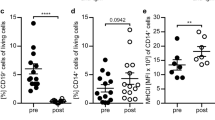
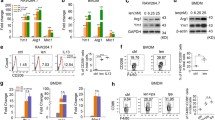
Abstract
Innate immune responses by myeloid cells decisively contribute to perpetuation of central nervous system (CNS) autoimmunity and their pharmacologic modulation represents a promising strategy to prevent disease progression in Multiple Sclerosis (MS). Based on our observation that peripheral immune cells from relapsing-remitting and primary progressive MS patients exhibited strongly decreased levels of the bile acid receptor FXR (farnesoid-X-receptor, NR1H4), we evaluated its potential relevance as therapeutic target for control of established CNS autoimmunity. Pharmacological FXR activation promoted generation of anti-inflammatory macrophages characterized by arginase-1, increased IL-10 production, and suppression of T cell responses. In mice, FXR activation ameliorated CNS autoimmunity in an IL-10-dependent fashion and even suppressed advanced clinical disease upon therapeutic administration. In analogy to rodents, pharmacological FXR activation in human monocytes from healthy controls and MS patients induced an anti-inflammatory phenotype with suppressive properties including control of effector T cell proliferation. We therefore, propose an important role of FXR in control of T cell-mediated autoimmunity by promoting anti-inflammatory macrophage responses.








Similar content being viewed by others
References
Ajami B, Bennett JL, Krieger C, McNagny KM, Rossi FM (2011) Infiltrating monocytes trigger EAE progression, but do not contribute to the resident microglia pool. Nat Neurosci 14:1142–1149. doi:10.1038/nn.2887
Ali AH, Carey EJ, Lindor KD (2015) Recent advances in the development of farnesoid X receptor agonists. Ann Transl Med 3:5. doi:10.3978/j.issn.2305-5839.2014.12.06
Allen IV (1991) Pathology of multiple sclerosis. In: Matthews WB (ed) McAlpine’s multiple sclerosis. Churchill Livingstone, Edinburgh, pp 341–378
Barnett MH, Henderson AP, Prineas JW (2006) The macrophage in MS: just a scavenger after all? Pathology and pathogenesis of the acute MS lesion. Mult Scler 12:121–132
Berer K, Mues M, Koutrolos M, Rasbi ZA, Boziki M, Johner C, Wekerle H, Krishnamoorthy G (2011) Commensal microbiota and myelin autoantigen cooperate to trigger autoimmune demyelination. Nature 479:538–541. doi:10.1038/nature10554
Bettelli E, Das MP, Howard ED, Weiner HL, Sobel RA, Kuchroo VK (1998) IL-10 is critical in the regulation of autoimmune encephalomyelitis as demonstrated by studies of IL-10- and IL-4-deficient and transgenic mice. J Immunol 161:3299–3306
Brusca SB, Abramson SB, Scher JU (2014) Microbiome and mucosal inflammation as extra-articular triggers for rheumatoid arthritis and autoimmunity. Curr Opin Rheumatol 26:101–107. doi:10.1097/BOR.0000000000000008
Butovsky O, Ziv Y, Schwartz A, Landa G, Talpalar AE, Pluchino S, Martino G, Schwartz M (2006) Microglia activated by IL-4 or IFN-gamma differentially induce neurogenesis and oligodendrogenesis from adult stem/progenitor cells. Mol Cell Neurosci 31:149–160. doi:10.1016/j.mcn.2005.10.006
Calkin AC, Tontonoz P (2012) Transcriptional integration of metabolism by the nuclear sterol-activated receptors LXR and FXR. Nat Rev Mol Cell Biol 13:213–224. doi:10.1038/nrm3312
Chawla A, Nguyen KD, Goh YP (2011) Macrophage-mediated inflammation in metabolic disease. Nat Rev Immunol 11:738–749. doi:10.1038/nri3071
Fairweather D, Cihakova D (2009) Alternatively activated macrophages in infection and autoimmunity. J Autoimmun 33:222–230. doi:10.1016/j.jaut.2009.09.012
