Abstract
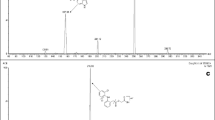
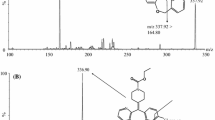
A sensitive and selective method for the determination of 4′-ethyl-3-methyl-3-piperidinopro-piophenone hydrochloride (eperisone hydrochloride) in human plasma was developed and validated. The procedure employed an internal standard and a solvent extraction step followed by chromatography on a Xterra C18 minibore column. Detection was by electrospray ionization tandem mass spectrometry with multiple reaction monitoring. The mass transitions of eperisone and tolperisone (IS) were m/z 260 → 98 and m/z 246 → 98, respectively. The method has a limit of detection of 0.1 pg/mL for eperisone based on the three times signal-to-noise value with a linear range from 0.01 to 10.0 ng/mL for the analyte. Extraction recovery was on average 98.6±7.2% (SD) for eperisone. The Intra- and inter-day assay accuracy ranged from 93 to 114% and precision (RSD) was better than 8.5%. The method was successfully employed to analyze plasma samples and evaluate pharmacokinetics of eperisone in healthy male volunteers.
Similar content being viewed by others
References
Ackermann, B. L., Berna, M. J., and Murphy, A.T., Recent advances in use of LC/MS/MS for quantitative high -throughput bioanalytical support of drug discovery.Curr. Top. Med. Chem., 2, 53–66 (2002).
Fujita, T., Takamatsu, T., Hisamoto, T., Tsutsumi, J., Kinoshita, K., and Kanai, T., Studies on the metabolic fate of 4′-ethtyl-2-methyl-3-piperidinopropiopenone hydrochloride(1):absorption, disposition and excretion in rat and guinea pigs.Pharmacometrics, 21, 835–846 (1981).
Iwase, S., Mano, T., Saito, M., and Ishida, G., Effect of a centrally-acting muscle relaxant eperisone hydrochloride, on muscle symmmpathetic nerve activity in humans.Funct Neurol., 7, 459–470 (1992).
Kuroiwa, Y., Sobue, I., Tazak, Y. I., Nakanishi, T., Ohtomo, E., and Itahara, K., Effects of E-0646 on cases of spasticity - A double blind comparision using tolperisone hydrochloride.Clin. Eval., 9, 391–419 (1981).
Li, D., Xin, W., Shengqiang, Z., Jianpin, S., and Yindi, Z., Rapid and sensitive liquid chromatography-electrospray ionization-mass specrtometry method for the determination of eperisone in human plasma: Method and Clinical Applications.J. Chromatogr Sci., 42, 254–258 (2004).
Matuszewski, B. K., Constanzer M. L., and Chavez-Eng, C. M., Strategies for the assessment of matrix effect in quantitative bioanalytical methods based on HPLC-MS/MS.Anal. Chem., 75, 3019–3030 (2003).
Mihara, K., Masumura, M., Yoshioka, E., Hanada, K., Nakasa, H., Ohmori, S., Kitada, M., and Ogata, H., Intestinal first-pass metabolism of eperisone in the rat.Pharm. Res., 18, 1131–37 (2001).
Takenaka, F., Hasui, M., and Ohkawa, T., Determination of eperisone in human plasma by gas chromatography -mass spectrometry.J. Chromatogr., 584, 261–266 (1992).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jeoung, M.K., Jeong, E.S., Kim, N.H. et al. Determination of eperisone in human plasma by liquid chromatography-ESI-tandem mass spectrometry. Arch Pharm Res 30, 1174–1178 (2007). https://doi.org/10.1007/BF02980255
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02980255