Summary
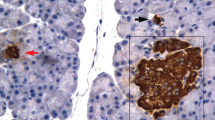
In vivo3H-Thymidine autoradiographic investigations of DNA synthesis in acinar, islet and duct cells in the pancreas of normal rats showed that activity was dependent on age. The proliferation of acinar and islet cells, which was high in young animals, decreased exponentially with age; proliferation of the ductal cells on the other hand, increased until the animals became mature. These findings suggest that the physiological regeneration of acinar and islet cells, as well as their replacement after injury in adult animals commences from pancreatic ducts.
Similar content being viewed by others
References
Bani D, Bani Sacchi T (1985a) The intermediate cells of the rat pancreas in normal conditions and following portocaval shunt. Z mikrosk-anat Forsch 99:353–361
Bani D, Bani Sacchi T, Billiotti G (1985b) Nesidioblastosis and intermediate cells in the pancreas of patients with hyperinsulinaemic hypoglycaemia. Virchows Arch [B] 48:19–32
Bani D, Bani Sacchi T, Billiotti G (1985c) Nesidioblastosis and islet cell changes related to endogenous hypergastrinaemia. Virchows Arch [B] 48:261–276
Blum B (1963) see- Hellerström G (1977) Growth pattern of pancreatic islets in animals. In: Volk BW, Wellman KF (eds) The diabetic pancreas. Plenum Press, New York, pp 70–97
Chick WL, Boston MD (1973) Bet-cell replication in pancreatic monolayer cultures. Diabetes 22:687–693
Chick WL (1973) Effects of glucose on Beta-cells in pancreatic monolayer cultures. Endocrinology 92:212–218
Cossel L (1984) Intermediate cells in the adult human pancreas. Virchows Arch [B] 47:313–328
Cossel L (1986) Electron microskopic demonstration of intermediate cells in the healthy human pancreas. Virchows Arch [B] 52:283–287
Cossel L (1987) Intermediärzellen im Pankreas und zelluläre Transformation. Zentralbl Allg Pathol 133:503–516
De Clercq L (1980) Nuclear events in Beta-Cells of young and senescent rat islets in organ cultures. Cell Biol Int Rep 4:817–826
Dore BA, Mc Lean GW, Madge GE, Webb SR (1981) Biphasic development of the postnatal mouse pancreas. Biol Neonate 40:209–217
Fitzgerald PJ, Vinijchaikul K, Carol B, Rosenstock L (1986) Pancreas acinar cell regeneration III. DNA-synthesis of pancreas nuclei as indicated by3H-Thymidine autoradiography. Am J Pathol 52:1039–1065
Hellerström C (1977) Growth pattern of pancreasic islets in animals. In: Volk BW, Wellman KF (eds) The diabetic pancreas. Plenum Publishing Corperation. New York, pp 61–97
Hellerström C (1984) The life story of the pancreatic Beta-cell. Diabetologia 26:393–400
Hellerström C, Swenne I (1985) Growth pattern of the islet organ. In: Volk BW, Aquilla ER (eds) The diabetic pancreas. Plenum Medical Bock Company, New York London, pp 53–79
Kern HF, Kern D (1968) Symposium der Deutschen Gesellschaft für Endokrinologie. 14:186–196
Kern HF, Logothetopoulos J (1970) Steroid diabetes in the guinea pig. Studies on islet-cell ultrastructure and regeneration. Diabetes 19:145–154
Klöppel G (1981) Endokrines Pankreas und Diabetes. In: Doerr W, Seiffert G (eds) Pathologie der endokrinen Organe. Bd. 11 /I. Springer Berlin Heidelberg New York, pp 523–728
Klöppel G (1983) Immunocytochemical morphometry of the endocrine pancreas in obese and non obese type II (non insuline dependent) diabetic patients. Diabetologia 25:171–179
Kruskal WH, Wallis WA (1952) Use of ranks in one criterion variance analysis. J Am Statist Assoc 47:614–617
Lazarus SS (1959) Pancreatic adaptation to diabetogenic hormones. Arch Pathol 67:456–467
Like AA, Chick WL (1970) Studies on the diabetic mutant mouse. I. Light microscopy and radioautography of pancreatic islets. Diabetologia 6:207–215
Logothetopoulos J (1972) Islet cell regeneration and neogenesis. In: Handbook of physiology, vol. 1, sect. 7. Am Physiol Soc Washington, pp 67–76
Lucke S, Ziegler B, Komolow I, Hahn HJ (1976) Beziehung zwischen Markierungsindex und Thymidineinbau in kultivierten Langerhansschen Inseln der Ratte. Stimulation der DNSSynthese durch FGH-S3. Acta Histochem 79:33–41
Lütcke H, Scheele GA, Kern HF (1987) Time course and cellular site of mitotic activity in the endocrine pancreas of the rat during susstained hormone stimulation. Cell Tiss Res 247:385–391
McEvoy RC (1981) Changes in the A-, B-, and D-cell population in the pancreatic islet during the postnatal development of the rat. Diabetes 30:813–817
Oates PS, Morgan RGH (1986) Changes in pancreatic acinar cell nuclear and DNA content during aging in the rat. Am J Anat 177:547–554
Rogers AW (1979) Techniques of Autoradiography. Elsevier North Holland Biomedical Press, Amsterdam New York Oxford
Schultze B (1969) Autoradiographie. Physikalische Methoden in der Biologie. Bd. III, Academic Press, New York
Swenne I (1982) The role of glucose in the in vitro regulation of cell cycle kinetics and proliferation of fetal pancreatic B-cells. Diabetes 31:754–760
Swenne I (1983) Effects of aging on the regenerative capacity of the pancreatic B-cell of the rat. Diabetes 32:14–19
Tsubouchi S, Kano E, Suzuki H (1987) Demonstration of expanding cell populations in mouse pancreatic acini and islets. Anat Rec 218:111–115
Warrens S, Root HF (1925) The pathology of diabets with special reference to pancreatic regeneration. Am J Pathol 1:415–429
Author information
Authors and Affiliations
Additional information
This study is part of the research project HFR 22 and was supported by the Health Departement of the GDR
Rights and permissions
About this article
Cite this article
Müller, R., Laucke, R., Trimper, B. et al. Pancreatic cell proliferation in normal rats studied by in vivo autoradiography with3H-Thymidine. Virchows Archiv B Cell Pathol 59, 133–136 (1990). https://doi.org/10.1007/BF02899397
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02899397