Farez MF, Quintana FJ, Gandhi R, Izquierdo G, Lucas M, Weiner HL (2009) Toll-like receptor 2 and poly(ADP-ribose) polymerase 1 promote central nervous system neuroinflammation in progressive EAE. Nat Immunol 10:958–964. doi:10.1038/ni.1775
Fassl SK, Austermann J, Papantonopoulou O, Riemenschneider M, Xue J, Bertheloot D, Freise N, Spiekermann C, Witten A, Viemann D et al (2015) Transcriptome assessment reveals a dominant role for TLR4 in the activation of human monocytes by the alarmin MRP8. J Immunol 194:575–583. doi:10.4049/jimmunol.1401085
Gadaleta RM, Oldenburg B, Willemsen EC, Spit M, Murzilli S, Salvatore L, Klomp LW, Siersema PD, van Erpecum KJ, van Mil SW (2011) Activation of bile salt nuclear receptor FXR is repressed by pro-inflammatory cytokines activating NF-kappaB signaling in the intestine. Biochim Biophys Acta 1812:851–858. doi:10.1016/j.bbadis.2011.04.005
Gadaleta RM, van Erpecum KJ, Oldenburg B, Willemsen EC, Renooij W, Murzilli S, Klomp LW, Siersema PD, Schipper ME, Danese S et al (2011) Farnesoid X receptor activation inhibits inflammation and preserves the intestinal barrier in inflammatory bowel disease. Gut 60:463–472. doi:10.1136/gut.2010.212159
Goodwin B, Jones SA, Price RR, Watson MA, McKee DD, Moore LB, Galardi C, Wilson JG, Lewis MC, Roth ME et al (2000) A regulatory cascade of the nuclear receptors FXR, SHP-1, and LRH-1 represses bile acid biosynthesis. Mol Cell 6:517–526
Gregori S, Tomasoni D, Pacciani V, Scirpoli M, Battaglia M, Magnani CF, Hauben E, Roncarolo MG (2010) Differentiation of type 1 T regulatory cells (Tr1) by tolerogenic DC-10 requires the IL-10-dependent ILT4/HLA-G pathway. Blood 116:935–944. doi:10.1182/blood-2009-07-234872
Hendriks JJ, Teunissen CE, de Vries HE, Dijkstra CD (2005) Macrophages and neurodegeneration. Brain Res Brain Res Rev 48:185–195. doi:10.1016/j.brainresrev.2004.12.008
Ho PP, Steinman L (2016) Obeticholic acid, a synthetic bile acid agonist of the farnesoid X receptor, attenuates experimental autoimmune encephalomyelitis. Proc Natl Acad Sci USA 113:1600–1605. doi:10.1073/pnas.1524890113
Hsu P, Santner-Nanan B, Hu M, Skarratt K, Lee CH, Stormon M, Wong M, Fuller SJ, Nanan R (2015) IL-10 Potentiates Differentiation of Human Induced Regulatory T Cells via STAT3 and Foxo1. J Immunol 195:3665–3674. doi:10.4049/jimmunol.1402898
Hucke S, Eschborn M, Liebmann M, Herold M, Freise N, Engbers A, Ehling P, Meuth SG, Roth J, Kuhlmann T et al (2016) Sodium chloride promotes pro-inflammatory macrophage polarization thereby aggravating CNS autoimmunity. J Autoimmun 67:90–101. doi:10.1016/j.jaut.2015.11.001
Hucke S, Flossdorf J, Grutzke B, Dunay IR, Frenzel K, Jungverdorben J, Linnartz B, Mack M, Peitz M, Brustle O et al (2012) Licensing of myeloid cells promotes central nervous system autoimmunity and is controlled by peroxisome proliferator-activated receptor gamma. Brain J Neurol 135:1586–1605. doi:10.1093/brain/aws058
Hucke S, Wiendl H, Knolle P, Klotz L (2013) Nuclear receptor control of myeloid cell responses—implications for CNS autoimmunity. Rheumatol Curr Res 3:1–12. doi:10.4172/2161-1149.1000124
Huitinga I, van Rooijen N, de Groot CJ, Uitdehaag BM, Dijkstra CD (1990) Suppression of experimental allergic encephalomyelitis in Lewis rats after elimination of macrophages. J Exp Med 172:1025–1033
International Multiple Sclerosis Genetics C, Beecham AH, Patsopoulos NA, Xifara DK, Davis MF, Kemppinen A, Cotsapas C, Shah TS, Spencer C, Booth D et al (2013) Analysis of immune-related loci identifies 48 new susceptibility variants for multiple sclerosis. Nature Gen 45:1353–1360. doi:10.1038/ng.2770
