Abstract
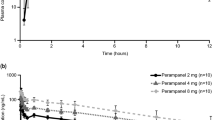
The pharmacokinetic variables of sufentanil were studied in 20 healthy children between two and eight years of age. The plasma concentrations of sufentanil were measured for up to 480 min after administration of a bolus of sufentanil, 1–3 μg · kg−1. The distribution half-life (t1/2β) was 5.2 ±2.2 (mean ±SD) min and the elimination half life (t1/2β) was 97.0 ±42.0 min. The volume of distribution at steady state (Vdss) was 2.9 ±0.6 L · kg−1 and the clearance was 30.5 ±8.8 ml · kg−1 · min−1. The Vdss was one and a half limes greater than that reported in adults when expressed as a function of body weight but similar to that of adults when expressed as a function of body surface area. According to our results, the clearance of sufentanil in normal children between two and eight years of age is twice as rapid as that described in adults and adolescents. A greater clearance of sufentanit in children suggests that they would require relativelygreater maintenance doses than adults.
Résumé
La pharmacocinétique du sufentanil a été étudiée chez 20 enfants agés de deux à huit ans. Les concentrations plasmatiques ont pu être mesurées jusqu ’à 480 min après l’administration d’un bolus de 1 à 3 μg · kg−1 de sufentanil. La demi-vie de distribution (t1/2α) était de 5.2 ±2,2 min et la demi-vie d’élimination (t1/2β) était de 97,0 ±42,0 min. Le volume de distribution à l’état d’équilibre (Vdss) était 2,9 ±0,6 L · kg−1 et la clairance 30,5 ±8,8 ml · kg−1 · min−1. Le Vdss est égal à environ une fois et demi celui de précédemment rapporté pour l’adulte lorsqu’il est exprimé en fonction du poids corporel et superposable à celui-ci lorsqu’il est exprimé en fonction de la surface corporelle. D’après nos résultats la clairance du sufentanil chez l’enfant normal de deux à huit ans est environ deux fois celle décrite chez l’adolescent et l’adulte. Une clairance augmentée suggère que les enfants nécessitent des doses d’entretien plus élevées.
Article PDF
Similar content being viewed by others
References
Hickey PR, Hansen DD. Fentanyl and sufentanil-oxygenpancuronium anesthesia for cardiac surgery in children. Anesth Analg 1984; 63: 117–24.
Moore RA, Yang SS, McNicholas KW, Gallagher JD.Clark DL. Hemodynamic and anesthetic effects of sufentanil as the sole anesthetic for pediatric cardiovascular surgery. Anesthesiology 1985; 62: 725–31.
Glenski JA, Friesen RH, Lane GA, Young S, Glascock J. Low-dose sufentanil as a supplement to halothane/N2O anesthesia in infants and children. Can J Anaesth 1988; 35: 379–84.
Vercauteren M, Boeckx E, Hangegreefs G, Noorduin H, Vanden Bussche G. Intranasal sufentanil for pre-operative sedation. Anaesthesia 1988; 43: 270–3.
Henderson JM, Brodsky DA, Fisher DM, Brett Cm, Hertzka RE. Pre-induction of anesthesia in pediatric patients with nasally administered sufentanil. Anesthesiology 1988; 68: 671–5.
Greeley WJ, De Bruijn NP, Davis DP. Sufentanil pharmacokinetics in pediatric cardiovascular patients. Anesth Analg 1987; 66: 1067–72.
Davis PJ, Cook DR, Stiller RL, Davin-Robinson KA. Pharmacodynamics and pharmacokinetics of high-dose sufentanil in infants and children undergoing cardiac surgery. Anesth Analg 1987; 66: 203–8.
Greeley WJ, De Bruijn NP. Changes in sufentanil pharmacokinetics within the neonatal period. Anesth Analg 1988; 67: 86–90.
Koren G, Goresky G, Crean P, Klein J, MacLeod SM. Unexpected alterations in fentanyl pharmacokinetics in children undergoing cardiac surgery: age related or disease related? Dev Pharmacol Ther 1986; 9: 183–91.
Michiels M, Hendriks R, Heykants J. Radioimmunoassay of the new opiate analgesics alfentanil and sufentanil. Preliminary pharmacokinetic profile in man. J Pharm Pharmacol 1983; 35: 86–93.
Gibaldi M, Perrier D. Pharmacokinetics. New York, Marcel Dekker. 1982, pp 409–17.
Bovill JG, Sebel PS, Blackburn CL, Oei-Lim V, Heykants JJ. The pharmacokinetics of sufentanil in surgical patients. Anesthesiology 1984; 61: 502–6.
Davis PJ, Stiller RL, Cook DR, Brandom BW, Davin-Robinson KA. Pharmacokinetics of sufentanil in adolescent patients with chronic renal failure. Anesth Analg 1988; 67: 268–71.
Davis PJ, Cook DR. Clinical pharmacokinetics of the newer intravenous anaesthetic agents. Clin Pharmacol 1986; 11: 18–35.
Chiou WL The phenomenon and rationale of marked dependance of drug concentration on blood sampling site: implications in pharmacokinetics, pharmacodynamics, toxicology and therapeutics (Part II). Clin Pharmacokinet 1989; 17:275–90.
Nies AS, Shand DG, Wilkinson GR. Altered hepatic blood flow and drug disposition. Clin Pharmacokinet 1976; 1: 135–55.
Balistreri WF. Development of hepatic and biliary structure and function. In: Behrman RE, Vaughan VC (Eds.). Nelson Textbook of Pediatrics, 13th ed., Philadelphia: WB Saunders Cie., 1987:821–4.
Loughan PM, Sitar DS, Oglivie RI, Eisen A, Fox F, Neims AH. Pharmacokinctic analysis of the disposition of intravenous theophylline in the young children. J Pediatr 1976; 88: 874–9.
Gichansky E, Weinberger M. Relationship of theophylline clearance to oral dosage in children with chronic asthma. J Pediatr 1977; 91: 655–60.
Hendeles L, Weinberger M, Bighley L. Disposition of theophylline after a single intravenous infusion of aminophylline. Am J Resp Dis 1978; 118: 97–103.
Gelman S, Fowler KC andSmith LR. Liver circulation and function during isoflurane and halothane anesthesia. Anesthesiology 1984; 61: 726–30.
Karlin JM, Kutt H. Acute diphenylhydantoin intoxication following halothane anesthesia. J Pediatr 1970; 76: 941–4.
White PF, Johnstone PR, Pudwill CR. Interaction of ketamine and halothane in rats. Anesthesiology 1975; 42: 179–86.
Stenson RE, Constantino RI, Harrison DC. Interrelationship of hepatic blood flow, cardiac output and blood levels of lidocaine in man. Circulation 1971; 43: 205–11.
Hepper GW, Vesell ES, Tantum KR. Reduced drug elimination in congestive heart failure: studies using aminopyrine as a model drug. Am J Med 1978; 65: 271–6.
Author information
Authors and Affiliations
Additional information
This study was supported in part by grants from Janssen Pharmaceutica Canada Ltd. and from the Inter-Service Club Council of Montreal
Rights and permissions
About this article
Cite this article
Guay, J., Gaudreault, P., Tang, A. et al. Pharmacokinetics of sufentanil in normal children. Can J Anaesth 39, 14–20 (1992). https://doi.org/10.1007/BF03008666
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF03008666