Abstract
Background
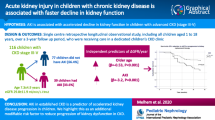
Diabetic ketoacidosis (DKA) and hyperglycaemia without ketoacidosis are common acute complications of diabetes. Their association with acute kidney injury (AKI) and diabetic kidney disease (DKD) was studied.
Methods
The study group consisted of 197 children with type 1 diabetes with average diabetes duration of 8.08 ± 2.32 years. The medical history of the patients was retrospectively reviewed. The number of children with severe hyperglycaemia, DKA and AKI was assessed. The association with the risk of chronic kidney disease (CKD) was analysed.
Results
AKI was found in 14% of cases hospitalised for DKA and 8% of cases hospitalised for hyperglycaemia. Patients with AKI showed a significantly increased corrected sodium (141.23 ± 5.09 mmol/L, p = 0.035). Patients with AKI in DKA showed a significant increase in WBC (20.73 ± 8.71 × 103/µL, p = 0.0009). Follow-up analysis after a minimum of 5 years of diabetes revealed that a single episode of DKA was found in 63 patients and a single episode of AKI in 18 patients. Two or more episodes of DKA were found in 18 patients, and nine cases were complicated by AKI. These patients showed a significant increase in urinary albumin excretion (44.20 ± 64.21 mg/24 h), the highest values of eGFR and the worst glycaemic control.
Conclusions
Diabetic children can develop AKI in the course of DKA and hyperglycaemia without ketoacidosis, which is associated with volume depletion and reflected by corrected sodium concentration. AKI in DKA seems to be complicated by stress and inflammation activation. AKI and poor glycaemic control with repeated DKA episodes can magnify the risk of progression to DKD.
Graphical abstract

A higher resolution version of the Graphical abstract is available as Supplementary information
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Diabetic kidney disease (DKD) is a leading cause of chronic kidney disease (CKD), with a high risk of dialysis and mortality [1]. Given the growing incidence of type 1 and type 2 diabetes in children and adolescents, DKD represents a significant public health problem [2, 3]. The pathophysiology of DKD is complex and multifactorial. Chronic and acute hyperglycaemia associated with diabetes leads to glomerular hypertrophy, glomerulosclerosis, tubulointerstitial inflammation and fibrosis. These result in the natural history in DKD of glomerular hyperfiltration, progressive albuminuria, declining glomerular filtration rate (GFR) and ultimately, kidney failure [1]. The earliest alterations in the kidney structure are apparent within 1.5–2 years of type 1 diabetes diagnosis, in the form of a thickening of the glomerular basement membrane. Mesangial volume expansion is detectable within 5–7 years after diabetes diagnosis and then increased albuminuria can also occur [4]. Although DKD is considered a glomerular disease, a growing body of evidence suggests that tubular-interstitial injury may be the first alteration in DKD [5].
The common risk factors for DKD include age, age at onset, duration of diabetes, genetics, gender, glycaemic control, blood pressure, cholesterol levels and smoking [6]. Another risk factor for CKD is acute kidney injury (AKI). Paediatric AKI is associated with increased morbidity and mortality [7,8,9]. For those who survive AKI, recent data suggest that they likely have permanent kidney damage. These findings have challenged the previous belief that AKI was a completely reversible event [10]. AKI can also occur in diabetes [11]. Acute hyperglycaemic events, chronic poor glycaemic control, and diabetic ketoacidosis (DKA) can lead to AKI [11, 12]. Hyperglycaemia has been shown to induce kidney inflammation and tubulopathy, and poor glycaemic control can lead to polyuria with resultant volume contraction and hypovolemia, which is subsequently associated with the development of pre-renal AKI [13, 14].
DKA is a common and severe acute complication of diabetes. It is characterised by a combination of hyperglycaemia, metabolic acidosis and the production of ketone bodies [15,16,17,18]. DKA is currently the leading cause of hospitalisation, morbidity and mortality in youth with type 1 diabetes [15, 19, 20]. Severe hyperglycaemia associated with DKA leads to osmotic diuresis, dehydration and significant pre-renal AKI [17, 18, 21]. In a study by Hursh et al., up to 64% of children with type 1 diabetes hospitalised for DKA developed AKI [15].