Jiang Z, Jiang JX, Zhang GX (2014) Macrophages: a double-edged sword in experimental autoimmune encephalomyelitis. Immunol Lett 160:17–22. doi:10.1016/j.imlet.2014.03.006
Klotz L, Burgdorf S, Dani I, Saijo K, Flossdorf J, Hucke S, Alferink J, Nowak N, Beyer M, Mayer G et al (2009) The nuclear receptor PPAR gamma selectively inhibits Th17 differentiation in a T cell-intrinsic fashion and suppresses CNS autoimmunity. J Exp Med 206:2079–2089. doi:10.1084/jem.20082771
Klotz L, Dani I, Edenhofer F, Nolden L, Evert B, Paul B, Kolanus W, Klockgether T, Knolle P, Diehl L (2007) Peroxisome proliferator-activated receptor gamma control of dendritic cell function contributes to development of CD4 + T cell anergy. J Immunol 178:2122–2131
Klotz L, Hucke S, Thimm D, Classen S, Gaarz A, Schultze J, Edenhofer F, Kurts C, Klockgether T, Limmer A et al (2009) Increased antigen cross-presentation but impaired cross-priming after activation of peroxisome proliferator-activated receptor gamma is mediated by up-regulation of B7H1. J Immunol 183:129–136. doi:10.4049/jimmunol.0804260
Klotz L, Schmidt M, Giese T, Sastre M, Knolle P, Klockgether T, Heneka MT (2005) Proinflammatory stimulation and pioglitazone treatment regulate peroxisome proliferator-activated receptor gamma levels in peripheral blood mononuclear cells from healthy controls and multiple sclerosis patients. J Immunol 175:4948–4955
Lassmann H (1998) Pathology of multiple sclerosis. In: Compston A, Ebers G, Lassmann H, McDonald I, Matthews B, Wekerle H (eds) McAlpinès multiple sclerosis, 3rd edn. Churchill Livingstone, London, pp 323–358
Lassmann H (2004) Recent neuropathological findings in MS—implications for diagnosis and therapy. J Neurol 251(Suppl 4):IV2–IV5. doi:10.1007/s00415-004-1402-3
Lee YK, Menezes JS, Umesaki Y, Mazmanian SK (2011) Proinflammatory T-cell responses to gut microbiota promote experimental autoimmune encephalomyelitis. Proc Natl Acad Sci USA 108(Suppl 1):4615–4622. doi:10.1073/pnas.1000082107
Lefebvre P, Cariou B, Lien F, Kuipers F, Staels B (2009) Role of bile acids and bile acid receptors in metabolic regulation. Physiol Rev 89:147–191. doi:10.1152/physrev.00010.2008
Lian F, Wang Y, Chen J, Xu H, Yang X, Liang L, Zhan Z, Ye Y, Chen M (2012) Activation of farnesoid X receptor attenuates liver injury in systemic lupus erythematosus. Rheumatol Int 32:1705–1710. doi:10.1007/s00296-011-1874-2
Liu C, Li Y, Yu J, Feng L, Hou S, Liu Y, Guo M, Xie Y, Meng J, Zhang H et al (2013) Targeting the shift from M1 to M2 macrophages in experimental autoimmune encephalomyelitis mice treated with fasudil. PLoS One 8:e54841. doi:10.1371/journal.pone.0054841
Melief SM, Schrama E, Brugman MH, Tiemessen MM, Hoogduijn MJ, Fibbe WE, Roelofs H (2013) Multipotent stromal cells induce human regulatory T cells through a novel pathway involving skewing of monocytes toward anti-inflammatory macrophages. Stem Cells 31:1980–1991. doi:10.1002/stem.1432
Mencarelli A, Renga B, Migliorati M, Cipriani S, Distrutti E, Santucci L, Fiorucci S (2009) The bile acid sensor farnesoid X receptor is a modulator of liver immunity in a rodent model of acute hepatitis. J Immunol 183:6657–6666. doi:10.4049/jimmunol.0901347
Mikita J, Dubourdieu-Cassagno N, Deloire MS, Vekris A, Biran M, Raffard G, Brochet B, Canron MH, Franconi JM, Boiziau C et al (2011) Altered M1/M2 activation patterns of monocytes in severe relapsing experimental rat model of multiple sclerosis. Amelioration of clinical status by M2 activated monocyte administration. Mult Scler 17:2–15. doi:10.1177/1352458510379243
