Abstract
The objective of this study was to evaluate the safety and effectiveness of cryopreserved umbilical cord (UC) allograft as a nerve wrap around the neurovascular bundle (NVB) in accelerating return to continence after radical prostatectomy. A single-center, retrospective study was performed on 200 patients who underwent bilateral, nerve-sparing robot-assisted radical prostatectomy (RARP) with and without placement of UC around the NVBs (n = 100/group). Patients were excluded if they had previous simple or transurethral prostatectomy or history of pelvic radiation. Post-operative continence, defined as 0 or 1 safety pad, was analyzed between groups at 1, 3, 6, and 12 months. Complications, biochemical recurrence and adverse events were assessed to determine safety. Patients who underwent RARP with UC were significantly more likely to be continent at 1 month (65% vs. 44%, p = 0.018), 3 months (83% vs. 70%, p = 0.03), and 12 months (97% vs. 87%, p = 0.009). Sample stratification revealed that UC is beneficial for obese patients and those > 60 years, both of which are high risk for post-RARP incontinence. Biochemical failure was noted in 2 (UC) and 4 (control) patients. No adverse events or complications related to UC were observed. The results suggest that UC allograft is safe and accelerates continence recovery in post-RARP patients. Prospective, randomized trials are warranted.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Prostate cancer is the most common cancer diagnosis among men, with more than 160,000 new cases in the United States each year [1]. Radical prostatectomy (RP) is the recommended, front-line treatment approach for patients with clinically localized prostate cancer and greater than 10-year life expectancy [2]. There are several surgical approaches for RP, with the majority of cases conducted via the minimally invasive da Vinci robotic-assisted surgical system [3, 4]. This technology provides surgeons with superior visualization, enhanced dexterity and greater precision, and as a result, improves surgical and functional outcomes. Despite these operative advances, urinary incontinence remains a problem for at least 50% of men who undergo robot-assisted prostatectomy [5,6,7,8]. Consequently, these men may have a poor quality of life and report anxiety, fear and embarrassment [8], as well as loss of sense of control, depression and decreased social interactions [6]. Unfortunately, post-prostatectomy incontinence is expected to rise due to the increasing number of procedures performed which increases burden on society [9].
Urethral sphincter incompetence is generally considered as the most important contributing factor to post-RP incontinence and is likely a result of damage to supporting structures and nerves rather than damage to the sphincter itself [10, 11]. It has been found that the neurovascular bundle (NVB) directly innervates the membranous urethra [12], and intraoperative stimulation of the NVB significantly increases urethral pressure [13]. These findings suggest that NVB damage affects the continence mechanism, and preservation leads to earlier recovery of continence following RP [14,15,16,17,18,19,20,21]. While NVB preservation minimizes incontinence, it is often difficult to completely mitigate NVB manipulation as anatomic studies show a plate-like formation of nerves rather than the traditional bundle [10]. Furthermore, dissection, traction, and surgical insult can elicit inflammation, mitigating the healing process and impacting continence after prostatectomy.
Amniotic and umbilical cord (UC) tissues have been used in a wide variety of clinical applications to reduce inflammation and promote regenerative healing including ocular surface reconstruction [22, 23], tendon repair [24, 25], wound healing [26,27,28,29,30,31,32,33], and burns [34,35,36,37]. Additionally, amniotic membrane has been shown to support nerve regeneration [38,39,40], with evidence of increased axons possessing myelin sheaths of normal thickness, as well as, less inter-axonal fibrosis [38]. While the clinical use of UC is well documented, its use in reducing inflammation and promoting regenerative healing in the NVB is relatively new. Several recent studies have demonstrated enhanced return to potency using dehydrated human amnion/chorion membrane [41,42,43], with only one of them reporting continence outcomes [41]. While the short-term results are encouraging, no studies have assessed continence outcomes past 2 months. The aim of this study was to assess the use of cryopreserved UC allograft as a nerve wrap during robot-assisted radical prostatectomy (RARP) in accelerating return to continence up to 1-year post-op.
Materials and methods
Full institutional board approval and waiver of informed consent was granted for this study. A retrospective medical chart review was conducted on patients who underwent bilateral nerve-sparing RARP with and without cryopreserved UC allograft (CLARIX CORD 1K, Amniox Medical Inc., Miami, FL, USA) from January 2015 to September 2017. Patients were eligible for inclusion in this study if they had at least 3 months of follow-up data. Patients were excluded if they had previous history of pelvic radiation or prostatectomy or NVB damage during surgery.
