Abstract
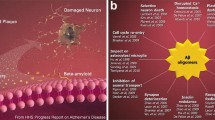
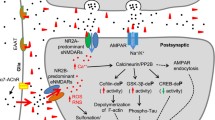
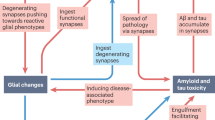
Individuals with early-stage Alzheimer’s disease (AD) suffer from profound failure to form new memories. A novel molecular mechanism with implications for therapeutics and diagnostics is now emerging in which the specificity of AD for memory derives from disruption of plasticity at synapses targeted by neurologically active Aβ oligomers (1). We have named these oligomers “ADDLs” (for pathogenic Aβ-Derived Diffusible Ligands). ADDLs constitute metastable alternatives to the disease-defining Aβ fibrils deposited in amyloid plaques. In AD brain, ADDLs accumulate primarily as Aβ 12mers (2) (∼54 kDa) and can be found in dot-like clusters distinct from senile plaques (3). Oligomers of equal mass have been reported to occur in tg-mouse AD models where they emerge concomitantly with memory failure (4), consistent with ADDL inhibition of LTP (1). In cell biology studies, ADDLs act as pathogenic gain-of-function ligands that target particular synapses, binding to synaptic spines at or near NMDA receptors (5,6). Binding produces ectopic expression of the memory-linked immediate early gene Arc. Subsequent ADDL-induced abnormalities in spine morphology and synaptic receptor composition (7) are predicted consequences of Arc overexpression, a pathology associated with memory dysfunction in tg-Arc mice. Significantly, the attack on synapses provides a plausible mechanism unifying memory dysfunction with major features of AD neuropathology; recent findings show that ADDL binding instigates synapse loss, oxidative damage, and AD-type tau hyperphosphorylation. Acting as novel neurotoxins that putatively account for memory loss and neuropathology, ADDLs present significant targets for disease-modifying therapeutics in AD.
Similar content being viewed by others
References
Lambert, M.P., Barlow, A.K., Chromy, B.A., Edwards, C., Freed, R., Liosatos, M., Morgan, T.E., Rozovsky, I., Trommer, B., Viola, K.L., Wals, P., Zhang, C., Finch, C.E., Krafft, G.A. & Klein, W.L. (1998) Diffusible, nonfibrillar ligands derived from Abetal-42 are potent central nervous system neurotoxins. Proc. Natl. Acad. Sci. U. S. A. 95, 6448–6453.
Gong, Y., Chang, L., Viola, K.L., Lacor, P.N., Lambert, M.P., Finch, C.E., Krafft, G.A. & Klein, W.L. (2003) Alzheimer’s disease-affected brain: presence of oligomeric A beta ligands (ADDLs) suggests a molecular basis for reversible memory loss. Proc. Natl. Acad. Sci. U. S. A. 100, 10417–10422.
Ishibashi, K., Tomiyama, T., Nishitsuji, K., Hara, M. & Mori, H. (2006) Absence of synaptophysin near cortical neurons containing oligomer Abeta in Alzheimer’s disease brain. J Neurosci. Res. 84, 632–636.
Lesne, S., Koh, M.T., Kotilinek, L., Kayed, R., Glabe, C.G., Yang, A., Gallagher, M. & Ashe, K.H. (2006) A specific amyloid-beta protein assembly in the brain impairs memory. Nature 440, 352–357.
Lacor, P.N., Buniel, M.C., Chang, L., Fernandez, S.J., Gong, Y., Viola, K.L., Lambert, M.P., Velasco, P.T., Bigio, E.H., Finch, C.E., Krafft, G.A. & Klein, W.L. (2004) Synaptic targeting by Alzheimer’s-related amyloid beta oligomers. J Neurosci. 24, 10191–10200.
De Felice, F.G., Velasco, P.T., Lambert, M.P., Viola, K., Fernandez, S.J., Ferreira, S.T. & Klein, W.L. (2007) Abeta oligomers induce neuronal oxidative stress through an N-methyl-D-aspartate receptor-dependent mechanism that is blocked by the Alzheimer drug memantine. J Biol Chem. 282, 11590–11601.