Mildner A, Mack M, Schmidt H, Bruck W, Djukic M, Zabel MD, Hille A, Priller J, Prinz M (2009) CCR2 + Ly-6Chi monocytes are crucial for the effector phase of autoimmunity in the central nervous system. Brain J Neurol 132:2487–2500. doi:10.1093/brain/awp144
Modica S, Gadaleta RM, Moschetta A (2010) Deciphering the nuclear bile acid receptor FXR paradigm. Nucl Recept Signal 8:e005. doi:10.1621/nrs.08005
Moreno M, Bannerman P, Ma J, Guo F, Miers L, Soulika AM, Pleasure D (2014) Conditional ablation of astroglial CCL2 suppresses CNS accumulation of M1 macrophages and preserves axons in mice with MOG peptide EAE. J Neurosci Off J Soc Neurosci 34:8175–8185. doi:10.1523/JNEUROSCI.1137-14.2014
Mosser DM (2003) The many faces of macrophage activation. J Leukoc Biol 73:209–212
Mosser DM, Edwards JP (2008) Exploring the full spectrum of macrophage activation. Nat Rev Immunol 8:958–969. doi:10.1038/nri2448
Mudaliar S, Henry RR, Sanyal AJ, Morrow L, Marschall HU, Kipnes M, Adorini L, Sciacca CI, Clopton P, Castelloe E et al (2013) Efficacy and safety of the farnesoid X receptor agonist obeticholic acid in patients with type 2 diabetes and nonalcoholic fatty liver disease. Gastroenterology 145(574–582):e571. doi:10.1053/j.gastro.2013.05.042
Murray PJ, Allen JE, Biswas SK, Fisher EA, Gilroy DW, Goerdt S, Gordon S, Hamilton JA, Ivashkiv LB, Lawrence T et al (2014) Macrophage activation and polarization: nomenclature and experimental guidelines. Immunity 41:14–20. doi:10.1016/j.immuni.2014.06.008
Murray PJ, Wynn TA (2011) Protective and pathogenic functions of macrophage subsets. Nat Rev Immunol 11:723–737. doi:10.1038/nri3073
Neuschwander-Tetri BA, Loomba R, Sanyal AJ, Lavine JE, Van Natta ML, Abdelmalek MF, Chalasani N, Dasarathy S, Diehl AM, Hameed B et al (2014) Farnesoid X nuclear receptor ligand obeticholic acid for non-cirrhotic, non-alcoholic steatohepatitis (FLINT): a multicentre, randomised, placebo-controlled trial. Lancet. doi 10.1016/S0140-6736(14)61933-4
Nijmeijer RM, Gadaleta RM, van Mil SW, van Bodegraven AA, Crusius JB, Dijkstra G, Hommes DW, de Jong DJ, Stokkers PC, Verspaget HW et al (2011) Farnesoid X receptor (FXR) activation and FXR genetic variation in inflammatory bowel disease. PLoS One 6:e23745. doi:10.1371/journal.pone.0023745
Nikolic I, Saksida T, Mangano K, Vujicic M, Stojanovic I, Nicoletti F, Stosic-Grujicic S (2014) Pharmacological application of carbon monoxide ameliorates islet-directed autoimmunity in mice via anti-inflammatory and anti-apoptotic effects. Diabetologia 57:980–990. doi:10.1007/s00125-014-3170-7
Porcheray F, Viaud S, Rimaniol AC, Leone C, Samah B, Dereuddre-Bosquet N, Dormont D, Gras G (2005) Macrophage activation switching: an asset for the resolution of inflammation. Clin Exp Immunol 142:481–489. doi:10.1111/j.1365-2249.2005.02934.x
Prineas JW (1985) The neuropathology of multiple sclerosis. In: Koetsier JC (ed) Demyelinating diseases. Elsevier Science Ltd., Amsterdam, pp 213–257
Prineas JW, Graham JS (1981) Multiple sclerosis: capping of surface immunoglobulin G on macrophages engaged in myelin breakdown. Ann Neurol 10:149–158. doi:10.1002/ana.410100205
Renga B, Mencarelli A, Cipriani S, D’Amore C, Carino A, Bruno A, Francisci D, Zampella A, Distrutti E, Fiorucci S (2013) The bile acid sensor FXR is required for immune-regulatory activities of TLR-9 in intestinal inflammation. PLoS One 8:e54472. doi:10.1371/journal.pone.0054472
Renga B, Migliorati M, Mencarelli A, Fiorucci S (2009) Reciprocal regulation of the bile acid-activated receptor FXR and the interferon-gamma-STAT-1 pathway in macrophages. Biochim Biophys Acta 1792:564–573. doi:10.1016/j.bbadis.2009.04.004