Treatment procedures
All RARP procedures were performed at Hackensack Meridian Health Hackensack University Medical Center (Hacensack, NJ, USA) and all patients were followed up at the New Jersey Center for Prostate Cancer and Urology (Maywood, NJ, USA) by three surgeons using the standard trans-peritoneal six-port technique with the da Vinci surgical system (Intuitive Surgical, Sunnyvale, CA, USA). Bilateral, athermal nerve-sparing RARP was performed on each patient, with bladder neck reconstruction, an anterior suspension stitch, and posterior reconstruction. A 6 × 3.0 cm UC allograft (CLARIX CORD 1K, Amniox Medical, Miami, FL, USA) was cut into two longitudinal pieces (1.5 cm in width) and placed circumferentially around each NVB as a nerve wrap through an assistant port.
Outcome measures
Data collected from the medical charts included demographic information, significant medical history including co-morbidities, prostate size, blood loss, perineural invasion, positive surgical margins, PSA levels, Gleason score, and clinical stage. The primary endpoint was the proportion of men with return of urinary continence at follow-up. Continence was defined as use of no or one safety pad. The secondary endpoint was the safety of UC for use as NVB wrap during NS-RARP by assessing both treatment related and treatment emergent adverse events through review of physical exams and assessments. Adverse events were further classified as procedure related or product related.
Statistical analysis
Post-operative outcomes were analyzed between groups using the Student’s t test for continuous factors and the Chi-Square or Fisher’s exact test for categorical factors. To identify and adjust for factors that could influence continence rates at each follow-up, multiple regression analysis was implemented. All statistical analyses were performed using SPSS v. 20.0 (IBM SPSS Statistics, Chicago, IL, USA). A p value < 0.05 was used to determine statistical significance.
Results
A total of 200 patients who underwent bilateral nerve-sparing RARP with and without cryopreserved UC (n = 100/group) met the eligibility criteria and were included for retrospective analysis. Demographics, pre-operative characteristics (Table 1), and intraoperative outcomes (Table 2) were comparable between the UC and control group. The mean age in the UC group was 61.9 ± 7.1 years, while the control group was 59.6 ± 7.0 years (p = 0.05). Additionally, the BMI was 28.5 ± 4.6 kg/m2 in the UC group and 29.2 ± 5.2 kg/m2 in the control group (p = 0.39). Clinical stage, surgical margins, prostate size, and PSA levels did not differ between groups.
Continence recovery rates at 1, 3, 6, and 12 months were significantly better for patients receiving UC when compared to the control group at all points in time, except at 6 months: 65% (42/65) vs. 44% (31/70) at 1 month (p = 0.018), 83% (83/100) vs. 70% (70/100) at 3 months (p = 0.03), 90% (90/100) vs. 84% (84/100) at 6 months (p = 0.21), and 97% (97/100) vs. 87% (87/100) at 12 months (p = 0.009) (Fig. 1). When defining continence as use of zero pads, continence recovery rates were significantly better for patients receiving UC at 1, 3, 6, and 12 months compared to the control group at all time points: 55% (36/65) vs. 30% (21/70) at 1 month (p = 0.003), 68% (68/100) vs. 52% (52/100) at 3 months (p = 0.021), 84% (84/100) vs. 64% (64/100) at 6 months (p = 0.001), and 90% (90/100) vs. 80% (80/100) at 12 months (p = 0.048).
To identify and adjust for factors that could influence continence at each follow-up, binary logistic regression analysis was implemented. The factors included in each model were treatment, age, BMI, diabetes, hypertension, Gleason score, perineural invasion, blood loss, and prostate size. After controlling for these factors, only treatment and age were significant predictors of continence outcomes at 1 month (p = 0.02 and 0.006, respectively), 3 months (p = 0.032 and 0.001, resp.) and 12 months post-RARP (p = 0.005 and 0.001, resp.). At 6 months, only age was predictive of continence (p = 0.001).