Lacor, P.N., Buniel, M.C., Furlow, P.W., Clemente, A.S., Velasco, P.T., Wood, M., Viola, K.L. & Klein, W.L. (2007) Abeta oligomer-induced aberrations in synapse composition, shape, and density provide a molecular basis for loss of connectivity in Alzheimer’s disease. J Neurosci. 27, 796–807.
Hardy, J. & Selkoe, D.J. (2002) The amyloid hypothesis of Alzheimer’s disease: progress and problems on the road to therapeutics. Science 297, 353–356.
Selkoe, D.J. (2002) Alzheimer’s disease is a synaptic failure. Science 298, 789–791.
El Agnaf, O.M., Walsh, D.M. & Allsop, D. (2003) Soluble oligomers for the diagnosis of neurodegenerative diseases. Lancet Neurol. 2, 461–462.
Caughey, B. & Lansbury, P.T., Jr. (2003) Protofibrils, Pores, Fibrils, and Neurodegeneration: Separating the Responsible Protein Aggregates from the Innocent Bystanders. Annu. Rev. Neurosci. 26, 267–298.
Mattson, M.P. (2004) Pathways towards and away from Alzheimer’s disease. Nature 430, 631–639.
Walsh, D.M. & Selkoe, D.J. (2004) Deciphering the molecular basis of memory failure in Alzheimer’s disease. Neuron 44, 181–193.
Walsh, D.M., Klyubin, I., Shankar, G.M., Townsend, M., Fadeeva, J.V., Betts, V., Podlisny, M.B., Cleary, J.P., Ashe, K.H., Rowan, M.J. & Selkoe, D.J. (2005) The role of cell-derived oligomers of Abeta in Alzheimer’s disease and avenues for therapeutic intervention. Biochem. Soc. Trans. 33, 1087–1090.
Rowan, M.J., Klyubin, I., Wang, Q. & Anwyl, R. (2005) Synaptic plasticity disruption by amyloid beta protein: modulation by potential Alzheimer’s disease modifying therapies. Biochem. Soc. Trans. 33, 563–567.
Glabe, C.G. (2006) Common mechanisms of amyloid oligomer pathogenesis in degenerative disease. Neurobiol. Aging 27, 570–575.
Standridge, J.B. (2006) Vicious cycles within the neuropathophysiologic mechanisms of Alzheimer’s disease. Curr. Alzheimer Res. 3, 95–108.
Klein, W.L., Lacor, P.N., Felice, F.G. & Ferreira, S.T. Molecules that disrupt memory circuits in Alzheimer’s disease: the attack on synapses by Abeta oligomers (ADDLs). In Memories: Molecules and Circuits. Bontempi, B., Silva, A.J. & Christen, Y. (eds.), pp. 155–179 (Springer, New York, 2006).
Smith, A. (2003) Amyloid-β: less bark, more bite! Nat. Rev. Drug Discov. 2, 771.
Valeo, T. (2004) Downsized target Sci. Am. 290, 26, 28.
Keating, C.D. (2005) Nanoscience enables ultrasensitive detection of Alzheimer’s biomarker. Proc. Natl. Acad. Sci. U. S. A. 102, 2263–2264.
Fradinger, E.A. & Bitan, G. (2005) En route to early diagnosis of Alzheimer’s disease-are we there yet? Trends Biotechnol. 23, 531–533.
Hardy, J.A. & Higgins, G.A. (1992) Alzheimer’s disease: the amyloid cascade hypothesis. Science 256, 184–185.
Dodart, J.C., Bales, K.R., Gannon, K.S., Greene, S.J., DeMattos, R.B., Mathis, C., DeLong, C.A., Wu, S., Wu, X., Holtzman, D.M. & Paul, S.M. (2002) Immunization reverses memory deficits without reducing brain Abeta burden in Alzheimer’s disease model. Nat Neurosci. 5, 452–457.