Schote AB, Turner JD, Schiltz J, Muller CP (2007) Nuclear receptors in human immune cells: expression and correlations. Mol Immunol 44:1436–1445. doi:10.1016/j.molimm.2006.04.021
Schwab N, Schneider-Hohendorf T, Posevitz V, Breuer J, Gobel K, Windhagen S, Brochet B, Vermersch P, Lebrun-Frenay C, Posevitz-Fejfar A et al (2013) L-selectin is a possible biomarker for individual PML risk in natalizumab-treated MS patients. Neurology 81:865–871. doi:10.1212/WNL.0b013e3182a351fb
Serbina NV, Pamer EG (2006) Monocyte emigration from bone marrow during bacterial infection requires signals mediated by chemokine receptor CCR2. Nat Immunol 7:311–317. doi:10.1038/ni1309
Shaik FB, Prasad DV, Narala VR (2014) Role of farnesoid X receptor in inflammation and resolution. Inflamm Res Off J Eur Histamine Res Soc [et al]. doi 10.1007/s00011-014-0780-y
Shechter R, London A, Varol C, Raposo C, Cusimano M, Yovel G, Rolls A, Mack M, Pluchino S, Martino G et al (2009) Infiltrating blood-derived macrophages are vital cells playing an anti-inflammatory role in recovery from spinal cord injury in mice. PLoS Med 6:e1000113. doi:10.1371/journal.pmed.1000113
Sica A, Mantovani A (2012) Macrophage plasticity and polarization: in vivo veritas. J Clin Investig 122:787–795. doi:10.1172/JCI59643
Sinal CJ, Tohkin M, Miyata M, Ward JM, Lambert G, Gonzalez FJ (2000) Targeted disruption of the nuclear receptor FXR/BAR impairs bile acid and lipid homeostasis. Cell 102:731–744
Tierney JB, Kharkrang M, La Flamme AC (2009) Type II-activated macrophages suppress the development of experimental autoimmune encephalomyelitis. Immunol Cell Biol 87:235–240. doi:10.1038/icb.2008.99
Trinschek B, Luessi F, Haas J, Wildemann B, Zipp F, Wiendl H, Becker C, Jonuleit H (2013) Kinetics of IL-6 production defines T effector cell responsiveness to regulatory T cells in multiple sclerosis. PLoS One 8:e77634. doi:10.1371/journal.pone.0077634
Trivedi PJ, Hirschfield GM, Gershwin ME (2016) Obeticholic acid for the treatment of primary biliary cirrhosis. Exp Rev Clin Pharmacol 9:13–26. doi:10.1586/17512433.2015.1092381
van Schaik FD, Gadaleta RM, Schaap FG, van Mil SW, Siersema PD, Oldenburg B, van Erpecum KJ (2012) Pharmacological activation of the bile acid nuclear farnesoid X receptor is feasible in patients with quiescent Crohn’s colitis. PLoS One 7:e49706. doi:10.1371/journal.pone.0049706
Vavassori P, Mencarelli A, Renga B, Distrutti E, Fiorucci S (2009) The bile acid receptor FXR is a modulator of intestinal innate immunity. J Immunol 183:6251–6261. doi:10.4049/jimmunol.0803978
Wang S, Gao X, Shen G, Wang W, Li J, Zhao J, Wei YQ, Edwards CK (2016) Interleukin-10 deficiency impairs regulatory T cell-derived neuropilin-1 functions and promotes Th1 and Th17 immunity. Sci Rep 6:24249. doi:10.1038/srep24249
Wang Y, Begum-Haque S, Telesford KM, Ochoa-Reparaz J, Christy M, Kasper EJ, Kasper DL, Robson SC, Kasper LH (2014) A commensal bacterial product elicits and modulates migratory capacity of CD39(+) CD4 T regulatory subsets in the suppression of neuroinflammation. Gut Microbes 5:552–561. doi:10.4161/gmic.29797
Weber MS, Prod’homme T, Youssef S, Dunn SE, Rundle CD, Lee L, Patarroyo JC, Stuve O, Sobel RA, Steinman L et al (2007) Type II monocytes modulate T cell-mediated central nervous system autoimmune disease. Nat Med 13:935–943. doi:10.1038/nm1620
Wu M, Nissen JC, Chen EI, Tsirka SE (2012) Tuftsin promotes an anti-inflammatory switch and attenuates symptoms in experimental autoimmune encephalomyelitis. PLoS One 7:e34933. doi:10.1371/journal.pone.0034933