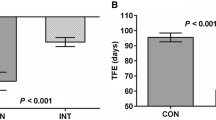
The study sample was further stratified into two groups according to BMI and age, and the continence recovery rates at 1, 3, 6, and 12 months were compared between the UC and control group. For patients ≥ 30 kg/m2 (n = 65), continence recovery rates were significantly better in the UC group than the control group at all time points (p < 0.05), while there was no significant difference between groups for patients < 30 kg/m2 (n = 120) (Fig. 2). In addition, for patients > 60 years old (n = 105), the UC group was significantly more continent than the control group at 1, 3, and 12 months (p < 0.05); whereas, continence did not significantly differ between groups in patients ≤ 60 years (n = 95) at any time point (Fig. 3).
Safety was assessed through review of physical exams and assessments for both treatment related and treatment emergent adverse events. No complications or adverse events related to UC were observed throughout the duration of the study. In addition, biochemical failure was noted in 2 (UC) and 4 (control) patients and a nonsignificant improvement in potency (SHIM > 16) was noted in patients who received UC.
Discussion
Despite recent advances in operative technique, there remains a convalescent period characterized by urinary incontinence even when the NVBs are well preserved [44,45,46]. This delay in continence recovery is believed to be a result of dissection or traction injury to the NVB and supporting structures which induces an inflammatory response [11, 47]. In this retrospective review, we assessed the clinical effectiveness and safety of cryopreserved UC as a NVB wrap in facilitating continence recovery in patients who underwent bilateral, NS-RARP due to the known anti-inflammatory actions of the UC. Our results showed that there was a significantly higher percentage of patients who became continent in the UC vs. control group at 1 month (65% vs. 44%), 3 months (83% vs. 70%,), and 12 months post-RARP (97% vs. 87%). After controlling for possible covariates influencing continence outcomes, UC and age were found to have a significant effect on continence recovery at 1, 3, and 12 months. Moreover, we found that UC significantly benefits patients with a BMI ≥ 30 kg/m2 or patients > 60 years old. These findings are especially promising given obesity [48,49,50,51] and old age [50, 52,53,54,55,56] are significant risk factors of post-RP incontinence, specifically at 6- and 12 months [57].
Other attempts have been made to reduce post-surgical inflammation after RARP. Some studies evaluated the induction of regional hypothermia via an endorectal cooling balloon; however, a recent randomized control trial demonstrated no significant benefit for continence recovery [58]. In addition, other studies have assessed the use of dehydrated AM tissue around the NVB in accelerating return to potency; however, only one study has reported continence outcomes [41]. In that study, Patel et al. found that the mean time to continence was shorter in the dHAM group by 0.62 months (p = 0.03); however, there was no significant difference in continence outcomes between the dHAM and control group at the 2-month follow-up (81% vs. 74%, p = 0.37). It is possible that the lack of significant findings is due to the surgical technique or method of AM preservation. While cryopreserved tissues retain their native architecture and biological components relevant to fresh UC tissue [59, 60], dehydrated AM tissues are structurally compromised and almost entirely lack key biological signaling complex, HC-HA/PTX3 [59].
The overall improvement in continence observed in this study may be explained by the anti-inflammatory and regenerative properties inherent to UC tissues [61,62,63]. UC induces apoptosis of neutrophils [64, 65], monocytes, and macrophages [66] and reduces infiltration of neutrophils [64, 65], macrophages [67, 68], and lymphocytes [69]. Additionally, UC is rich in cytokines and neurotrophic factors, particularly nerve growth factor, which plays an important role in nerve regeneration and epithelial healing [70,71,72,73]. One key biological modulator present in cryopreserved UC tissues, HC-HA/PTX3, upregulates IL-10, downregulates IL-12, and polarizes macrophages toward the M2 phenotype, all of which play an integral role in the healing process [61,62,63]. Together, these conditions provide an optimal environment for healing and facilitate recovery of the NVB post-RARP.
While our data are encouraging, this study is limited by its lack of prospective randomization and potential placebo bias. Patients treated with UC were also predominantly treated after April 2016 compared to control patients treated prior to them; however, the surgical technique was similar during this period. Several pre-operative characteristics significantly differed between groups; however, the treatment group represented a higher risk sample as age and Gleason scores were more advanced. At our practice, UC is used for patients undergoing only bilateral nerve-sparing RARP, with < T4 prostate cancer, without erectile dysfunction pre-op, and without neurogenic bladders. Future health economic studies are needed to determine cost and benefit analysis of using the UC (at a cost of approximately $2500). Prospective, randomized clinical trials with validated questionnaires are necessary to further ascertain the effect of UC on urinary incontinence following NS-RARP.