AKI is currently defined by the Kidney Disease Improving Global Outcomes (KDIGO) consensus classification based on conventional serum creatinine and urine output (UO) criteria [22]. However, despite the strict standards of AKI and the marked intravascular volume depletion that occurs in DKA, kidney injury in DKA in children has not been systematically studied. Moreover, the impact of DKA on AKI and chronic diabetic kidney injury has not been studied.
The primary objective of this study was to determine the proportion of children with type 1 diabetes hospitalised for the disease that developed severe hyperglycaemia, DKA and AKI. As a secondary objective, we wanted to determine whether developing DKA and AKI was associated with an increased risk for CKD in children with type 1 diabetes for more than 5 years.
Materials and methods
Study design and participants
The study group consisted of 197 adolescents with type 1 diabetes (104 girls and 93 boys) with a mean age of 14.69 ± 2.64 years and with a duration of diabetes of more than 5 years, who were hospitalised in the Department of Paediatric Diabetes and Obesity at Poznan University of Medical Sciences, Poland (Fig. 1). The data was collected in 2019 and 2020. All patients were Caucasian. The medical history of the patients and detailed information, including gender, age, height, weight, body mass index and pre-existing CKD, were retrospectively reviewed based on electronic hospital records. We analysed data obtained at the onset of diabetes and later acute complications, as well as at planned medical control after a minimum of 5 years of diabetes duration. The planned hospitalisation needed to have taken place a minimum of 3 months after the last episode of acute complications of diabetes. However, because in most cases acute complications of diabetes were at the onset of the disease, the observational period was almost as long as the duration of diabetes. No patients had CKD for reasons besides diabetes.
The cases hospitalised due to acute complications of diabetes were divided into the following groups:
-
N — cases with hyperglycaemia without DKA or AKI
-
DKA — cases with DKA without AKI
-
AKI — cases with AKI
On admission, because of acute complications, biological parameters, including pH, HCO3, serum creatinine, blood glucose (Glu), serum sodium (Na), haematocrit (HCT) and white blood cell (WBC) count, were collected. The serum creatinine was assessed within 24 h of admission. Corrected sodium (cNa) during DKA episodes was calculated using the following formula:
For our patients’ treatment and fluid regimen, we follow the International Society for Pediatric and Adolescent Diabetes (ISPAD) guidelines. Nevertheless, although the resuscitation bolus consisted of 0.9% saline in all our DKA patients, subsequent rehydration in most patients was carried out with balanced salt solution (Optilyte) with the initial maintenance rate of fluid administration as indicated in the guidelines [23].
The biochemical criteria for the diagnosis of DKA were.
-
Hyperglycaemia (blood glucose > 11 mmol/L [200 mg/dL])
-
Venous pH < 7.3 or serum bicarbonate < 15 mmol/L
-
Ketonemia or ketonuria [23]
The severity of DKA was categorised by the degree of acidosis:
-
Mild — venous pH < 7.3 or serum bicarbonate < 15 mmol/L
-
Moderate — pH < 7.2 or serum bicarbonate < 10 mmol/L
-
Severe — pH < 7.1 or serum bicarbonate < 5 mmol/L [23]
AKI was defined according to the KDIGO Clinical Practice Guideline by any of the following:
-
Increase in serum creatinine by ≥ 0.3 mg/dL within 48 h
-
Increase in serum creatinine to ≥ 1.5 times baseline, known or presumed to have occurred within the prior 7 days
-
Urine volume < 0.5 mL/kg/h for 6 h [24]
In acute complications of diabetes, the full age spectrum (FAS) equation (eGFRFAS) was used for estimating the glomerular filtration rate [25]. eGFRFAS was calculated using the following formula:
- SCr:
-
serum creatinine
- Q:
-
values [= median serum creatinine in mg/dL] for the FAS equation, according to height-specific healthy populations
Because no study participants had available baseline serum creatinine values before admission at the onset of diabetes or acute complications, we used an estimated GFR of 120 mL/min/1.73 m2 to calculate an expected baseline creatinine level (EBC). A GFR of 120 mL/min/1.73 m2 was selected based on previously established standards in paediatric AKI studies [26, 27]. Stage 1 AKI occurred if a creatinine value was 1.5 times to less than two times the EBC, stage 2 AKI occurred if a creatinine value was two to less than three times the EBC, and stage 3 AKI occurred if a creatinine value was three times the EBC. KDIGO AKI UO criteria were not used because the recording of hourly UO rates was inconsistent among cases.