Xu Z, Huang G, Gong W, Zhou P, Zhao Y, Zhang Y, Zeng Y, Gao M, Pan Z, He F (2012) FXR ligands protect against hepatocellular inflammation via SOCS3 induction. Cell Signal 24:1658–1664. doi:10.1016/j.cellsig.2012.04.015
Ye Z, Huang H, Hao S, Xu S, Yu H, Van Den Hurk S, Xiang J (2007) IL-10 has a distinct immunoregulatory effect on naive and active T cell subsets. J interf Cytokine Res Off J Int Soc Interf Cytokine Res 27:1031–1038. doi:10.1089/jir.2006.0144
Zhang H, Liu Y, Bian Z, Huang S, Han X, You Z, Wang Q, Qiu D, Miao Q, Peng Y et al (2014) The critical role of myeloid-derived suppressor cells and FXR activation in immune-mediated liver injury. J Autoimmun 53:55–66. doi:10.1016/j.jaut.2014.02.010
Zhu W, Yu J, Nie Y, Shi X, Liu Y, Li F, Zhang XL (2014) Disequilibrium of M1 and M2 macrophages correlates with the development of experimental inflammatory bowel diseases. Immunol Invest 43:638–652. doi:10.3109/08820139.2014.909456
Acknowledgments
We thank Annika Engbers, Andrea Pabst and Corinna Hölscher for excellent technical support as well as Anita Posevitz-Fejfar and Verena Schütte for bio-banking of human PBMC samples. We thank Heike Blum for creating an excellent illustration (Fig. 8). We thank M.E. Schwab and H.-J. Radzun for providing antibodies against NogoA and KiM1P to perform histological analysis. This study was supported by the German Research Foundation Grant Number CRC 128 A8 to LK, CRC 128 Z1 to TK, and CRC 128 Z2 to HW,FZ, BH and the Interdisciplinary center for clinical research (IZKF) Grant Number Kl2/015/14 to LK.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing financial interests. MH, ML, NF, ML, AF, SZ, ST, JF, DRE, BH, JR have nothing to declare. SH received speaker honoraria from Novartis. DB received compensation for activities with Bayer HealthCare, BiogenIdec, MerckSerono, and Novartis, she was supported by the Abirisk and the PML Consortium. FL received travel grants from Teva Pharmaceuticals and Merck Serono. SGM received honoraria for lecturing and travel expenses for attending meetings and financial research support from Bayer, Bayer Schering, Biogen Idec, Genzyme, Merck Serono, MSD, Novartis, Omniamed, Novo Nordisk, Sanofi-Aventis and Teva. FZ has received research grants from Teva, Merck Serono, Novartis and Bayer as well as consultation funds from Teva, Merck Serono, Novartis, Bayer Healthcare, Biogen Idec Germany, ONO, Genzyme, Sanofi-Aventis and Octapharma, her travel compensation has been provided for by the aforementioned companies. TK received speaker honoraria from Novartis, Biogen Idec Canada,and Teva; she received compensation as a consultant from Genzyme. HW received compensation for serving on Scientific Advisory Boards/Steering Committees for Bayer Healthcare, Biogen Idec, Genzyme, Merck Serono, Novartis and Sanofi-Aventis; he received speaker honoraria and travel support from Bayer Vital GmbH, Bayer Schering AG, Biogen Idec, CSL Behring, Fresenius Medical Care, Genzyme, Glaxo Smith Kline, GW Pharmaceuticals, Lundbeck, Merck Serono, Omniamed, Novartis and Sanofi-Aventis; he received compensation as a consultant from Biogen Idec, Merck Serono, Novartis and Sanofi-Aventis, has received research support from Bayer Vital, Biogen Idec, Genzyme Merck Serono, Novartis, Sanofi-Aventis Germany, Sanofi US. LK received compensation for serving on Scientific Advisory Boards for Genzyme and Novartis; she received speaker honoraria and travel support from Novartis, Merck Serono and CSL Behring; she receives research support from Novartis and Biogen Idec.