Conclusion
The results of this retrospective study suggest that UC can be safely used to facilitate return to continence following NS-RARP. Prospective, randomized trials are warranted to further evaluate the potential benefit of UC.
References
Litwin MS, Tan HJ (2017) The diagnosis and treatment of prostate cancer: a review. JAMA 317:2532–2542
Heidenreich A, Bastian PJ, Bellmunt J, Bolla M, Joniau S, van der Kwast T et al (2014) EAU guidelines on prostate cancer. Part 1: screening, diagnosis, and local treatment with curative intent-update 2013. Eur Urol 65:124–137
Carlucci JR, Nabizada-Pace F, Samadi DB (2009) Robot-assisted laparoscopic radical prostatectomy: technique and outcomes of 700 cases. Int J Biomed Sci: IJBS. 5:201–208
Ficarra V, Cavalleri S, Novara G, Aragona M, Artibani W (2007) Evidence from robot-assisted laparoscopic radical prostatectomy: a systematic review. Eur Urol 51:45–55 (discussion 6)
Alivizatos G, Skolarikos A (2005) Incontinence and erectile dysfunction following radical prostatectomy: a review. Sci World J 5:747–758
Ko WF, Sawatzky JA (2008) Understanding urinary incontinence after radical prostatectomy: a nursing framework. Clin J Oncol Nurs 12:647–654
Liatsikos EN, Assimakopoulos K, Stolzenburg JU (2008) Quality of life after radical prostatectomy. Urol Int 80:226–230
Fan X, Heyes S, King L (2012) Men’s experiences of urinary incontinence after prostatectomy. Cancer Nurs Pract. 11:29–34
Wessells H, Peterson AC (2012) Surgical treatment of male sphincteric urinary incontinence: the male perineal sling and artificial urinary sphincter. In: Wein AJ et al (eds) Campbell-Walsh urology, 10th ed, vol 3. Saunders, Philadelphia, pp 2290–2305
Hoyland K, Vasdev N, Abrof A, Boustead G (2014) Post-radical prostatectomy incontinence: etiology and prevention. Rev Urol 16:181–188
van der Poel HG, de Blok W, Joshi N, van Muilekom E (2009) Preservation of lateral prostatic fascia is associated with urine continence after robotic-assisted prostatectomy. Eur Urol 55:892–900
Strasser H, Bartsch G (2004) Anatomic basis for the innervation of the male pelvis. Der Urologe Ausg A 43:128–132
Nelson CP, Montie JE, McGuire EJ, Wedemeyer G, Wei JT (2003) Intraoperative nerve stimulation with measurement of urethral sphincter pressure changes during radical retropubic prostatectomy: a feasibility study. J Urol 169:2225–2228
Ko YH, Coelho RF, Chauhan S, Sivaraman A, Schatloff O, Cheon J et al (2012) Factors affecting return of continence 3 months after robot-assisted radical prostatectomy: analysis from a large, prospective data by a single surgeon. J Urol 187:190–194
Loughlin KR, Prasad MM (2010) Post-prostatectomy urinary incontinence: a confluence of 3 factors. J Urol 183:871–877
Song C, Doo CK, Hong JH, Choo MS, Kim CS, Ahn H (2007) Relationship between the integrity of the pelvic floor muscles and early recovery of continence after radical prostatectomy. J Urol 178:208–211
Sacco E, Prayer-Galetti T, Pinto F, Fracalanza S, Betto G, Pagano F et al (2006) Urinary incontinence after radical prostatectomy: incidence by definition, risk factors and temporal trend in a large series with a long-term follow-up. BJU Int 97:1234–1241
Catarin MV, Manzano GM, Nobrega JA, Almeida FG, Srougi M, Bruschini H (2008) The role of membranous urethral afferent autonomic innervation in the continence mechanism after nerve sparing radical prostatectomy: a clinical and prospective study. J Urol 180:2527–2531
Ozdemir MB, Eskicorapci SY, Baydar DE, Cumhur M, Onderoglu S, Ozen H (2007) A cadaveric histological investigation of the prostate with three-dimensional reconstruction for better results in continence and erectile function after radical prostatectomy. Prostate Cancer Prost Dis 10:77–81