During planned medical control after a minimum of 5 years of diabetes duration, the patients were divided into the following groups:
-
0 — patients who had never had DKA or AKI
-
DKA1 — patients who had a single episode of DKA without AKI
-
AKI1 — patients who had a single episode of AKI with or without DKA
-
DKAM — patients who had multiple (two or more) episodes of DKA, including episodes of DKA complicated with AKI
The urinary albumin excretion (UAE), cystatin C, glycosylated haemoglobin (HbA1c) total cholesterol, triglycerides and serum uric acid (UA) were collected. UAE was assessed by 24-h urine collection; kidney function was estimated by glomerular filtration rate (eGFR) according to the Filler formula based on cystatin C [28, 29]; long-term glycaemic control was based on haemoglobin A1c (HbA1c) levels [30]; serum glucose, serum creatinine in blood samples and the examination of albuminuria were measured by an automated biochemical analyser, Alinity c (ABBOTT, USA); WBCs were measured by a blood routine analyser XN-1000 (SYSMEX, Japan); pH was tested in arterial blood by a blood gas analyser ABL 835 (RADIOMETER, Denmark), and HbA1c was measured by glycosylated haemoglobin analyser Alinity c (ABBOTT, USA).
Statistical analysis
Statistical analysis was performed using Statistica ver. 8 (StatSoft, Tulsa, OK) and MedCalc. The statistical analysis results of the studied parameters were normally distributed and expressed as the mean ± standard deviation (SD). Continuous variables were tested using the analysis of variance Scheffe post hoc tests. The level of statistical significance was p < 0.05.
Results
Analysis of acute complications of diabetes (Tables 1 and 2; Fig. 2)
Retrospective analysis revealed 234 hospitalisations due to acute complications of diabetes. Among them were 98 cases of hyperglycaemia without DKA or AKI.
Diabetic ketoacidosis was diagnosed in 127 cases (54% hospitalisations). In 18 cases, it was complicated with AKI (AKIDKA; 14% of cases patients were hospitalised for DKA). In 109 cases of DKA, an episode of AKI occurred.
AKI was diagnosed in 27 cases in total. Apart from 18 episodes of AKI in DKA, nine episodes of AKI occurred in children with hyperglycaemia without DKA (AKIN; 8% of cases hospitalised for hyperglycaemic events). The highest prevalence of AKI was noted in patients at the onset of diabetes (22/27 AKI in total). All AKI cases were diagnosed on admission to the hospital. In 21 cases, AKI was in stage 1, and in six cases, it was in stage 2. None were treated in the paediatric intensive care unit (PICU). The presence of AKI did not influence PICU admission. All AKI cases were resolved during the first week of hospitalisation.
Analysis of biochemical parameters during acute complications revealed that patients with AKI showed a significantly increased concentration of corrected sodium (141.23 ± 5.09 mmol/L) when compared to patients who only had DKA. Patients with DKA or AKI showed a significant increase in WBC when compared to group N. The seriousness of acidosis based on pH was comparable in the AKI and DKA groups, whereas the concentration of HCO3 was even increased in the AKI group.
Interestingly, the comparison of children with AKI revealed that patients with AKIDKA showed significantly higher WBC levels than children with AKIN. Group AKIN presented the highest mean concentration of glucose, but it was not significant (Table 2).
Analysis of chronic complications of diabetes (Table 3; Fig. 3)
UAE, GFRF, HbA1c and diabetes duration among groups in children after 5 years’ duration of diabetes. Group 0 — patients who had never had DKA; DKA1 — patients with a single episode of DKA during diabetes duration without AKI; AKI1 — patients with a single episode of DKA complicated by AKI; DKAM — patients who had multiple (two or more) episodes of DKA during diabetes duration
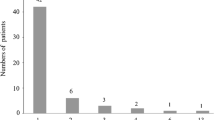
Among the 197 patients with diabetes, 91 had DKA (46%). A single episode of DKA was diagnosed in 73 patients (37%), usually at the onset of the disease. In 63 patients, it was a single episode of DKA without AKI (group DKA1).
