Abstract
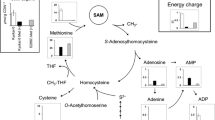
The ability to accumulate S-adenosylmethionine (SAM) of 572 yeast strains isolated from environmental sources were surveyed. An S-adenosylmethionine enriching strain S42-12, identified asCandida sp., was chose to develop a SAM-accumulating mutant successfully. The final SAM-accumulating mutant strain YQ-5 was isolated by UV radiation or by NTG treatment using ethionine selection and nystatin selection method. The mutant strain YQ-5 accumulated 112.1 mg per gram biomass, was 3.14-fold higher than the original strain S42-12. When cultivated in the optimal medium with a favourable fermentation conditions, SAM content of the mutant strain reached at 1740 mg L−1. Trend of SAM and ergosterol contents and methionine adenosyltransferase activity of SAM-accumulating mutants during fermentation were analysed. The results suggested that one of the reasons why the mutants accumulated SAM in significantly high amounts may be the lower consumption of SAM for ergosterol biosynthesis, other than improvement of methionine adenosyltransferase activity.
Similar content being viewed by others
References
Arthington-Skaggs B.A., Jardi H., Desai T., Morrison C.J. (1999). Quantitation of ergosterol content: novel method for determination of fluconazole susceptibility of Candida albicans. J. Clin. Microbiol., 37 (10): 3332–3337.
Bottiglieri T., Hyland K. (1994). S-adenosylmethionine levels in psychiatric and neurological disorders: a review. Acta Neurol Scand Suppl., 154:19–26.
Chen X., Wang Y., Zheng Y., Shen Y. (2004). Optimization of Fermentation Conditions and Cultural Media for S-Adenosylmethionine Production. China Biotechnology, 24(11): 65–69.
Kurtzman C.P. (2001). Four new Candida species from geographically diverse locations. Atntonie van leeuwenhoek, 79: 353–361.
Lieber C.S., Packer L. (2002). S-Adenosylmethionine: molecular, biological, and clinical aspects—an introduction. Am. J. Clin. Nutr., 76(5): 1148–1150
Mincheva K.P., Balutsov V.M. (2002). Isolation of aKluyveromyces lactis mutant enriched in S-adenosyl-L-methionine and growing in whey medium. Applied Biochemistry and Microbiology, 38(4): 335–338.
Philippsen P., Stotz A. and Scherf C. (1991). DNA of Saccharomyces cerevisiae. Methods Enzymol., 194: 169–182.
Schlenk F., Depalma R.E. (1957). The formation of S-adenosylmethionine in yeast. J. Biol. Chem., 229(2): 1037–1050.
Shiozaki S., Shimizu S. and Yamada H. (1984). Unusual intracellular accumulation of S-adenosyl- L-methionine by microorganisms. Agric. Biol. Chem., 48(9): 2293–2300.
Shobayashi M., Mukai N., Iwashita K., Hiraga Y., Iefuji H. (2006). A new method for isolation of S-adenosylmethionine (SAM)-accumulating yeast. Appl. Microbiol. Biotechnol., 69(6): 704–710.
Smit E., Leeflang P., Glandorf B., Elsas J.D. and Wernars K. (1999). Analysis of fungal diversity in the wheat rhizosphere by sequencing of cloned PCR-amplified genes encoding 18S rRNA and temperature gradient gel electrophoresis. Appl. Environ. Microbiol., 65: 2614–2621.
Tabor C.W., Tabor H. (1984). Methionine adenosyltransferase (S-adenosylmethionine synthetase) andS-adenosylmethionine decarboxylase. Adv. Enzymol., 56: 251–282.
Yarrow D. (1998). Method for the isolation, maintenance and identification of yeasts. InThe yeasts a Taxonomic Study, 4th edn. Kurtzman C.P. and Fell J. W. Elsevier, Amsterdam, 77–100.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Li, W., Ye, S., Luo, K. et al. Isolation and characterisation ofCandida sp. mutants enriched in S-adenosylmethionine (SAM). Ann. Microbiol. 57, 383–387 (2007). https://doi.org/10.1007/BF03175077
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF03175077