Kaye DR, Hyndman ME, Segal RL, Mettee LZ, Trock BJ, Feng Z et al (2013) Urinary outcomes are significantly affected by nerve sparing quality during radical prostatectomy. Urology. 82:1348–1353
Burkhard FC, Kessler TM, Fleischmann A, Thalmann GN, Schumacher M, Studer UE (2006) Nerve sparing open radical retropubic prostatectomy—does it have an impact on urinary continence? J Urol 176:189–195
Tseng SC (2001) Amniotic membrane transplantation for ocular surface reconstruction. Biosci Rep 21:481–489
Gheorghe A, Pop M, Burcea M, Serban M (2016) New clinical application of amniotic membrane transplant for ocular surface disease. J Med Life 9:177–179
Gao M, Zhao H, Tian D, Yu K, Bai J, Dong R et al (2013) Experimental study on human amniotic membrane for repairing tendon sheath defect. Chin J Rep Reconst Surg. 27:335–339
Liu C, Yu K, Bai J, Tian D, Liu G (2018) Experimental study of tendon sheath repair via decellularized amnion to prevent tendon adhesion. PLoS One 13:e0205811
Snyder RJ, Shimozaki K, Tallis A, Kerzner M, Reyzelman A, Lintzeris D et al (2016) A prospective, randomized, multicenter, controlled evaluation of the use of dehydrated amniotic membrane allograft compared to standard of care for the closure of chronic diabetic foot ulcer. Wounds Compen Clin Res Pract 28:70–77
Faulk WP, Matthews R, Stevens PJ, Bennett JP, Burgos H, Hsi BL (1980) Human amnion as an adjunct in wound healing. Lancet (London, England). 1:1156–1158
Gruss JS, Jirsch DW (1978) Human amniotic membrane: a versatile wound dressing. Can Med Assoc J 118:1237–1246
Kirsner RS, Sabolinski ML, Parsons NB, Skornicki M, Marston WA (2015) Comparative effectiveness of a bioengineered living cellular construct vs. a dehydrated human amniotic membrane allograft for the treatment of diabetic foot ulcers in a real world setting. Wound Repair Regener. 23:737–744
Kraus I, Sabolinski ML, Skornicki M, Parsons NB (2017) The comparative effectiveness of a human fibroblast dermal substitute versus a dehydrated human amnion/chorion membrane allograft for the treatment of diabetic foot ulcers in a real-world setting. Wounds Compend Clin Res Pract 29:125–132
Pacaccio DJ, Cazzell SM, Halperin GJ, Kasper MA, Neutel JM, O’Carroll BD et al (2018) Human placental membrane as a wound cover for chronic diabetic foot ulcers: a prospective, postmarket. CLOSURE study. J Wound Care 27:S28–S37
Raphael A (2016) A single-centre, retrospective study of cryopreserved umbilical cord/amniotic membrane tissue for the treatment of diabetic foot ulcers. J Wound Care. 25:S10–S17
Shun A, Ramsey-Stewart G (1983) Human amnion in the treatment of chronic ulceration of the legs. Med J Aust 2:279–283
Branski LK, Herndon DN, Celis MM, Norbury WB, Masters OE, Jeschke MG (2008) Amnion in the treatment of pediatric partial-thickness facial burns. Burns J Int Soc Burn Injur 34:393–399
Bose B (1979) Burn wound dressing with human amniotic membrane. Ann R Coll Surg Engl 61:444–447
Walker AB, Cooney DR, Allen JE (1977) Use of fresh amnion as a burn dressing. J Pediatr Surg 12:391–395
Singh R, Purohit S, Chacharkar MP, Bhandari PS, Bath AS (2007) Microbiological safety and clinical efficacy of radiation sterilized amniotic membranes for treatment of second-degree burns. Burns J Int Soc Burn Injur 33:505–510
Fesli A, Sari A, Yilmaz N, Comelekoglu U, Tasdelen B (2014) Enhancement of nerve healing with the combined use of amniotic membrane and granulocyte-colony-stimulating factor. J Plast Reconstruct Aesth Surg JPRAS. 67:837–843
Karaman M, Tuncel A, Sheidaei S, Senol MG, Karabulut MH, Deveci I et al (2013) Amniotic membrane covering for facial nerve repair. Neural Regener Res 8:975–982
John T, Tighe S, Sheha H, Hamrah P, Salem ZM, Cheng AMS et al (2017) Corneal nerve regeneration after self-retained cryopreserved amniotic membrane in dry eye disease. J Ophthalmol 2017:6404918