Kotilinek, L.A., Bacskai, B., Westerman, M., Kawarabayashi, T., Younkin, L., Hyman, B.T., Younkin, S. & Ashe, K.H. (2002) Reversible memory loss in a mouse transgenic model of Alzheimer’s disease. J. Neurosci. 22, 6331–6335.
Frackowiak, J., Zoltowska, A. & Wisniewski, H.M. (1994) Non-fibrillar beta-amyloid protein is associated with smooth muscle cells of vessel walls in Alzheimer disease. J. Neuropathol. Exp. Neurol. 53, 637–645.
Kuo, Y.M., Emmerling, M.R., Vigo-Pelfrey, C., Kasunic, T.C., Kirkpatrick, J.B., Murdoch, G.H., Ball, M.J. & Roher, A.E. (1996) Water-soluble Abeta (N-40, N-42) oligomers in normal and Alzheimer disease brains. J. Biol. Chem. 271, 4077–4081.
Chromy, B.A., Nowak, R.J., Lambert, M.P., Viola, K.L., Chang, L., Velasco, P.T., Jones, B.W., Fernandez, S.J., Lacor, P.N., Horowitz, P., Finch, C.E., Krafft, G.A. & Klein, W.L. (2003) Self-assembly of Abeta(1-42) into globular neurotoxins. Biochemistry 42, 12749–12760.
Boutaud, O., Montine, T.J., Chang, L., Klein, W.L. & Oates, J.A. (2006) PGH-derived levuglandin adducts increase the neurotoxicity of amyloid betal-42. J Neurochem. 96, 917–923.
Wang, H.W., Pasternak, J.F., Kuo, H., Ristic, H., Lambert, M.P., Chromy, B., Viola, K.L., Klein, W.L., Stine, W.B., Krafft, G.A. & Trommer, B.L. (2002) Soluble oligomers of beta amyloid (1-42) inhibit long-term potentiation but not long-term depression in rat dentate gyrus. Brain Res. 924, 133–140.
Cleary, J.P., Walsh, D.M., Hofmeister, J.J., Shankar, G.M., Kuskowski, M.A., Selkoe, D.J. & Ashe, K.H. (2005) Natural oligomers of the amyloid-beta protein specifically disrupt cognitive function. Nat. Neurosci. 8, 79–84.
Lambert, M.P., Viola, K.L., Chromy, B.A., Chang, L., Morgan, T.E., Yu, J., Venton, D.L., Krafft, G.A., Finch, C.E. & Klein, W.L. (2001) Vaccination with soluble Abeta oligomers generates toxicity-neutralizing antibodies. J. Neurochem. 79, 595–605.
Lambert, M.P., Velasco, P.T., Chang, L., Viola, K.L., Fernandez, S., Lacor, P.N., Khuon, D., Gong, Y., Bigio, E.H., Shaw, P., De Felice, F.G., Krafft, G.A. & Klein, W.L. (2007) Monoclonal antibodies that target pathological assemblies of Abeta. J Neurochem. 100, 23–35.
Kayed, R., Head, E., Thompson, J.L., McIntire, T.M., Milton, S.C., Cotman, C.W. & Glabe, C.G. (2003) Common structure of soluble amyloid oligomers implies common mechanism of pathogenesis. Science 300, 486–489.
Chang, L., Bakhos, L., Wang, Z., Venton, D.L. & Klein, W.L. (2003) Femtomole immunodetection of synthetic and endogenous Amyloid-β oligomers and its application to Alzheimer’s Disease drug candidate screening. J. Mol. Neurosci. 20, 305–313.
Georganopoulou, D.G., Chang, L., Nam, J.M., Thaxton, C.S., Mufson, E.J., Klein, W.L. & Mirkin, C.A. (2005) Nanoparticle-based detection in cerebral spinal fluid of a soluble pathogenic biomarker for Alzheimer’s disease. Proc. Natl. Acad. Sci. U. S. A. 102, 2273–2276.
Schneider, A., Schulz-Schaeffer, W., Hartmann, T., Schulz, J.B. & Simons, M. (2006) Cholesterol depletion reduces aggregation of amyloid-beta peptide in hippocampal neurons. Neurobiol. Dis. 23, 573–577.
