L) ions. The dialysis mode was hemodialysis. Each hemodialysis session started at 8 a.m. and lasted for 4 h.
The study protocol was approved by the Ethics Committee of the PLAGH (S2021-424-01). All patients provided written informed consent. Double encryption was used, and all data were stored on a secure computer that was not connected to a network to protect patient data.
Calculation of Glucose Profiles
A single dialysis-on day extended from 8 a.m. on the day of dialysis to 8 a.m. on the following day; the subsequent 24 h were defined as a dialysis-off day. The following variables were calculated from the CGMS for each patient: (1) mean blood glucose (MBG) level (the mean of 288 measured values during the 24-h CGMS monitoring period; (2) standard deviation of blood glucose (SDBG) (standard deviation values during the 24-h CGMS or observation period [normal reference value of less than 1.4 mmol/L]); (3) large amplitude of glycemic excursion (LAGE) (the difference between maximum and minimum blood glucose levels during blood glucose monitoring [normal reference value of less than 5.7 mmol/L]); (4) mean amplitude of glycemic excursion (MAGE) (mean value of effective fluctuation blood glucose fluctuation range, effective fluctuation means amplitude is greater than one standard deviation [normal reference value of less than 3.4 mmol/L]); (5) TBR<3.9; (6) TIR3.9–10; and (7) TAR>13.8.
Statistical Analysis
All statistical analyses were performed using IBM SPSS Statistics 23.0 (IBM Corporation, Armonk, NY, USA). Tests for normality were conducted using the Shapiro–Wilk test. All normally distributed data are expressed as mean ± standard deviation, and non-normally distributed data are expressed as median (range). Comparisons of normally distributed variables between dialysis-on and dialysis-off days were made with the t test; the Wilcoxon test was used for non-normally distributed variables. The level of statistical significance was set at P < 0.05.
Results
Patient Demographic and Clinical Characteristics
All patients had type 2 DM. The mean patient age was 82.58 ± 8.97 years. The mean duration of DM was 13.29 ± 11.34 years, and the mean duration of hemodialysis was 2.97 ± 2.16 years. The proportions of HbA1c and glycated albumin were 6.23 ± 0.89% and 19 ± 5.58%, respectively (Table 1).
Blood Glucose Levels on Dialysis-on Days and Dialysis-off Days
The MBG level was significantly lower on dialysis-on days (6.96 ± 2.57 mmol/L) than on dialysis-off days (7.68 ± 2.31 mmol/L, P = 0.03). LAGE (5.82 ± 2.86 mmol/L versus 4.21 ± 1.71 mmol/L, P = 0.04), MAGE (4.78 ± 1.68 mmol/L versus 3.89 ± 1.67 mmol/L, P = 0.04), SDBG (1.55 ± 0.89 mmol/L versus 1.03 ± 0.4 mmol/L, P = 0.04), and the percentage of hypoglycemia time (8.27% versus 4.25%, P = 0.02) were significantly greater on dialysis-on days than on dialysis-off days (all P < 0.05) (Table 2). Blood glucose fluctuations for each patient were the same as the overall trend (Table 3).
Blood Glucose Drift, LAGE, and MAGE of Different Periods on Dialysis-on Days and Dialysis-off Days
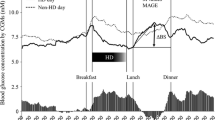
The blood glucose curves on dialysis-on and dialysis-off days differed from the beginning of hemodialysis to 2 h post-hemodialysis (8 a.m. to 2 p.m.); an inverted U-shape was observed. In other periods, the blood glucose curves were not different (Fig. 1).
LAGE from the beginning of hemodialysis to 2 h post-hemodialysis (8 a.m. to 2 p.m.) was significantly greater on dialysis-on days (3.6 ± 1.74 mmol/L) compared to that on dialysis-off days (2.8 ± 1.33 mmol/L; P < 0.05). In the other two periods, the difference was not significant (P > 0.05). MAGE from the beginning of hemodialysis to 2 h post-hemodialysis (8 a.m. to 2 p.m.) was significantly greater on dialysis-on days (4.01 ± 1.03 mmol/L) compared to that on dialysis-off days (3.12 ± 0.97 mmol/L; P < 0.05). In the other two periods, the difference was not significant (P > 0.05) (Table 4).
