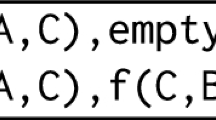Abstract
Problems concerned with learning the relationships between molecular structure and activity have been important test-beds for Inductive Logic programming (ILP) systems. In this paper we examine these applications and empirically evaluate the extent to which a first-order representation was required. We compared ILP theories with those constructed using standard linear regression and a decision-tree learner on a series of progressively more difficult problems. When a propositional encoding is feasible for the feature-based algorithms, we show that such algorithms are capable of matching the predictive accuracies of an ILP theory. However, as the complexity of the compounds considered increased, propositional encodings becomes intractable. In such cases, our results show that ILP programs can still continue to construct accurate, understandable theories. Based on this evidence, we propose future work to realise fully the potential of ILP in structure-activity problem.
Article PDF
Similar content being viewed by others
References
Bahler, D. and Bristol, D., “The Induction of Rules for Predicting Chemical Carcinogenesis,” inProceedings of the 26th Hawaii International Conference on System Sciences, Los Alamitos, IEEE Computer Society Press, 1993.
Breiman, L., Friedman, J. H., Olshen, R. A., and Stone, C. J.,Classification and Regression Trees, Wadsworth, Belmont, 1984.
Debnath, A. K., Lopez de Compadre, R. L., Debnath, G., Schusterman, A. J., and Hansch, C., “Structure-Activity Relationship of Mutagenic Aromatic and Hetero-aromatic Nitro Compounds. Correlation with Molecular Orbital Energies and Hydrophobicity,”Journal of Medicinal Chemistry, 34, 2, pp. 786–797, 1991.
Hansch, C., Malong, P. P., Fujita, T., and Muir, M.,Nature, 194, pp. 178–180, 1962.
Hirst, J. D., King, R. D., and Sternberg, M. J. E., “Quantitative Structure-Activity Relationships by Neural Networks and Inductive Logic Programming. ii. The Inhibition of Dihydrofolate Reductase by Triazines,”Journal of Computer-Aided Molecular Design, 8, pp. 421–432, 1994.
King, R., Muggleton, S., and Sternberg, M. J. E., “Drug Design by Machine Learning: The Use of Inductive Logic Programming to Model the Structure-Activity Relationships of Trimethoprim Analogues Binding to Dihydrofolate Reductase,”Proc. of the National Academy of Sciences, 89, 23, pp. 11322–11326, 1992.
King, R. D., Muggleton, S., Srinivasan, A., Feng, C., Lewis, R. A., and Sternberg, M. J. E., “Drug Design Using Inductive Logic Programming,” inProceedings of the 26th Hawaii International Conference on System Sciences, Los Alamitos, IEEE Computer Society Press, 1993.
Marsh, P., “Prescribing All the Way to the Bank,”New Scientist, 18 Nov. 1989.
Martin, Y. C.,Quantitative Drug Design: A Critical Introduction, Marcel Drekker Inc., New York, 1978.
Muggleton, S., “Inverse Entailment and Progol,” inNew Generation Computing, 13, Springer-Verlag and Ohmsha, Ltd., 1995.
Muggleton, S. H. and Feng, C., “Efficient Induction of Logic Programs,” inProceedings of the First Conference on Algorithmic Learning Theory, Tokyo, Ohmsha, 1990.
Ramsden, C. A.,Comprehensive Medicinal Chemistry (vol. 4), Pergamon Press, Oxford, 1979.
Srinivasan, A., Muggleton, S. H., King, R. D., and Sternberg, M. J. E., “Mutagenesis: ILP Experiments in a Non-Determinate Biological Domain,” inProceedings of the Fourth International Inductive Logic Programming Workshop (S. Wrobel, ed.), Gesellschaft fur Mathematik und Datenverarbeitung MBH, 1994.GMD-Studien Nr 237.
Author information
Authors and Affiliations
Additional information
Ross D. King, Ph. D: He currently works as a postdoctoral fellow in the Biomolecular Modelling Laboratory of the Imperial Cancer Research Fund. His interests are in the application of Machine Learning to the understanding of the relationship between molecular structure and function. He received his B. Sc. in Microbiology from the University of Aberdeen in 1983, his M. Sc. in Computer Science from the University of Newcastle Upon Tyne in 1985, and his Ph. D. from the Turing Institute, University of Strathclyde in 1988.
Michael J. E. Sternberg, D. Phil.: He is head of the Biomolecular Modelling Laboratory at the Imperial Cancer Research Fund, London. He studied Natural Sciences at Cambridge obtaining a B. A. in 1972. He obtained an M. Sc. in Computing from Imperial College London in 1974. His research in computer modelling of biologcal molecules was his Ph. D. thesis research in Oxford (1974–78) and he has continued in this area.
Ashwin Srinivasan, Ph. D: He currently works as a postdoctoral resercher at the University of Oxford computing Laboratory. His interests are in developing ILP systems and applying them to difficult real-world problems in molecular biology and chemistry. He received his B. E. in Electrical Engineering and Computer Science from the University of New South Wales, where he also received his Ph. D in 1991.
An erratum to this article is available at http://dx.doi.org/10.1007/BF03037220.
About this article
Cite this article
King, R.D., Sternberg, M.J.E. & Srinivasan, A. Relating chemical activity to structure: An examination of ILP successes. NGCO 13, 411–433 (1995). https://doi.org/10.1007/BF03037232
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF03037232