In 18 cases (group AKI1), a single episode of AKI was diagnosed (in 10 cases during DKA and in eight cases during severe hyperglycaemia).
Two or more episodes of DKA were found in 18 patients (9%, group DKAM). In group DKAM there were 54 episodes of DKA in total, including nine episodes of AKI in eight patients. A girl who had seven episodes of DKA showed two episodes of AKI.
In 98 patients (49%), no episodes of DKA or AKI occurred (group 0).
The UAE was significantly increased in group DKAM (44.20 ± 64.21 mg/24 h) compared to groups 0, DKA1, and AKI1. The AKI1 subgroup showed an increase in UAE levels, but it was not significant.
The eGFR did not differ much among the studied groups. However, the highest values were in DKAM. Group DKAM had the worst glycaemic control and the highest levels of lipids (total cholesterol and triglycerides). The duration of diabetes was significantly increased only in AKI1, and these patients were the youngest group at the onset of diabetes.
Discussion
This study revealed that among all acute complications of diabetes, around half of the patients had DKA, mostly a single episode. It usually occurred at the onset of the disease. This is consistent with other reports, which show frequencies of DKA at the beginning of diabetes from approximately 15 to 70%, depending on the age at diagnosis, origin, ethnicity, and access to medical care. The risk of DKA in established type 1 diabetes is estimated to be between 1 and 10% in each patient every year, similar to the findings of our study, in which 9% of established patients showed two or more episodes of DKA [31, 32].
However, our study revealed that only 14% of children hospitalised for DKA developed AKI. This is much less than what other authors have reported. Hursh et al. presented that 64.2% of children hospitalised for DKA had AKI, mostly stage 2 [15]. Baalaaji et al. showed AKI in 35.4% of children with DKA admitted to a single PICU. In contrast, Myers et al. showed that 43.0% of children with DKA had AKI (1359 episodes) [33, 34]. In a recently published study by Al Khalifah et al., the AKI incidence reached 80.75% of all children with DKA. However, none of these studies investigated the development of AKI during hyperglycaemia without ketoacidosis.
Baalaaji used the pRIFLE classification based on decreased estimated creatinine clearance [27]. Using the Schwartz formula, Hursh, Myers and Al Khalifah used serum creatinine measurements using the KDIGO criteria and eGFR of 120 mL/min/1.73 m2 to calculate an expected baseline creatinine level. In this study, we used the FAS formula as it has more validity for patients with different heights or ages. Nevertheless, the percentage of AKI in DKA was still low compared to other studies.
In the present study, all patients with AKI showed a significantly increased corrected sodium, considered a good indicator of dehydration in diabetes. In many studies, the corrected sodium was increased in AKI patients and usually correlated with the severity of AKI. All studies analysed AKI in diabetic ketoacidosis [15, 16, 21, 33, 34].
Interestingly, our study revealed that AKI can also occur in children without DKA during severe hyperglycaemia, and it represents 8% of cases of hyperglycaemic events. All AKIN episodes were established at the onset of diabetes and were in stage 1. These children presented the highest mean levels of glucose. In the setting of diabetes, as shown by others, the extracellular volume depletion and pre-renal AKI are commonly induced by glucosuria and osmotic diuresis because of poorly controlled diabetes [15, 35]. Severe hyperglycaemia seems to be enough to cause AKI.
In our study, children with AKI during DKA above increased corrected sodium also showed the highest WBC concentration (20.73 ± 8.71 × 103/µL). An increased WBC was noticed in an earlier study in diabetic adults, in which AKI patients showed a WBC of 16.51 × 103/µL vs. 9.38 × 103/µL in DKA without AKI [36].
Leucocytosis is very common in hyperglycaemic crises, but its origin is still unknown [23]. It seems to respond to metabolic stress during DKA without apparent infection [37]. Significant dehydration, haemoconcentration and hyperglycaemia lead to the release of catecholamine and cortisol from adrenal glands, increasing leucocyte levels [38,39,40]. Moreover, a lack of insulin and a lack of its possible anti-inflammatory effect can stimulate the production of neutrophils in bone marrow [41].
