Abstract
Background
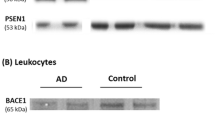
Members of membrane-bound disintegrin metalloproteinases (ADAMs) were shown to be capable of cleaving amyloid precursor protein (APP) at the α-cleavage site in different cell systems. One of the candidate α-secretases identified in this family is ADAM10. The present study addresses the following major questions: 1) Are the levels of an α-secretase candidate (i.e., ADAM10) reduced in accessible cells of Alzheimer Disease (AD) patients? 2) Are ADAM10 levels in the peripheral cells of AD patients related to a concomitant decrease in αAPPs?
Materials and Methods
Western Blot analysis of ADAM10 is performed on platelet homogenates from 33 sporadic AD patients and on 26 age-matched control subjects. Moreover, the levels of α-secretase metabolite (αAPPs) are tested both in platelets and cerebrospinal fluid (CSF) of the same pool of subjects by means of Western blot with a specific antibody.
Results
A significant decrease of platelet ADAM10 levels is observed in patients affected by probable AD when compared to control subjects and this is paralleled by a reduced level of αAPPs released from platelets. Moreover, in the same pool of AD patients, αAPPs levels were reduced concomitantly in CSF.
Conclusions
ADAM10 is expressed in platelets. A reduced level of ADAM10 is observed in platelets obtained from AD patients compared to age-matched controls. Further, in the same pool of AD patients, a qualitatively and quantitatively similar decrease in αAPPs is present both in thrombin-activated platelets and CSF, thus suggesting that alterations of APP processing might occur both in the neuronal compartment and peripheral cells.






Similar content being viewed by others
References
Sisodia SS. (1992) Beta-amyloid precursor protein cleavage by a membrane-bound protease. Proc. Natl. Acad. Sci. U.S.A. 89: 6075–6079.
Tomita S, Kirino Y, Suzuki T. (1998) Cleavage of Alzheimer’s amyloid precursor protein (APP) by secretases occurs after O-glycosylation of APP in the protein secretory pathway—Identification of intracellular compartment in which APP cleavage occurs without using toxic agents that interfere with protein metabolism. J. Biol. Chem. 273: 6277–6284.
Weidemann A, Konig G, Bunke D, et al. (1989) Identification, biogenesis, and localization of precursors of Alzheimer’s disease A4 amyloid protein. Cell 57: 115–126.
Haass C, Selkoe DJ. (1993) Cellular processing of beta-amyloid precursor protein and the genesis of amyloid beta-peptide. Cell 75: 1039–1042.
Walter J, Capell A, Hung AY, et al. (1997) Ectodomain phosphorylation of beta-amyloid precursor protein at two distinct cellular locations. J. Biol. Chem. 272: 1896–1903.
Buxbaum JD, Liu KN, Luo X, et al. (1998) Evidence that tumor necrosis factor alpha-converting enzyme is involved in regulated alpha-secretase cleavage of the Alzheimer amyloid precursor protein. J. Biol. Chem. 273: 27765–27767.
Nitsch RM, Slack BE, Wurtman RJ, et al. (1992) Release of Alzheimer amyloid precursor derivatives stimulated by activation of muscarinic acetylcholine receptors. Science 258: 304–307.
Koike H, Tomioka S, Sorimachi H, et al. (1999) Membrane-anchored metalloprotease MDC9 has an alpha-secretase activity responsible for processing the amyloid precursor protein. Biochem. J. 343: 371–375.
Nelson KK, Schlondorff J, Blobel CP. (1999) Evidence for an interaction of the metalloprotease-disintegrin tumor necrosis factor alpha convertase (TACE) with mitotic arrest deficient 2 (MAD2), and of the metalloprotease-disintegrin MDC9 with a novel MAD2-related protein, MAD2beta. Biochem. J. 343: 673–680.
Lammich S, Kojro E, Postina R, et al. (1999) Constitutive and regulated α-secretase cleavage of Alzheimer’s amyloid precursor protein by a disintegrin metalloprotease. Proc. Natl. Acad. Sci. U.S.A. 96: 3922–3927.
Kojro E, Giml G, Lammich S, Marz W, Fahrenholz F. (2001) Low cholesterol stimulates the nonamyloidogenic pathway by its effect on the α-secretase. Proc. Natl. Acad. Sci. U.S.A. 98: 5815–5820.
Slack BE, Ma LK, Seah CC. (2001) Constitutive shedding of the amyloid precursor protein ectodomain is up-regulated by tumour necrosis factor-alpha converting enzyme. Biochem. J. 357: 787–794.