Patel VR, Samavedi S, Bates AS, Kumar A, Coelho R, Rocco B et al (2015) Dehydrated human amnion/chorion membrane allograft nerve wrap around the prostatic neurovascular bundle accelerates early return to continence and potency following robot-assisted radical prostatectomy: propensity score-matched analysis. Eur Urol 67:977–980
Ogaya-Pinies G, Palayapalam-Ganapathi H, Rogers T, Hernandez-Cardona E, Rocco B, Coelho RF et al (2018) Can dehydrated human amnion/chorion membrane accelerate the return to potency after a nerve-sparing robotic-assisted radical prostatectomy? Propensity score-matched analysis. J Robot Surg 12:235–243
Razdan S, Bajpai RR, Razdan S, Sanchez MA (2018) A matched and controlled longitudinal cohort study of dehydrated human amniotic membrane allograft sheet used as a wraparound nerve bundles in robotic-assisted laparoscopic radical prostatectomy: a puissant adjunct for enhanced potency outcomes. J Robot Surg 13(3):475–481
Salomon L, Droupy S, Yiou R (2015) Soulie M [Functional results and treatment of functional dysfunctions after radical prostatectomy]. Progres en urologie : journal de l’Association francaise d’urologie et de la Societe francaise d’urologie 25:1028–1066
Steineck G, Helgesen F, Adolfsson J, Dickman PW, Johansson JE, Norlen BJ et al (2002) Quality of life after radical prostatectomy or watchful waiting. N Engl J Med 347:790–796
Nguyen LN, Head L, Witiuk K, Punjani N, Mallick R, Cnossen S et al (2017) The risks and benefits of cavernous neurovascular bundle sparing during radical prostatectomy: a systematic review and meta-analysis. J Urol 198:760–769
Menon M, Kaul S, Bhandari A, Shrivastava A, Tewari A, Hemal A (2005) Potency following robotic radical prostatectomy: a questionnaire based analysis of outcomes after conventional nerve sparing and prostatic fascia sparing techniques. J Urol. 174:2291–2296
Wolin KY, Luly J, Sutcliffe S, Andriole GL, Kibel AS (2010) Risk of urinary incontinence following prostatectomy: the role of physical activity and obesity. J Urol 183:629–633
Ahlering TE, Eichel L, Edwards R, Skarecky DW (2005) Impact of obesity on clinical outcomes in robotic prostatectomy. Urology. 65:740–744
Sanchez-Salas R, Prapotnich D, Rozet F, Mombet A, Cathala N, Barret E et al (2010) Laparoscopic radical prostatectomy is feasible and effective in ‘fit’ senior men with localized prostate cancer. BJU Int 106:1530–1536
Wiltz AL, Shikanov S, Eggener SE, Katz MH, Thong AE, Steinberg GD et al (2009) Robotic radical prostatectomy in overweight and obese patients: oncological and validated-functional outcomes. Urology. 73:316–322
Heesakkers J, Farag F, Bauer RM, Sandhu J, De Ridder D, Stenzl A (2017) Pathophysiology and contributing factors in postprostatectomy incontinence: a review. Eur Urol 71:936–944
Limani K, Albisinni S, Aoun F, Le Dinh D, Biaou I, Hawaux E et al (2017) Quality of life after robotic prostatectomy: impact of BMI and age on urinary incontinence. Progres en urologie : journal de l’Association francaise d’urologie et de la Societe francaise d’urologie. 27:244–252
Kim PH, Pinheiro LC, Atoria CL, Eastham JA, Sandhu JS, Elkin EB (2013) Trends in the use of incontinence procedures after radical prostatectomy: a population based analysis. J Urol 189:602–608
Prabhu V, Sivarajan G, Taksler GB, Laze J, Lepor H (2014) Long-term continence outcomes in men undergoing radical prostatectomy for clinically localized prostate cancer. Eur Urol 65:52–57
Catalona WJ, Carvalhal GF, Mager DE, Smith DS (1999) Potency, continence and complication rates in 1,870 consecutive radical retropubic prostatectomies. J Urol 162:433–438
Matsushita K, Kent MT, Vickers AJ, von Bodman C, Bernstein M, Touijer KA et al (2015) Preoperative predictive model of recovery of urinary continence after radical prostatectomy. BJU Int 116:577–583