In addition to increased WBC, the elevation of cytokines such as TNF-α and IL-6 can also occur in DKA [42]. These cytokines, along with IL-1β, regulate the production of acute-phase proteins [43]. In DKA, increased reactive oxygen species production leads to increased cytokine levels and the emergence of growth factor receptors [44]. Cytokines released during DKA may result in capillary perturbation and thus may contribute to developing acute clinical complications (i.e., cerebral or pulmonary oedema). The pathophysiology of these complications remains uncertain, but they likely involve some capillary perturbation that begins before the management of DKA and is accentuated by it [45].
In other words, increased WBC may reflect the severity of stress and inflammatory activation during DKA, resulting in capillary perturbation and AKI. The combination of poor glycaemic control with pre-renal AKI and inflammatory activation during DKA can intensify AKI in DKA. In our study, the most severe AKI (stage 2) was diagnosed only in patients with DKA.
AKI is an independent factor associated with more extended hospital stays and a higher mortality rate for children [46]. However, AKI is also associated with long-term health outcomes. In a review that included 13 cohort studies of adults with AKI, a single episode of AKI was associated with an increased risk of developing CKD, with a pooled adjusted hazard ratio of 8.8 (95% CI, 2.1–25.5) [11].
In the present study, children with AKI (groups AKI1 and DKAM) showed the highest concentration of albuminuria during planned medical control. Moreover, in group DKAM, changes in eGFR were noticed and were highest, reflecting the tendency to hyperfiltration, which is often the initial sign of DKD [4].
The development of DKD is associated with many alterations in the structure of multiple kidney compartments, which can start very early, even within 1.5–2 years of diabetes diagnosis. It is paralleled by capillary and tubular basement membrane thickening. Other glomerular changes include the loss of endothelial fenestrations, mesangial matrix expansion and loss of podocytes with effacement of foot processes. The longer the duration of diabetes, the higher the risk of DKD [4]. In the present study, the mean diabetes duration was 8 years. However, only patients with AKI or more than two episodes of DKA with poor glycaemic control showed changes in UAE. This emphasises that AKI and repeated episodes of DKA with poor glycaemic control are an essential risk for chronic kidney injury. In particular, the AKI1 group had the longest duration of diabetes (10.60 ± 2.61 years). This finding highlights that the duration of diabetes is an important risk factor for CKD.
Interestingly, the DKAM group showed the highest increase in HbA1c and lipids compared to other groups. In other words, children with repeated DKA showed the worst glycaemic control with the highest risk of CKD and progression to DKD. The AKI incidence in this study might be an underestimation because it is possible that in the DKAM group, there were more episodes of AKI than were diagnosed.
A limitation of this study is the relatively small subgroup with AKI and a short follow-up period. It was also a single-centre study, and further cohort studies are needed to clarify the impact of AKI and DKA on DKD in children.
Conclusions
Acute complications of diabetes mellitus are risk factors for AKI. This can occur in children with DKA and those with hyperglycaemia without ketoacidosis. AKI incidences in diabetes are associated with volume depletion reflected by corrected sodium concentration. In children with DKA, AKI incidences seem to be complicated by stress and inflammation activation, reflected by increased WBC. AKI and repeated DKA with poor glycaemic control can magnify CKD and progression to DKD. Prospective longitudinal studies are needed to better understand the risk factors and long-term implications of AKI and DKA in children with diabetes.
