Abstract
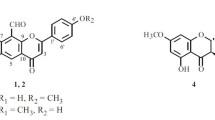
The chromatographic separation of the MeOH extract from the twigs ofAcer tegmentosum led to the isolation of ten phenolic compounds. The structures of these compounds were determined using spectroscopic methods as 3,7,3′,4′-tetramethyl-quercetin (1), 5,3′-dihydroxy-3,7,4′-trimethoxy flavone (2), 2,6-dimethoxy-p-hydroquinone (3), (-)-catechin (4), morin-3-O-α-L-lyxoside (5),p-hydroxy phenylethyl-O-ß-D-glucopyranoside (6), 3,5-dimethoxy-4-hydroxy phenyl-1-O-ß-D-glucoside (7), fraxin, (8), 3,5-dimethoxy-benzyl alcohol 4-O-ß-D-glucopyrano-side (9) and 4-(2,3-dihydroxy propyl)-2,6-dimethoxy phenyl ß-D-glucopyranoside (10). The compounds were examined for their cytotoxic activity against five cancer cell lines. Compound3 exhibited good cytotoxic activity against five human cancer cell lines with ED50 values ranging from 1.32 to 3.85 μM.
Similar content being viewed by others
References
Ahn, D. K., Illustrated Book of Korean Medicinal Herbs. Kyo-Hak publishing, Seoul, p 523 (1998).
Adolf, H., Peter, P., and Eric, E. Conn., Dhurrin, (-)-catechin, flavonol glycosides and flavones fromChamaebatia foliolosa.Phytochemistry, 26, 1546–1547 (1987).
Bilia, A. R., Morelli, I., Hamburger, M., and Hostettmann, K., Flavanes and a-type proanthocyanidins fromPrunus prostrata.Phytochemistry, 43, 887–892 (1996).
Emi, O., Tetsuya, H., Takamitsu, M., Haruhiro, F., Masami, I., and Mikio, Y., Analgesic Components of Saposhnikovia Root (Saposhnikovia divaricata).Chem. Pharm. Bull., 49, 154–160 (2001).
Hatano, T., Hattori, S., Ikeda, Y., Shingu, T., and Okuda, Y., Tannins of Aceraceous plants. Part II. Gallotannins having a 1,5-anhydro-D-glucitol core and some ellagitannins from Acer species.Chem. Pharm. Bull., 38, 1902–1905 (1990).
Hefeng, P. and Lennart, N. L., Phenolics from inner bark ofPinus sylvestris., Phytochemistry, 42, 1185–1189 (1996).
Hideaki, O., Mami, T., Shogo, I., Tomohiro, S., and Kazuo, Y., Phenolic compounds fromCoix lachryma-jobi var.ma-yuen., Phytochemistry, 28, 883–886 (1989).
Hidetoshi, A. and Gen, I. D., Isolation of antimicrobial compounds from Guava (Pisdium guajava L.) and their structural elucidation.Biosci. Biotechnol. Biochem., 66, 1727–1730 (2002).
Hideyuki, M., Hiroyuki, M., Chikako, A., Midori, A., Teruhiko, Y., and Junya, M., Isolation of α-glucosidase inhibitors from hyssop (Hyssopus officinalis).Phytochemistry, 65, 91–97 (2004).
Jorn, L., Alfred, B., Thomas, D., Turgen, S., Vidtor, W., Dierk, S., Dieter, S., and Sabine, R., Accumulation of tyrosol glucoside in transgenic potato plants expressing a parsley tyrosine decarboxylase. Hiroko Oguchi, lonone and lignan glycosides fromEpimedium diphyllum.Phytochemistry, 60, 683–689 (2002).
Junichi, K., Toru, I., Yasuko, T., Masateru, O., Yasuyuki, I., and Toshihiro, N., Water soluble constituents of Fennel. V Glycosides of Aromatic compounds.Chem. Pharm. Bull., 46, 1587–1590 (1998).
Kangi, I., Gen, I. N., and Itsuo, N., Flavan-3-ol and procyanidin glycosides fromQuercus miygii. Phytochemistry, 26, 1167–1170 (1987).
Kanji, I., Hiroshi, S., Motoyoshi, S., and Koichiro, S., Phenyl glucosides from hairy root culture ofSwertia jponica.Phytochemistry, 29, 3823–3825 (1990).
Kubo, M., Inoue, T., and Nagai, M., Studies on the constituents of aceraceae plants. III. Structure of acerogenin B fromAcer nikoense Maxim.Chem. Pharm. Bull., 28, 1300–1303 (1980).
Kubo, M., Nagai, M., and Inoue, T., Studies on the constituents of ceraceae plants. Carbon-13 nuclear magnetic resonance spectra of acerogenin A, rhododendrol, and related compounds, and structure of aceroside fromAcer nikoense.Chem. Pharm. Bull., 31, 1917–1922 (1983).
Masataka, S. and Masao, K., Studies on the constituents ofOsmantus species. X. Structures of phenolic glucosides form the leaves ofOsmanthus asiaticus Nakai.Chem. Pharm. Bull., 40, 325–326 (1992).
Renmin, L., Qinghua, S., Ailing, S., and Jichun, C., Isolation and purification of coumarin compounds fromCortex fraxinus by high-speed counter-current chromatography.J. Chromatogr. A., 1072, 195–199 (2005).
Stephen, J. P., Louise, N. J., and David, C. P., High-resolution1H- and13C-NMR. Spectra of D-glucopyranose, 2-acetamido-2-deoxy-D-glucopyranose, and related compounds in aqueous media.Carbohydrate Research, 59, 19–34 (1977).
Ying, W., Matthias, H., Joseph, G., and Kurt, H., Antimicrobial flavonoids fromPsiadia trinervia and their methylated and acetylated derivatives.Phytochemistry, 28, 2323–2327 (1989).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Park, K.M., Yang, M.C., Lee, K.H. et al. Cytotoxic phenolic constituents ofAcer tegmentosum maxim. Arch Pharm Res 29, 1086–1090 (2006). https://doi.org/10.1007/BF02969296
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02969296