Abstract
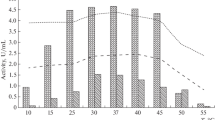
Alkaliphilic actinomycetes isolated from sediment samples of the Izmir Gulf, Turkey were studied for the production of protease activity. Strain MA1-1 was selected as a good alkaline protease producer as measured by the clear zone diameter by the hydrolysis of skim-milk and casein. The alkaline protease production from the marine alkaliphilic actinomycete MA1-1 was studied by using different carbon and nitrogen sources in medium containing glycerol, peptone, KCl, MgSO4, K2HPO4, and trace elements at 30°C for 72 h. Among the different carbon and nitrogen sources, fructose, starch, maltose, D(+) glucose, yeast extract, malt extract, beef extract and peptone provided higher production of protease. Starch was also found to be effective for growth and enzyme production with highest specific activity at 699 U mg−1. Purification was achieved by adsorption on Diaion HP 20 which resulted in a recovery rate of 68% with a specific activity of 7618 U mg−1 protein and 40-fold purification. The optimum pH and temperature of the partially purified protease were determined as pH 9.0 and 50°C, but high activity was also observed at pH 8.0–13.0 and 35–50°C. The inhibition profile exhibited by phenylmethylsulphonyl fluoride (PMSF) showed that this enzyme belongs to the serine-protease group.
Similar content being viewed by others
References
Arbige M.V., Pitcher W.H. (1989). Industrial enzymology: a look towards the future. Trends Biotechnol., 7: 330–335.
Ban O-H, Han S-S, Lee Y.N. (2003). Identification of a potent protease-producing bacterial isolate,Bacillus amyloliquefaciens CMB01. Ann. Microbiol., 53: 95–103.
Bradford M.M. (1976). A rapid and sensitive method for the quantification of protein using the principle of protein-dye binding. Anal. Biochem., 72: 248–254.
Coral G., Arikan B., Ünaldi M.N., Güvenmez H. (2003). Thermostable alkaline protease produced by anAspergillus niger strain. Ann. Microbiol., 53: 491–498.
Gupta R., Beg Q.K., Lorenz P. (2002). Bacterial alkaline proteases: molecular approaches and industrial applications. Appl. Microbiol. Biotechnol., 59: 15–32.
Imada I. (2005). Enzyme inhibitors and other bioactive compounds from marine actinomycetes. Antonie van Leeuwenhoek., 87: 59–63.
Jensen P.R., Gontang E., Mafnas C., Mincer T.J., Fenical W. (2005). Culturable marine actinomycete diversity from tropical Pacific Ocean sediments. Environ. Microbiol., 7 (7): 1039–1048.
Joo H.S., Park G.C., Kim K.T., Paik S.R., Chang C.S. (2001). Simple methods for alkaline protease purification from the polychaeta,Periserrula leucophryna. Process Biochem., 37: 299–303.
Kumar C.G., Takagi H. (1999). Microbial alkaline proteases: From a bioindustrial viewpoint. Biotechnol. Adv., 17: 561–594.
Mehta V.J., Thumar J.T., Singh S.P. (2006). Production of alkaline protease from an alkaliphilic actinomycete. Bioresource Technol., 97 (14): 1650–1654.
Petinate S.D.G., Branquinha M.H., Coelho R.R.R., Vermelho A.B., Giovanni-De-Simone S. (1999). Purification and partial characterization of an extracellular serine-proteinase ofStreptomyces cyaneus isolated from Brazilian cerrado soil. J. Appl. Microbiol., 87: 557–563.
Rahman R.N.Z.R.A., Basri M., Salleh A.B. (2003). Thermostable alkaline protease fromBacillus stearothermophilus F1; nutitional factors affecting protease production. Ann. Microbiol. 53: 199–210.
Rao M.B., Tanksale A.M., Ghatge M.S., Deshpande V.V. (1998). Molecular and biotechnological aspects of microbial proteases. Microbiol. Mol. Biol. Rev., 62(3): 597–635.
Takami H., Akiba T., Horikoshi A. (1989). Production of extremely thermostable alkaline protease fromBacillus sp. No. AH-101. Appl. Microbiol. Biotechnol., 30: 120–124.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hameş-Kocabaş, E.E., Uzel, A. Alkaline protease production by an actinomycete MA1-1 isolated from marine sediments. Ann. Microbiol. 57, 71–75 (2007). https://doi.org/10.1007/BF03175053
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF03175053