References
Alicic RZ, Rooney MT, Tuttle KR (2017) Diabetic kidney disease: challenges, progress, and possibilities. Clin J Am Soc Nephrol 12:2032–2045. https://doi.org/10.2215/CJN.11491116
Afkarian M (2015) Diabetic kidney disease in children and adolescents. Pediatr Nephrol 30:65–74; quiz 70–71. https://doi.org/10.1007/s00467-014-2796-5
Pulgaron ER, Delamater AM (2014) Obesity and type 2 diabetes in children: epidemiology and treatment. Curr Diab Rep 14:508. https://doi.org/10.1007/s11892-014-0508-y
Kidney Disease: Improving Global Outcomes (2013) KDIGO 2012 clinical practice guideline for the evaluation and management of chronic kidney disease. Kidney Int Suppl 3:1–163
Makris K, Spanou L (2016) Acute kidney injury: definition, pathophysiology and clinical phenotypes. Clin Biochem Rev 37:85–98
Harjutsalo V, Groop PH (2014) Epidemiology and risk factors for diabetic kidney disease. Adv Chronic Kidney Dis 21:260–266. https://doi.org/10.1053/j.ackd.2014.03.009
Duzova A, Bakkaloglu A, Kalyoncu M, Poyrazoglu H, Delibas A, Ozkaya O, Peru H, Alpay H, Soylemezoglu O, Gur-Guven A, Bak M, Bircan Z, Cengiz N, Akil I, Ozcakar B, Uncu N, Karabay-Bayazit A, Sonmez F; Turkish Society for Pediatric Nephrology Acute Kidney Injury Study Group (2010) Etiology and outcome of acute kidney injury in children. Pediatr Nephrol 25:1453–1461. https://doi.org/10.1007/s00467-010-1541-y
Alkandari O, Eddington KA, Hyder A, Gauvin F, Ducruet T, Gottesman R, Phan V, Zappitelli M (2011) Acute kidney injury is an independent risk factor for pediatric intensive care unit mortality, a longer length of stay and prolonged mechanical ventilation in critically ill children: a two-center retrospective cohort study. Crit Care 15:R146–R157. https://doi.org/10.1186/cc10269
Schneider J, Khemani R, Grushkin C, Bart R (2010) Serum creatinine as stratified in the RIFLE score for acute kidney injury is associated with mortality and length of stay for children in the pediatric intensive care unit. Crit Care Med 38:933–939. https://doi.org/10.1097/CCM.0b013e3181cd12e1
Askenazi DJ, Feig DI, Graham NM, Hui-Stickle S, Goldstein SL (2006) 3–5 year longitudinal follow-up of pediatric patients after acute renal failure. Kidney Int 69:184–189. https://doi.org/10.1038/sj.ki.5000032
Coca SG, Singanamala S, Parikh CR (2012) Chronic kidney disease after acute kidney injury: a systematic review and meta-analysis. Kidney Int 81:442–448. https://doi.org/10.1038/ki.2011.379
Piani F, Reinicke T, Borghi C, Tommerdahl KL, Cara-Fuentes G, Johnson RJ, Bjornstad P (2021) Acute kidney injury in pediatric diabetic kidney disease. Front Pediatr 9:668033. https://doi.org/10.3389/fped.2021.668033
Vallon V (2014) Do tubular changes in the diabetic kidney affect the susceptibility to acute kidney injury? Nephron Clin Pract 127:133–138. https://doi.org/10.1159/000363554
Gordillo R, Ahluwalia T, Woroniecki R (2016) Hyperglycemia and acute kidney injury in critically ill children. Int J Nephrol Renovasc Dis 9:201–204. https://doi.org/10.2147/IJNRD.S115096
Hursh BE, Ronsley R, Islam N, Mammen C, Panagiotopoulos C (2017) Acute kidney injury in children with type 1 diabetes hospitalised for diabetic ketoacidosis. JAMA Pediatr 171:e170020. https://doi.org/10.1001/jamapediatrics.2017.0020
Orban JC, Maiziere EM, Ghaddab A, Van Obberghen E, Ichai C (2014) Incidence and characteristics of acute kidney injury in severe diabetic ketoacidosis. PLoS One 9:e110925. https://doi.org/10.1371/journal.pone.0110925
Castellanos L, Tuffaha M, Koren D, Levitsky LL (2020) Management of diabetic ketoacidosis in children and adolescents with type 1 diabetes mellitus. Paediatr Drugs 22:357–367. https://doi.org/10.1007/s40272-020-00397-0