Townsend, M., Shankar, G.M., Mehta, T., Walsh, D.M. & Selkoe, D.J. (2006) Effects of secreted oligomers of amyloid beta-protein on hippocampal synaptic plasticity: a potent role for trimers. J Physiol 572, 477–492.
Kokubo, H., Kayed, R., Glabe, C.G. & Yamaguchi, H. (2005) Soluble Abeta oligomers ultrastructurally localize to cell processes and might be related to synaptic dysfunction in Alzheimer’s disease brain. Brain Res. 1031, 222–228.
Zhao, W.Q., De Felice, F.G., Fernandez, S., Chen, H., Lambert, M.P., Quon, M., Krafft, G.A. & Klein, W.L. (2007) Amyloid beta oligomers induce impairment of neuronal insulin receptors. FASEB J.
Guzowski, J.F., Lyford, G.L., Stevenson, G.D., Houston, F.P., McGaugh, J.L., Worley, P.F. & Barnes, C.A. (2000) Inhibition of activity-dependent arc protein expression in the rat hippocampus impairs the maintenance of long-term potentiation and the consolidation of long-term memory. J. Neurosci. 20, 3993–4001.
Guzowski, J.F. (2002) Insights into immediate-early gene function in hippocampal memory consolidation using antisense oligonucleotide and fluorescent imaging approaches. Hippocampus 12, 86–104.
Kaufmann, W.E. & Moser, H.W. (2000) Dendritic anomalies in disorders associated with mental retardation. Cereb. Cortex 10, 981–991.
Halpain, S., Spencer, K. & Graber, S. (2005) Dynamics and pathology of dendritic spines. Prog. Brain Res. 147, 29–37.
Zhao, W.Q. & Alkon, D.L. (2001) Role of insulin and insulin receptor in learning and memory. Mol. Cell Endocrinol. 177, 125–134.
Ferrer, I., Boada, R.M., Sanchez Guerra, M.L., Rey, M.J. & Costa-Jussa, F. (2004) Neuropathology and pathogenesis of encephalitis following amyloid-beta immunization in Alzheimer’s disease. Brain Pathol. 14, 11–20.
De Felice, F.G., Vieira, M.N., Saraiva, L.M., Figueroa-Villar, J.D., Garcia-Abreu, J., Liu, R., Chang, L., Klein, W.L. & Ferreira, S.T. (2004) Targeting the neurotoxic species in Alzheimer’s disease: inhibitors of Abeta oligomerization. FASEB J 18, 1366–1372.
Wang, Z., Chang, L., Klein, W.L., Thatcher, G.R. & Venton, D.L. (2004) Per-6-substituted-per-6-deoxy beta-cyclodextrins inhibit the formation of beta-amyloid peptide derived soluble oligomers. J Med. Chem. 47, 3329–3333.
Walsh, D.M., Townsend, M., Podlisny, M.B., Shankar, G.M., Fadeeva, J.V., Agnaf, O.E., Hartley, D.M. & Selkoe, D.J. (2005) Certain inhibitors of synthetic amyloid beta-peptide (Abeta) fibrillogenesis block oligomerization of natural Abeta and thereby rescue long-term potentiation. J Neurosci. 25, 2455–2462.
Wu, Y., Wu, Z., Butko, P., Christen, Y., Lambert, M.P., Klein, W.L., Link, C.D. & Luo, Y. (2006) Amyloid-beta-induced pathological behaviors are suppressed by Ginkgo biloba extract EGb 761 and ginkgolides in transgenic Caenorhabditis elegans. J Neurosci. 26, 13102–13113.
Witt, A., Macdonald, N. & Kirkpatrick, P. (2004) Memantine hydrochloride. Nat Rev. Drug Discov. 3, 109–110
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Viola, K.L., Velasco, P.T. & Klein, W.L. Why Alzheimer’s is a disease of memory: The attack on synapses by Aß oligomers (ADDLs). J Nutr Health Aging 12 (Suppl 1), S51–S57 (2008). https://doi.org/10.1007/BF02982587
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02982587