MBG Level from 8 a.m. to 2 p.m. on Dialysis-on Days and Dialysis-off Days
The MBG level did not change significantly from 1 h pre-hemodialysis to 1–3 h post-hemodialysis between dialysis-on days and dialysis-off days (P > 0.05); the changes were significant from the 2nd hour of hemodialysis to 2 h post-hemodialysis (P < 0.05). The most significant changes were observed from the 4th hour of hemodialysis to 1 h post-hemodialysis (Fig. 2).
Hypoglycemia
Hypoglycemia occurred 12 times on dialysis-on days and three times on dialysis-off days. None of the hypoglycemic episodes were associated with any symptoms (Table 5).
Discussion
According to data from the 10th edition of the International Diabetes Federation Diabetes Atlas, 537 million adults lived with diabetes worldwide in 2021, and the age-adjusted comparative prevalence of diabetes is estimated to be 9.8%. The proportion of diabetes-related deaths is as high as 32.6% [20]. Hypoglycemia and blood glucose fluctuation can result in serious complications, such as diabetes-related macrovascular and microvascular disorders. Acute hypoglycemia can result in coma and subsequent mortality. Patients with both diabetes and ESRD are at a high risk of developing acute cardiovascular and cerebrovascular events; therefore, good glycemic control is necessary to improve their long-term prognosis [21] and quality of life. Previous studies identified CGMS as an appropriate and reliable tool for the detection of glycemic variations and hypoglycemic episodes in individuals with diabetes, particularly on the day of hemodialysis [22].
A previous study showed that HbA1c was weakly correlated with MBG level in patients with type 2 DM undergoing hemodialysis compared to those not undergoing hemodialysis [15]. The continuous monitoring of blood glucose facilitates the detection of hyperglycemia and hypoglycemia, especially in patients with diabetes and advanced chronic kidney disease—such monitoring was recommended by the 2020 KDIGO guideline—because of the drawbacks of currently established glycemic biomarkers, such as HbA1c [19]. In the present study, we determined the influence of hemodialysis on blood glucose by using the CGMS to evaluate blood glucose parameters and compare them between dialysis-on days and dialysis-off days in older adult patients. We found that fluctuations in blood glucose levels were larger on dialysis-on days, particularly from the start of hemodialysis to 2 h post-hemodialysis, than on dialysis-off days. Hypoglycemia, as indicated by the time below a target glucose range of less than 3.9 mmol/L, occurred more frequently on dialysis-on days than on dialysis-off days.
Blood glucose fluctuations contribute to the development of diabetes complications, such as cardiovascular and cerebrovascular events, and have been shown to cause more serious harm than continuous hyperglycemia [23,24,25]. Our results indicated that MBG level was generally lower on dialysis-on days than on dialysis-off days. Furthermore, coefficient of variation (CV), LAGE, and SDBG were higher on dialysis-on days than on dialysis-off days. This indicates that blood glucose levels tend to fluctuate more substantially on dialysis-on days. While prior reports have also documented similar trends for blood glucose fluctuations, not all studies have found consistent differences in MBG levels between dialysis-on days and dialysis-off days [12, 14, 15, 17]. For example, Jung et al. [14] observed fluctuations in blood glucose levels using the CGMS in nine patients with type 2 DM undergoing maintenance hemodialysis; however, there was no difference in the MBG level between dialysis-on days and dialysis-off days. The results of their subgroup analysis showed that glucose levels decreased significantly with hemodialysis initiation in patients who were maintained on antidiabetic agents on dialysis days; this is consistent with our results. The lack of a difference in the MBG level may have been related to the reduction in hypoglycemic treatment in some patients on the day of dialysis. Divani et al. [17] did not find a difference in the mean 24-h CGM-derived glucose level between dialysis-on and dialysis-off days in 37 individuals with diabetes; however, the 24-h CV of glucose readings and the TBR<70 were significantly higher during dialysis-on days than dialysis-off days. Mirani et al. [12] assessed 12 patients with DM on hemodialysis for 2 days (1 hemodialysis day and the following non-hemodialysis day) and found that the MBG level and glycemic variability were significantly higher on the day of hemodialysis. The results of this study are consistent with our findings. Riveline et al. [15] compared glucose levels in 19 hemodialyzed and 39 non-hemodialyzed patients with type 2 DM in a double-center study; a CGMS was used for 4-day monitoring (2 days with and without dialysis). While the mean glucose level was not significantly different between dialysis-on and dialysis-off days, it was remarkably lower in the first 3 h of dialysis. In the present study, we also observed a blood glucose drift from the beginning of hemodialysis to 2 h post-hemodialysis. This finding emphasizes the importance of monitoring changes in blood glucose not only during but also after dialysis, especially in older adult patients.