Gilbert RE (2017) Proximal tubulopathy: prime mover and key therapeutic target in diabetic kidney disease. Diabetes 66:791–800. https://doi.org/10.2337/db16-0796
Realsen J, Goettle H, Chase HP (2012) Morbidity and mortality of diabetic ketoacidosis with and without insulin pump care. Diabetes Technol Ther 14:1149–1154. https://doi.org/10.1089/dia.2012.0161
Razavi Z (2010) Frequency of ketoacidosis in newly diagnosed type 1 diabetic children. Oman Med J 25:114–117. https://doi.org/10.5001/omj.2010.31
Al Khalifah R, Al-Eyadhy A, Musibeeh N, Alshalawi A, Alanazi N, Alhboob A, Hassan G, Temsah MH, Alhaboob AAN, Raina R, Alhasan K (2022) Risk factors, outcomes, and predictors of resolution of acute kidney injury in children with diabetic ketoacidosis. Pediatr Nephrol. https://doi.org/10.1007/s00467-022-05578-2
Ricci Z, Romagnoli S (2018) Acute kidney injury: diagnosis and classification in adults and children. Contrib Nephrol 193:1–12. https://doi.org/10.1159/000484956
Wolfsdorf JI, Glaser N, Agus M, Fritsch M, Hanas R, Rewers A, Sperling MA, Codner E (2018) ISPAD Clinical Practice Consensus Guidelines 2018: diabetic ketoacidosis and the hyperglycemic hyperosmolar state. https://doi.org/10.1111/pedi.12701
KDIGO Clinical Practice Guideline for Acute Kidney Injury (2012) Volume2/Issue 1/March. Kidney Int Suppl 2:8–12. https://doi.org/10.1038/kisup.2012.7
Pottel H, Hoste L, Dubourg L, Ebert N, Schaeffner E, Eriksen BO, Melsom T, Lamb EJ, Rule AD, Turner ST, Glassock RJ, De Souza V, Selistre L, Mariat C, Martens F, Delanaye P (2016) An estimated glomerular filtration rate equation for the full age spectrum. Nephrol Dial Transplant 31:798–806. https://doi.org/10.1093/ndt/gfv454
Basu RK, Kaddourah A, Terrell T, Mottes T, Arnold P, Jacobs J, Andringa J, Goldstein SL; Prospective Pediatric AKI Research Group (ppAKI) (2015) Assessment of worldwide acute kidney injury, renal angina and epidemiology in critically ill children (AWARE): study protocol for a prospective observational study. BMC Nephrol 16:24. https://doi.org/10.1186/s12882-015-0016-6
Akcan-Arikan A, Zappitelli M, Loftis LL, Washburn KK, Jefferson LS, Goldstein SL (2007) Modified RIFLE criteria in critically ill children with acute kidney injury. Kidney Int 71:1028–1035. https://doi.org/10.1038/sj.ki.5002231
Filler G, Lepage N (2003) Should the Schwartz formula for estimation of GFR be replaced by cystatin C formula? Pediatr Nephrol 18:981–985. https://doi.org/10.1007/s00467-003-1271-5
Perrin N, Berg UB (2015) Estimated glomerular filtration rates cannot replace measured GFR in type 1 diabetes patients with hyperfiltration. Acta Paediatr 104:730–737. https://doi.org/10.1111/apa.12993
Rewers M, Pihoker C, Donaghue K, Hanas R, Swift P, Klingensmith GJ (2009) Assessment and monitoring of glycemic control in children and adolescents with diabetes. Pediatr Diabetes 10(Suppl 12):71–81. https://doi.org/10.1111/j.1399-5448.2009.00582.x
Hanas R, Lindgren F, Lindblad B (2009) A 2-yr national population study of pediatric ketoacidosis in Sweden: predisposing conditions and insulin pump use. Pediatr Diabetes 10:33–37. https://doi.org/10.1111/j.1399-5448.2008.00441.x
Maahs DM, Hermann JM, Holman N, Foster NC, Kapellen TM, Allgrove J, Schatz DA, Hofer SE, Campbell F, Steigleder-Schweiger C, Beck RW, Warner JT, Holl RW; National Paediatric Diabetes Audit and the Royal College of Paediatrics and Child Health, the DPV Initiative, and the T1D Exchange Clinic Network (2015) Rates of diabetic ketoacidosis: international comparison with 49,859 pediatric patients with type 1 diabetes from England, Wales, the U.S., Austria, and Germany. Diabetes Care 38:1876–1882.https://doi.org/10.2337/dc15-0780
