Abstract
Background: Lidocaine has been shown to inhibit neural conduction and to have anti-inflammatory properties. The purpose of this study was to determine whether intraoperative lidocaine infusion reduces opioid consumption in the postanesthesia care unit (PACU).
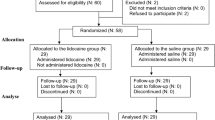
Methods: Fifty patients were enrolled in this prospective, randomized and observer-blinded study. At induction of anesthesia the control group (n=25) received fentanyl 3 µg·kg−1 while the lidocaine group received fentanyl 1.5 µg·kg−1 and a bolus of lidocaine 1.5 mg·kg−1 followed by a continuous infusion of lidocaine 2 mg·kg−1·hr−1. General anesthesia included propofol, rocuronium, and desflurane titrated to maintain blood pressure and heart rate within set parameters, and the bispectral index between 35 and 50. No supplemental opioids were given during surgery. All patients received acetaminophen, ketorolac, dexamethasone, droperidol and local anesthetics in the skin incision. Patients received fentanyl and ondansetron in the PACU. The primary outcome variable was the amount of fentanyl required in the PACU to establish and to maintain visual analogue scale pain scores<3.
Results: Most patients received fentanyl for pain relief in the PACU, but the cumulative mean dose was lower in the lidocaine group compared to the control group (98±54 µg,vs154±3 99 µg, respectively,P=0.018). Lidocaine infusion reduced by 10% the amount of desflurane required (P=0.012). White-Song scoresτ;12 were attained by all patients in both groups within 30 min of their arrival in the PACU. Median time from arrival to the PACU to discharge home was similar in both groups, 167.5 min in the control groupvs 180 min in the lidocaine group (P=0.649).
Conclusion: Intraoperative lidocaine infusion reduces opioid consumption in the PACU and intraoperative requirements of desflurane.
Résumé
Contexte: Il a été démontré que la lidocaïne inhibe la conduction nerveuse et possède des propriétés anti-inflammatoires. L’objectif de cette étude était de déterminer si une perfusion peropératoire de lidocaïne réduisait la consommation d’opioïdes dans la salle de réveil.
Méthode: Cinquante patients ont été recrutés dans le cadre de cette étude prospective, randomisée et à double insu. Lors de l’induction de l’anesthésie, le groupe témoin (n=25) a reçu 3 µg·kg−1 de fentanyl, et le groupe lidocaïne a reçu 1,5 µg·kg−1 de fentanyl ainsi qu’un bolus de 1,5 mg·kg−1 de lidocaïne suivi d’une perfusion continue de 2 mgsdkg−1·hr−1 de lidocaïne. L’anesthésie générale était composée de propofol, de rocuronium et de desflurane titré afin de maintenir la pression artérielle et la fréquence cardiaque dans la limite de paramètres préétablis, et l’index bispectral entre 35 et 50. Aucun opioïde supplémentaire n’a été administré pendant la chirurgie. Tous les patients ont reçu de l’acétaminophène, du kétorolac, de la dexaméthasone, du dropéridol et des agents anesthésiques locaux au niveau de l’incision cutanée. Les patients ont reçu du fentanyl et de l’ondansétron en salle de réveil. Le critère d’efficacité principal était la quantité de fentanyl nécessaire en salle de réveil pour établir et maintenir des scores de douleur<3 sur une échelle visuelle analogique.
Résultats: La plupart des patients ont reçu du fentanyl en salle de réveil pour soulager la douleur, mais la dose moyenne cumulative était plus basse dans le groupe lidocaïne que dans le groupe témoin (98±54 µg, vs 154±99 µg, respectivement, P=0,018). La perfusion de lidocaïne a réduit la quantité de desflurane requise de 10 % (P=0.012). Des scores de White-Song τ; 12 ont été obtenus chez tous les patients dans les deux groupes au cours des 30 premières minutes après leur transfer t à la salle de réveil. Le temps médian entre l’arrivée en salle de réveil et le congé de l’hôpital était semblable dans les deux groupes, soit 167,5 min dans le groupe témoin vs 180 min dans le groupe lidocaïne (P=0,649).
Conclusion: Une perfusion peropératoire de lidocaïne réduit la consommation d’opiacés en salle de réveil et les besoins peropératoires de desflurane.
Article PDF
Similar content being viewed by others
References
White PF. The role of non-opioid analgesic techniques in the management of pain after ambulatory surgery. Anesth Analg 2002; 94: 577–85.
Watt-Watson J, Chung F, Chan VW, McGillion M. Pain management following discharge after ambulatory same-day surgery. J Nurs Manag 2004; 12: 153–61.
Bisgaard T, Klarskov B, Rosenberg J, Kehlet H. Characteristics and prediction of early pain after laparoscopic cholecystectomy. Pain 2001; 90: 261–9.