Hypoglycemia is a common and serious complication of diabetes. The clinical manifestations of hypoglycemia in older adult patients may vary due to poor physical fitness and self-consciousness. Quality of life may be severely impacted, and impaired consciousness and coma are associated with a high rate of disability and fatality [26]. Increased susceptibility to hypoglycemia in individuals with diabetes and ESRD is attributed to impairment of renal gluconeogenesis and insulin clearance. Dialysis also has a large influence on blood glucose levels; indeed, our results demonstrated that the frequency and duration of hypoglycemia were greater on dialysis-on days than on dialysis-off days. In individuals with diabetes, hypoglycemia is defined by blood glucose levels of less than 3.9 mmol/L [27]. Thus, we used TBR<3.9 as a hypoglycemia assessment index; values were higher on dialysis-on days than on dialysis-off days. We observed two periods during which blood glucose levels were relatively low, and most hypoglycemia events occurred. The first was a blood glucose drift from 10 p.m. to 8 a.m. on the following morning, on both dialysis-on days and dialysis-off days. The second blood glucose drift only occurred on dialysis-on days, from the initiation of hemodialysis to 2 h post-hemodialysis (8 a.m. to 2 p.m.); this was indicated by an inverted U. We identified 15 cases of asymptomatic hypoglycemia in three patients; 12 (80%) of these cases occurred during hemodialysis days. One case of hypoglycemia lasted from the start of hemodialysis to 2 h post-hemodialysis (360 min). Kazempour-Ardebili et al. [11] reported that hemodialysis was associated with a higher risk of hypoglycemia; this was evident within 24 h of dialysis, during which most instances of asymptomatic hypoglycemia and glucose nadir occurred. The association between dialysis and hypoglycemia was also evaluated by Jung et al. [14], who used the CGMS to evaluate nine patients with DM on hemodialysis; five of these patients experienced a total of 10 episodes of hypoglycemia, with 80% occurring on the day of hemodialysis. Most of these episodes were asymptomatic and occurred during the first 12 h on the day of dialysis. Riveline et al. [15] also reported two cases of intradialytic hypoglycemia. Furthermore, Mori et al. [13] documented an acute decline in blood glucose levels during a hemodialysis session in a patient with DM. Therefore, increased vigilance is warranted to detect and prevent asymptomatic hypoglycemia, especially on the day of dialysis, in patients with DM.
The average age of the patients in the present study was 82.52 ± 8.97 years, while that in previous studies has ranged from 61 ± 9 to 65 ± 13 years; participants in this study were the oldest on average among all relevant studies. Our study also has some limitations, including the relatively small sample size and the lack of a long-term follow-up. As a result of the limited number of cases, it is uncertain whether the results can be generalized to all older adult patients; additional studies with larger samples sizes are required. Furthermore, as we did not perform capillary blood glucose monitoring, we were unable to assess CGMS precision.
Conclusion
The results of this study indicated that hemodialysis not only increased the amplitude of glycemic excursion but also increased the risk of hypoglycemia in older individuals with diabetes undergoing maintenance hemodialysis. Furthermore, the effect of dialysis on blood glucose levels was usually maintained from the initiation of hemodialysis to 2 h post-hemodialysis. Our results emphasize the importance of monitoring changes in blood glucose levels in older adult patients undergoing dialysis. As a result of the limited sample size, it is uncertain whether the results of this study are applicable to all older adult patients; additional studies with larger sample sizes are needed. Future interventional studies are required to determine optimal methods for reducing blood glucose fluctuations and the incidence of hypoglycemia.
References
Fiorina P, Lattuada G, Ponari O, Silvestrini C, DallAglio P. Impaired nocturnal melatonin excretion and changes of immunological status in ischaemic stroke patients. Lancet. 1996;347:692–3. https://doi.org/10.1016/s0140-6736(96)91246-5.
Abbott KC, Bakris GL. Treatment of the diabetes mellitus patient: focus on cardiovascular and renal risk reduction. Prog Brain Res. 2002;139:289–98. https://doi.org/10.1016/s0079-6123(02)39025-3.