Additional information
S. Hucke and M. Herold contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
401_2016_1593_MOESM1_ESM.jpg
Supplementary Figure 1. FXR expression in peripheral immune cells is reduced in CNS autoimmunity. a-d Nuclear receptor (NR) expression in human and murine immune cells was determined by quantitative real-time RT-PCR. Data were normalized to endogenous 18s expression and are displayed as relative expression compared to control group, which was set to 1. Shown are boxplots where each box displays the upper and lower quartiles of the respective distribution, median (line), and mean (+). Box whiskers represent the maximum and minimum range. a FXR mRNA expression in HD and MS patients from cohort 1 split according to treatment allocation, i.e., untreated, first-line immune-modulatory treatment, or escalation therapy. b+c Equal 18s expression by PBMCs from HD and MS patients demonstrates comparable RNA quality. Expression of NRs in b cohort 1 (n=14 healthy donors (HD), n=30 MS patients (RRMS)) and c cohort 2 (n=15 HD, n=15 MS patients (RRMS)). d Expression of the indicated NRs was analyzed in splenocytes from EAE-diseased mice (mean clinical score of 3.4±0.62, n=15) compared to splenocytes from healthy C57BL/6 mice. (n=16 per group) *p<0.05; **p<0.01; ***p<0.001. (JPEG 2551 kb)
401_2016_1593_MOESM2_ESM.jpg
Supplementary Figure 2. FXR activation does not generally alter cytokine production of lymph node T cells during EAE. EAE was performed as described in Fig. 2c. On day 15 of EAE, lymph node cells were ex vivo restimulated with αCD3 for 48h. Then, cytokine production in supernatants was analyzed by Luminex® Multiplex Assay. Shown are boxplots where each box displays the upper and lower quartiles of the respective distribution and median (line). Box whiskers represent the maximum and minimum range. (n=8 per group) (JPEG 1507 kb)
401_2016_1593_MOESM3_ESM.jpg
Supplementary Figure 3. In vivo FXR activation induces expression of FXR target genes but does not alter composition and phenotype of myeloid cell populations in gut and intestine. a+b EAE was performed as described in Fig. 2b. a On day 15, liver and intestine of EAE-diseased mice treated with GW4064 in 0.5 % CMC or vehicle only (DMSO in 0.5 % CMC) were collected and expression of FXR target genes ApoE and SHP was analyzed by quantitative real-time RT-PCR. Data were normalized to endogenous 18s expression and are displayed as mean relative expression compared to DMSO-treated EAE mice ± SEM. (n=3 per group) *p<0.05; **p<0.01. b In addition, liver (n=15) and intestine (n=3) were subjected to HE staining and histological analysis. Liver and intestine exhibit normal histology, which indicates that GW4064-treatment does not exhibit toxic side effects. c–f After 7 days of oral treatment of healthy mice with GW4064 or vehicle only (w/o EAE) or on day 10 after EAE induction (EAE) in GW4064- or control-treated mice, single cell suspensions from livers c+d or intestine e+f were analyzed for composition of different myeloid cell subpopulations by flow cytometry as described in supplementary table II (n=8). Panels c (liver) and e (intestine) depict our gating strategy for myeloid cell subpopulations using the markers CD11b, Gr1, Ly-6C, Ly-6G, and F4/80 in livers c and intestines e. c-f Additionally, we assessed expression levels of the well-known activation markers MHC class II, CD40, and CD80, as well as the anti-inflammatory markers B7H1 and MR, on different myeloid cell subsets as gated in c and e, respectively. g Intestinal cell suspensions from EAE-diseased mice were further used to determine mRNA expression levels of classical M1 and M2 markers by quantitative real-time RT-PCR (n=8). Data were normalized to endogenous HPRT expression ± SEM. (JPEG 4429 kb)
401_2016_1593_MOESM4_ESM.jpg