Parvathy S, Hussain I, Karran EH, Turner AJ, Hooper NM. (1998) Alzheimer’s amyloid precursor protein alpha-secretase is inhibited by hydroxamic acid-based zinc metalloprotease inhibitors: Similarities to the angiotensin converting enzyme secretase. Biochemistry 37: 1680–1685.
Di Luca M, Pastorino L, Cattabeni F, et al. (1996) Abnormal pattern of platelet APP isoforms in Alzheimer disease and Down syndrome. Arch. Neurol. 53: 1162–1166.
Davies TA, Long HJ, Tibbles HE, et al. (1997) Moderate and advanced Alzheimer’s patients exhibit platelet activation differences. Neurobiol. Aging 18: 155–162.
Davies TA, Long HJ, Sgro K. (1997) Activated Alzheimer disease platelets retain more beta amyloid precursor protein. Neurobiol. Aging 18: 147–153.
Rosenberg RN, Baskin F, Fosmire JA, et al. (1997) Altered amyloid protein processing in platelets of patients with Alzheimer disease. Arch. Neurol. 54: 139–144.
Bush AI, Tanzi RE. (1998) Alzheimer disease-related abnormalities of amyloid beta precursor protein isoforms in the platelets—the brain delegates in the periphery? Arch. Neurol. 55: 1179–1180.
Di Luca M, Pastorino L, Bianchetti A, et al. (1998) Differential pattern of platelet APP isoforms: An early marker for Alzheimer disease. Arch. Neurol. 55: 1195–1200.
Baskin F, Rosenberg RN, Iyer L, et al. (2000) Platelet APP isoform ratios correlate with declining cognition in AD. Neurology 54: 1907–1909.
Borroni B, Colciaghi F, Pastorino L, et al. (2001) Amyloid precursor protein in platelets of patients with Alzheimer disease—Effect of acetylcholinesterase inhibitor treatment. Arch. Neurol. 58: 442–446.
Padovani L, Pastorino B, Borroni et al. (In press) Amyloid precursor protein in platelets: A peripheral marker for the diagnosis of sporadic AD. Neurology.
Van Nostrand WE, Schmaier AH, Farrow JS, Cunningham DD. (1990) Protease nexin II (amyloid beta protein precursor): A platelet α-granule protein. Science 248: 745–748.
Li QX, Whyte S, Tanner JE, et al. (1998) Secretion of Alzheimer’s disease A beta amyloid peptide by activated human platelets. Lab. Invest. 78: 461–469.
Abraham CR, Marshall DC, Tibbles HE, et al. (1999) Platelets and DAMI megakaryocytes possess beta-secretase-like activity. J. Lab. Clin. Med. 133: 507–515.
Bush AI, Martins RN, Rumble B, et al. (1990) The amyloid precursor protein of Alzheimer’s disease is released by human platelets. J. Biol. Chem. 265: 15977–15983.
Lannfelt L, Basun H, Wahlund LO, et al. (1995) Decreased alpha-secretase-cleaved amyloid precursor protein as a diagnostic marker for Alzheimer’s disease. Nat. Med. 1: 829–832.
Almkvist O, Basun H, Wagner S, et al. (1997) Cerebrospinal fluid levels of alpha-secretase-cleaved soluble amyloid precursor protein mirror cognition in a Swedish family with Alzheimer disease and a gene mutation. Arch. Neurol. 54: 641–644.
Sennvik K, Fastbom J, Blomberg M, et al. (20000 Levels of a and β secretase cleaved amyloid precursor protein in the cerebrospinal fluid of Alzheimer’s disease patients. Neurosci. Lett. 278: 169–172.
Marcinkiewicz M, Seidah NG. (2000) Coordinated expression of β-amyloid precursor protein and putative β-secretase BACE and α-secretase ADAM10 in mouse and human brain. J. Neurochem. 75: 2133–2143.
Skovronsky DM, Lee VMY, Praticò D. (2001) Amyloid precursor protein and amyloid b peptide in human platelets. J. Biol. Chem. 286: 17036–17043.
Ferrarese C, Begni B, Canevari C, et al. (2000) L. Glutamate uptake is decreased in platelets from Alzheimer’s disease patients. Ann. Neurol. 47: 641–643.
Acknowledgments
This work is supported by CNR grants # 98.01097.CT14 and # 99.01234.CT14 to M.D.L., MURST 40% 1999–2000, N° 9906158271-004 to M.D.L., and Progetto Alzheimer Min. Sanita’2001–2003 to F.C. and M.D.L.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Colciaghi, F., Borroni, B., Pastorino, L. et al. α-Secretase ADAM10 as Well as αAPPs Is Reduced in Platelets and CSF of Alzheimer Disease Patients. Mol Med 8, 67–74 (2002). https://doi.org/10.1007/BF03402076
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF03402076