Haviv YS, Sharkia M, Safadi R. Hypoglycemia in patients with renal failure. Ren Fail. 2000;22:219–23. https://doi.org/10.1081/jdi-100100866.
Rigalleau V, Gin H. Carbohydrate metabolism in uraemia. Curr Opin Clin Nutr Metab Care. 2005;8:463–9. https://doi.org/10.1097/01.mco.0000172590.32564.b9.
Burmeister JE, Scapini A, da Rosa MD, da Costa MG, Campos BM. Glucose-added dialysis fluid prevents asymptomatic hypoglycemia in regular hemodialysis. Nephrol Dial Transplant. 2007;22:1184–9. https://doi.org/10.1093/ndt/gfl710.
Sangill M, Pedersen EB. The effect of glucose added to the dialysis fluid on blood pressure, blood glucose, and quality of life in hemodialysis patients: a placebo-controlled crossover study. Am J Kidney Dis. 2006;47:636–43. https://doi.org/10.1053/j.ajkd.2006.01.007.
Gai M, Merlo I, Dellepiane S, et al. Glycemic pattern in diabetes mellitus patients on hemodialysis: continuous glucose monitoring (CGM) analysis. Blood Purif. 2014;38:68–73. https://doi.org/10.1159/000362863.
Chantrel F, Sissoko H, Kepenekian L, et al. Influence of dialysis on the glucose profile in patients with diabetes: usefulness of continuous glucose monitoring. Horm Metab Res. 2014;46:810–3. https://doi.org/10.1055/s-0034-1370963.
Jin YP, Su XF, Yin GP, et al. Blood glucose fluctuations in hemodialysis patients with end stage diabetes mellitus nephropathy. J Diabetes Complicat. 2015;29:395–9. https://doi.org/10.1016/j.jdiacomp.2014.12.015.
Joubert M, Fourmy C, Henri P, Ficheux M, Lobbedez T, Reznik Y. Effectiveness of continuous glucose monitoring in dialysis patients with diabetes: the DIALYDIAB pilot study. Diabetes Res Clin Pract. 2015;107:348–54. https://doi.org/10.1016/j.diabres.2015.01.026.
Kazempour-Ardebili S, Lecamwasam VL, Dassanyake T, et al. Assessing glycemic control in maintenance hemodialysis patients with type 2 diabetes. Diabetes Care. 2009;32:1137–42. https://doi.org/10.2337/dc08-1688.
Mirani M, Berra C, Finazzi S, et al. Inter-day glycemic variability assessed by continuous glucose monitoring in insulin-treated type 2 diabetes patients on hemodialysis. Diabetes Technol Ther. 2010;12:749–53. https://doi.org/10.1089/dia.2010.0052.
Mori K, Emoto M, Abe M, Inaba M. Visualization of blood glucose fluctuations using continuous glucose monitoring in patients undergoing hemodialysis. J Diabetes Sci Technol. 2019;13:413–4. https://doi.org/10.1177/1932296818818429.
Jung HS, Kim HI, Kim MJ, et al. Analysis of hemodialysis-associated hypoglycemia in patients with type 2 diabetes using a continuous glucose monitoring system. Diabetes Technol Ther. 2010;12:801–7. https://doi.org/10.1089/dia.2010.0067.
Riveline JP, Teynie J, Belmouaz S, et al. Glycaemic control in type 2 diabetes mellitus patients on chronic haemodialysis: use of a continuous glucose monitoring system. Nephrol Dial Transpl. 2009;24:2866–71. https://doi.org/10.1093/ndt/gfp181.
Beck R, Connor C, Mullen DM, et al. The fallacy of average: how using HbA1c alone to assess glycemic control can be misleading. Diabetes Care. 2017;40:994–9. https://doi.org/10.2337/dc17-0636.
Divani M, Georgianos PI, Didangelos T, et al. Assessment of hyperglycemia, hypoglycemia and inter-day glucose variability using continuous glucose monitoring among diabetic patients on chronic hemodialysis. J Clin Med. 2021;10:4116. https://doi.org/10.3390/jcm10184116.
Battelino T, Danne T, Bergenstal RM, et al. Clinical targets for continuous glucose monitoring data interpretation: recommendations from the international consensus on time in range. Diabetes Care. 2019;42:1593–603. https://doi.org/10.2337/dci19-0028.