Supplementary Figure 4. GW4064 does not affect survival and proliferation of immune cells. a+b Human CD4+ T cells were isolated from the peripheral blood of healthy donors (n=3). T cells were treated with 15 μM GW4064 and stimulated in the presence of αCD3 and αCD28. a Survival and b proliferation were analyzed by flow cytometric analysis after 48h and 72h. a Graph depicts the mean percentage of live cells ± SEM after 48h and 72h, which were identified as Hoechst33342- demonstrating no toxic effect on T cells. b Graph depicts the mean percentage of proliferating T cells ± SEM, which is unaltered under GW4064-treatment. c Human CD14+ monocytes were isolated from the peripheral blood of healthy donors (n=3) and were treated with 15 μM GW4064. Graph depicts the mean percentage of live cells ± SEM after 48h and 72h, which were identified as Hoechst33342- demonstrating no toxic effect on monocytes. d+e Analysis of survival and proliferation of murine splenic CD4+ T cells treated with 15 μM GW4064 and stimulated in the presence of αCD3 and αCD28 was performed as in a+b. f BMMs were differentiated in the presence of 15 μM GW4064. On day 7 of differentiation, BMMs were harvested and survival was determined by flow cytometric analysis of Hoechst33342 staining after 48h and 72h of cultivation. Graph depicts the mean percentage of live cells ± SEM demonstrating no toxic effect on macrophages. (JPEG 1228 kb)
401_2016_1593_MOESM5_ESM.jpg
Supplementary Figure 5. GW4064 does not affect cytokine production by macrophages. BMMs were differentiated in the presence of 15 μM GW4064. On day 7 of differentiation, BMMs were harvested and seeded to determine cytokine production, which was analyzed by Luminex® Multiplex Assay after 48h of cultivation in the presence or absence of GW4064. Graphs display the pooled result from 4 independent experiments. Shown are boxplots where each box displays the upper and lower quartiles of the respective distribution and median (line). Box whiskers represent the maximum and minimum range. (JPEG 1016 kb)
401_2016_1593_MOESM6_ESM.jpg
Supplementary Figure 6. FXR activation in human myeloid cells represents a new therapeutic target. a CD14+ monocytes were isolated from the peripheral blood of healthy donors (HD) and treated with 15 μM GW4064. After 24h, monocytes were washed twice and subsequently cocultured with CD3+ T cells isolated from the blood of syngen or allogen healthy donors patients in the presence of 1 μg/ml anti-CD3 and 1 μg/ml anti-CD28. After 72h, proliferation was assessed by flow cytometric analysis of proliferation dye eFluor670 (see histograms Fig.7b). Graphs display the mean percentage of proliferated CD8+ T cells as well as the mean division index ± SEM. b CD14+ monocytes were isolated from the peripheral blood of healthy donors (HD) or MS patients (MS) and treated with 15 μM GW4064. After 24h, monocytes were washed twice and subsequently cocultured with CD3+ T cells isolated from the blood of allogen MS patients in the presence of 1 μg/ml anti-CD3 and 1 μg/ml anti-CD28. After 72h, proliferation was assessed by flow cytometric analysis of proliferation dye eFluor670 (see histograms Fig. 7c). Graphs display the mean percentage of proliferated CD8+ T cells as well as the mean division index ± SEM. c Upper row: Summary of all assays run in Fig. 7b+c. Lower row: In addition, the relative proliferation of CD8+ T cells was calculated by dividing the percentage of dividing cells (GW4064-treated monocyte group) / (control monocyte group). *p<0.05; **p<0.01; ***p<0.001. (JPEG 1504 kb)
401_2016_1593_MOESM10_ESM.docx
Supplementary Table IV. Primers, QuantiTect Primer Assays (Qiagen) or TaqMan probes used for real-time RT-PCR analysis (DOCX 19 kb)
Rights and permissions
About this article
Cite this article
Hucke, S., Herold, M., Liebmann, M. et al. The farnesoid-X-receptor in myeloid cells controls CNS autoimmunity in an IL-10-dependent fashion. Acta Neuropathol 132, 413–431 (2016). https://doi.org/10.1007/s00401-016-1593-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00401-016-1593-6