Baalaaji M, Jayashree M, Nallasamy K, Singhi S, Bansal A (2018) Predictors and outcome of acute kidney injury in children with diabetic ketoacidosis. Indian Pediatr 55:311–314
Myers SR, Glaser NS, Trainor JL, Nigrovic LE, Garro A, Tzimenatos L, Quayle KS, Kwok MY, Rewers A, Stoner MJ, Schunk JE, McManemy JK, Brown KM, DePiero AD, Olsen CS, Casper TC, Ghetti S, Kuppermann N; Pediatric Emergency Care Applied Research Network (PECARN) DKA FLUID Study Group (2020) Frequency and risk factors of acute kidney injury during diabetic ketoacidosis in children and association with neurocognitive outcomes. JAMA Netw Open 3:e2025481. https://doi.org/10.1001/jamanetworkopen.2020.25481
Schrier RW (2010) ARF, AKI, or ATN? Nat Rev Nephrol 6:125. https://doi.org/10.1038/nrneph.2010.1
Chen J, Zeng H, Ouyang X, Zhu M, Huang Q, Yu W, Ling L, Lan HY, Xu A, Tang Y (2020) The incidence, risk factors, and long-term outcomes of acute kidney injury in hospitalized diabetic ketoacidosis patients. BMC Nephrol 21:48. https://doi.org/10.1186/s12882-020-1709-z
Flood RG, Chiang VW (2001) Rate and prediction of infection in children with diabetic ketoacidosis. Am J Emerg Med 19:270–273. https://doi.org/10.1053/ajem.2001.24473
Karavanaki K, Karanika E, Georga S, Bartzeliotou A, Tsouvalas M, Konstantopoulos I, Fotinou A, Papassotiriou I, Karayianni C (2011) Cytokine response to diabetic ketoacidosis (DKA) in children with type 1 diabetes (T1DM). Endocr J 58:1045–1053. https://doi.org/10.1507/endocrj.ej11-0024
Hansen TK, Thiel S, Wouters PJ, Chrstiansen JS, Vanden Berghe G (2003) Intensive insulin therapy exerts anti-inflammatory effects in critically ill patients and counteracts the adverse effect of low mannose-binding lectin levels. J Clin Endocrinol Metab 88:1082–1088. https://doi.org/10.1210/jc.2002-021478
Dandona P, Aljada A, Mohanty P (2002) The anti-inflammatory and potential antiatherogenic effect of insulin: a new paradigm. Diabetologia 45:924–930. https://doi.org/10.1007/s00125-001-0766-5
Moradi S, Kerman SR, Rohani F, Salari F (2012) Association between diabetes complications and leukocyte counts in Iranian patients. J Inflam Res 5:7–11. https://doi.org/10.2147/JIR.S26917
Goebel MU, Mills PJ, Irwin MR, Ziegler MG (2000) Interleukin-6 and tumor necrosis factor-a production after acute psychological stress, exercise and infused isoproterenol: differential effects and pathways. Psychosom Med 62:591–598. https://doi.org/10.1097/00006842-200007000-00019
Suffredini AF, Fiantazzi G, Badolto R, Oppenheimer J, O’Grady N (1999) New insights into the biology of acute phase response. J Clin Immunol 19:203–214. https://doi.org/10.1023/a:1020563913045
Kitabchi AE, Stentz FB, Umpierrez GE (2004) Diabetic ketoacidosis induces in vivo activation of T-lymphocytes. Biochem Biophys Res Commun 315:404–407. https://doi.org/10.1016/j.bbrc.2004.01.065
Hoffman WH, Burek CL, Waller JL, Fisher LE, Khichi M, Mellick LB (2003) Cytokine response to diabetic ketoacidosis and its treatment. Clin Immunol 108:175–181. https://doi.org/10.1016/s1521-6616(03)00144-x
Uber AM, Sutherland SM (2020) Acute kidney injury in hospitalized children: consequences and outcomes. Pediatr Nephrol 35:213–220. https://doi.org/10.1007/s00467-018-4128-7
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Soltysiak, J., Krzysko-Pieczka, I., Gertig-Kolasa, A. et al. Acute kidney injury and diabetic kidney disease in children with acute complications of diabetes. Pediatr Nephrol 38, 1643–1652 (2023). https://doi.org/10.1007/s00467-022-05735-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00467-022-05735-7