Kidney Disease: Improving Global Outcomes (KIDGO) Diabetes work group. KDIGO. Clinical Practice Guideline for Diabetes Management in Chronic Kidney Disease. Kidney Int. 2020;2020(98):S1-115. https://doi.org/10.1016/j.kint.2020.06.019.
International Diabetes Federation (2021) IDF Diabetes Atlas, 10th edn. Brussels: IDF. 2021. https://www.diabetesatlas.org
Maruyama N, Abe M. Targets and therapeutics for glycemic control in diabetes patients on hemodialysis. Contrib Nephrol. 2018;196:37–43. https://doi.org/10.1159/000485695.
Gallieni M, De Salvo C, Lunati ME, et al. Continuous glucose monitoring in patients with type 2 diabetes on hemodialysis. Acta Diabetol. 2021;58:975–81. https://doi.org/10.1007/s00592-021-01699-6.
Ceriello A, Colagiuri S. International Diabetes Federation guideline for management of post meal glucose: a review of recommendations. Diabet Med. 2008;25:1151–6. https://doi.org/10.1111/j.1464-5491.2008.02565.x.
Piconi L, Quagliaro L, Assaloni R, et al. Constant and intermittent high glucose enhances endothelial cell apoptosis through mitochondrial superoxide overproduction. Diabetes Metab Res Rev. 2006;22:198–203. https://doi.org/10.1002/dmrr.613.
Buscemi S, Re A, Batisi JA, et al. Glycaemic variability using continuous glucose monitoring and endothelial function in the metabolic syndrome and in type 2 diabetes. Diabet Med. 2010;27:872–8. https://doi.org/10.1111/j.1464-5491.
Lee JA, Sunwoo S, Kim YS, et al. The effect of sleep quality on the development of type 2 diabetes in primary care patients. J Korean Med Sci. 2016;31:240–6. https://doi.org/10.3346/jkms.2016.31.2.240.
Workgroup on Hypoglycemia, American Diabetes Association. Defining and reporting hypoglycemia in diabetes: a report from the American diabetes association workgroup on hypoglycemia. Diabetes Care. 2005;28:1245–9. https://doi.org/10.2337/diacare.28.5.1245.
Acknowledgements
We would like to thank the Department of Geriatric Endocrinology for implementing the continuous glucose monitoring system. We thank the Department of Geriatric Dialysis for its assistance and the nurses at the Geriatric Nephrology department for measuring the capillary blood glucose levels in this study. We also acknowledge the support of the Department of Geriatric Gerontology in conducting the laboratory index test.
Funding
The rapid service fee for this work was supported by grants from the Second Medical Centre of Chinese PLA General Hospital.
List of Investigators
Jinzhu Li, Ruiqin Zhang, Zhen Wu, Jiayu Guo, Zhiying Wang, Shuhui Li, Chunlin Li, Guang Yang, Xiaoling Cheng.
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Authorship Contributions
Conceptualization: Jinzhu Li and Guang Yang; Methodology: Jinzhu Li and Guang Yang; Patient enrollment: Ruiqin Zhang, Zhiying Wang, and Shuhui Li; Statistical analysis: Jiayu Guo and Zhen Wu; Drafting of the initial version of the manuscript: Jinzhu Li and Guang Yang; Supervision: Chunlin Li and Xiaoling Cheng. All authors have read and approved the final manuscript.
Disclosures
Jinzhu Li, Ruiqin Zhang, Zhen Wu, Jiayu Guo, Zhiying Wang, Shuhui Li, Chunlin Li, Guang Yang, and Xiaoling Cheng have nothing to disclose.
Compliance with Ethics Guidelines
This study was performed in line with the principles of the Declaration of Helsinki. Approval was granted by the Ethics Committee of the Chinese People’s Liberation Army General Hospital (S2021-424-01, July 29, 2021). All patients provided written informed consent to participate in the study. All patients consent to having their data published in a journal article.
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Li, J., Zhang, R., Wu, Z. et al. Blood Glucose Fluctuation in Older Adults with Diabetes Mellitus and End-Stage Renal Disease on Maintenance Hemodialysis: An Observational Study. Diabetes Ther 13, 1353–1365 (2022). https://doi.org/10.1007/s13300-022-01274-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13300-022-01274-4