Bisgaard T. Analgesic treatment after laparoscopic cholecystectomy: a critical assessment of the evidence. Anesthesiology 2006; 104: 835–46.
Groudine SB, Fisher HA, Kaufman RPJr,et al. Intravenous lidocaine speeds the return of bowel function, decreases postoperative pain, and shortens hospital stay in patients undergoing radical retropubic prostatectomy. Anesth Analg 1998; 86: 235–9.
Koppert W, Weigand M, Neumann F, et al. Perioperative intravenous lidocaine has preventive effects on postoperative pain and morphine consumption after major abdominal surgery. Anesth Analg 2004; 98: 1050–5.
Ness TJ. Intravenous lidocaine inhibits visceral nociceptive reflexes and spinal neurons in the rat. Anesthesiology 2000; 92: 1685–91.
Hollmann MW, Durieux ME. Local anesthetics and the inflammatory response: a new therapeutic indication? Anesthesiology 2000; 93: 858–75.
Wu CT, Borel CO, Lee MS, et al. The interaction effect of perioperative cotreatment with dextromethorphan and intravenous lidocaine on pain relief and recovery of bowel function after laparoscopic cholecystectomy. Anesth Analg 2005; 100: 448–53.
Kaba A, Laurent SR, Detroz BJ, et al. Intravenous lidocaine infusion facilitates acute rehabilitation after laparoscopic colectomy. Anesthesiology 2007; 106: 11–8; discussion 5–6.
White PF, Song D. New criteria for fast-tracking after outpatient anesthesia: a comparison with the modified Aldrete’s scoring system. Anesth Analg 1999; 88: 1069–72.
Collard V, Mistraletti G, Taqi A, et al. Intraoperative esmolol infusion in the absence of opioids spares postoperative fentanyl in patients undergoing ambulatory laparoscopic cholecystectomy. Anesth Analg 2007; 105: 1255–62.
Pypendop BH, Ilkiw JE. The effects of intravenous lidocaine administration on the minimum alveolar concentration of isoflurane in cats. Anesth Analg 2005; 100: 97–101.
Hollmann MW, Gross A, Jelacin N, Durieux ME. Local anesthetic effects on priming and activation of human neutrophils. Anesthesiology 2001; 95: 113–22.
Herroeder S, Pecher S, Schonherr ME, et al. Systemic lidocaine shortens length of hospital stay after colorectal surgery: a double-blinded, randomized, placebo-controlled trial. Ann Surg 2007; 246: 192–200.
Lau H, Brooks DC. Contemporary outcomes of ambulatory laparoscopic cholecystectomy in a major teaching hospital. World J Surg 2002; 26: 1117–21.
Kehlet H, Gray AW, Bonnet F, et al. A procedurespecific systematic review and consensus recommendations for postoperative analgesia following laparoscopic cholecystectomy. Surg Endosc 2005; 19: 1396–415.
Song D, White PF. Remifentanil as an adjuvant during desflurane anesthesia facilitates early recovery after ambulatory surgery. J Clin Anesth 1999; 11: 364–7.
Himes RS Jr,DiFazio CA, Burney RG. Effects of lidocaine on the anesthetic requirements for nitrous oxide and halothane. Anesthesiology 1977; 47: 437–40.
Hollmann MW, Strumper D, Herroeder S, Durieux ME. Receptors, G proteins, and their interactions. Anesthesiology 2005; 103: 1066–78.
Martin F, Cherif K, Gentili ME, et al. Lack of impact of intravenous lidocaine on analgesia, functional recovery, and nociceptive pain threshold after total hip arthroplasty. Anesthesiology 2008; 109: 118–23.
Author information
Authors and Affiliations
Corresponding author
Additional information
Dr S. Lauwick was awarded a clinical fellowship from the Steinberg-Bernstein Centre for Minimally Invasive Surgery and the Montreal General Hospital Foundation, and a clinical research grant from the CHU of LIEGE, Belgium. Dr. G. Michelagnoli received a research fellowship from the Department of Anesthesia, University of Florence, Italy. Dr. G. Mistraletti was supported by the Istituto di Anestesiologia e Rianimazione, University of Milan, San Paolo Hospital, Milan, Italy.
This work was supported by internal funds, Department of Anesthesia, McGill University Health Centre.
None of the authors has any conflict of interest related to this study.
Rights and permissions
About this article
Cite this article
Lauwick, S., Kim, D.J., Michelagnoli, G. et al. Intraoperative infusion of lidocaine reduces postoperative fentanyl requirements in patients undergoing laparoscopic cholecystectomy. Can J Anesth 55, 754–760 (2008). https://doi.org/10.1007/BF03016348
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF03016348