Huynh LM, Skarecky D, Porter J, Wagner C, Witt J, Wilson T et al (2018) A randomized control trial of anti-inflammatory regional hypothermia on urinary continence during robot-assisted radical prostatectomy. Sci Rep 8:16352
Cooke M, Tan EK, Mandrycky C, He H, O’Connell J, Tseng SC (2014) Comparison of cryopreserved amniotic membrane and umbilical cord tissue with dehydrated amniotic membrane/chorion tissue. J Wound Care 23(465–74):76
Kia Tan E, Cooke M, Mandrycky C, Mahabole M, He H, O’Connell J et al (2014) Structural and biological comparison of cryopreserved and fresh amniotic membrane tissues. J Biomater Tissue Eng 4(5):379–388
He H, Li W, Tseng DY, Zhang S, Chen SY, Day AJ et al (2009) Biochemical characterization and function of complexes formed by hyaluronan and the heavy chains of inter-alpha-inhibitor (HC*HA) purified from extracts of human amniotic membrane. J Biol Chem 284:20136–20146
He H, Zhang S, Tighe S, Son J, Tseng SC (2013) Immobilized heavy chain-hyaluronic acid polarizes lipopolysaccharide-activated macrophages toward M2 phenotype. J Biol Chem 288:25792–25803
Koh TJ, DiPietro LA (2011) Inflammation and wound healing: the role of the macrophage. Expert Rev Mol Med 13:e23
Park WC, Tseng SC (2000) Modulation of acute inflammation and keratocyte death by suturing, blood, and amniotic membrane in PRK. Invest Ophthalmol Vis Sci 41:2906–2914
Wang MX, Gray TB, Park WC, Prabhasawat P, Culbertson W, Forster R et al (2001) Reduction in corneal haze and apoptosis by amniotic membrane matrix in excimer laser photoablation in rabbits. J Cataract Refract Surg 27:310–319
Shimmura S, Shimazaki J, Ohashi Y, Tsubota K (2001) Antiinflammatory effects of amniotic membrane transplantation in ocular surface disorders. Cornea 20:408–413
Bauer D, Wasmuth S, Hermans P, Hennig M, Meller K, Meller D et al (2007) On the influence of neutrophils in corneas with necrotizing HSV-1 keratitis following amniotic membrane transplantation. Exp Eye Res 85:335–345
Heiligenhaus A, Bauer D, Meller D, Steuhl KP, Tseng SC (2001) Improvement of HSV-1 necrotizing keratitis with amniotic membrane transplantation. Invest Ophthalmol Vis Sci 42:1969–1974
Bauer D, Wasmuth S, Hennig M, Baehler H, Steuhl KP, Heiligenhaus A (2009) Amniotic membrane transplantation induces apoptosis in T lymphocytes in murine corneas with experimental herpetic stromal keratitis. Invest Ophthalmol Vis Sci 50:3188–3198
Lambiase A, Sacchetti M, Bonini S (2012) Nerve growth factor therapy for corneal disease. Curr Opin Ophthalmol 23:296–302
Aloe L, Tirassa P, Lambiase A (2008) The topical application of nerve growth factor as a pharmacological tool for human corneal and skin ulcers. Pharmacol Res 57:253–258
Touhami A, Grueterich M, Tseng SC (2002) The role of NGF signaling in human limbal epithelium expanded by amniotic membrane culture. Invest Ophthalmol Vis Sci 43:987–994
Banerjee A, Nurnberger S, Hennerbichler S, Riedl S, Schuh CM, Hacobian A et al (2014) In toto differentiation of human amniotic membrane towards the Schwann cell lineage. Cell Tissue Bank. 15:227–239
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
Amniox Medical (Miami, FL, USA) funded IRB administrative costs (Grant CR-2018). There are no other conflicts of interest.
Research involving human participants
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
The IRB found that this research meets the requirements for a waiver of consent under 45 CFR 46.116(d).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Ahmed, M., Esposito, M. & Lovallo, G. A single-center, retrospective review of robot-assisted laparoscopic prostatectomy with and without cryopreserved umbilical cord allograft in improving continence recovery. J Robotic Surg 14, 283–289 (2020). https://doi.org/10.1007/s11701-019-00972-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11701-019-00972-9