Summary
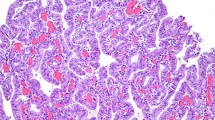
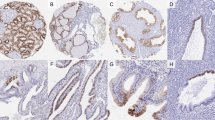
We studied the expression of villin, a microfilament-associated, actin-binding protein typical of brush-border microvilli, in a variety of human carcinomas by applying immunofluorescence microscopy to frozen sections and immunoblotting methods to tissue extracts using a rabbit antiserum and a monoclonal antibody specific for villin. All of the 24 primary and metastatic colorectal adenocarcinomas tested were uniformly and strongly positive for villin, with the immunocytochemical labeling concentrated at the luminal cell margin. In poorly differentiated tumor areas, rudimentary tubules were stained. All of the six tubular adenocarcinomas of the stomach studied as well as two adenocarcinomas of the gall bladder and a hepatocellular carcinoma were also villin-positive. Villin was detectable in 12 of 14 adenocarcinomas of the pancreas; in some of these cases, its distribution was heterogeneous. Among 21 renal cell carcinomas investigated, positivity for villin was seen in nine of 13 clear cell tumors (especially those of grade II), and in all four chromophilic cell tumors; however, all four chromophobe cell tumors studied were negative. Four of 11 endometrial but none of nine ovarian carcinomas were (uniformly or focally) villin positive. Of 18 adenocarcinomas of the lung studied, one was uniformly and four focally positive for villin, while the remainder were negative. All of the other epithelial tumors studied, including 12 adenocarcinomas of the breast and seven epithelial or biphasic pleural mesotheliomas, were villin negative. Our results show that the expression of villin in intestinal epithelial cells is consistently maintained in their corresponding carcinomas, even when the organized brush-border structure has been lost. The presence of villin in some endometrial and pulmonary adenocarcinomas — in contrast to its absence in the respective normal epithelia — suggests that this protein is newly expressed during hyperplasia, dysplasia, or carcinogenesis. Determining the presence or absence of villin and its immunocytochemical staining pattern in metastatic adenocarcinomas may be of some help in determining the type and site of the primary tumor.
Similar content being viewed by others
References
Achtstätter T, Moll R, Anderson A, Kuhn C, Pitz S, Schwechheimer K, Franke WW (1986) Expression of glial filament protein (GFP) in nerve sheaths and non-neural cells re-examined using monoclonal antibodies, with special emphasis on the co-expression of GFP and cytokeratins in epithelial cells of human salivary gland and pleomorphic adenomas. Differentiation 31:206–227
Atkinson BF, Ernst CS, Herlyn M, Steplewski Z, Sears HF, Koprowski H (1982) Gastrointestinal cancer-associated antigen in immunoperoxidase assay. Cancer Res 42:4820–4823
Bast RC Jr, Feeney M, Lazarus H, Nadler LM, Colvin RB, Knapp RC (1981) Reactivity of monoclonal antibody with human ovarian carcinoma. J Clin Invest 68:1331–1337
Bast RC Jr, Klug TL, John ES, Jenison E, Niloff JM, Lazarus H, Berkowitz RS, Leavitt T, Griffiths T, Parker L, Zurowski VR Jr, Knapp RC (1983) A radioimmunoassay using a monoclonal antibody to monitor the course of epithelial ovarian cancer. New Engl J Med 309:883–887
Bretscher A, Weber K (1979) Villin: the major microfilamentassociated protein of the intestinal microvillus. Proc Natl Acad Sci USA 76:2321–2325
Bretscher A, Weber K (1980) Villin is a major protein of the microvillus cytoskeleton which binds both G and F actin in a calcium-dependent manner. Cell 20:839–847
Bretscher A, Osborn M, Wehland J, Weber K (1981) Villin associates with specific microfilamentous structures as seen by immunofluorescence microscopy on tissue sections and cells microinjected with villin. Exp Cell Res 135:213–219
Burnette WN (1981) “Western Blotting”: Electrophoretic transfer of proteins from sodium dodecyl sulfate polyacrylamide gels to unmodified nitrocellulose and radiographic detection with antibody and radioiodinated protein A. Anal Biochem 112:195–203
Charpin C, Bhan AK, Zurawski VR Jr, Scully RE (1982) Carcinoembryonic antigen (CEA) and carbohydrate determinant 19-9 (CA 19-9) localization in 121 primary and metastatic ovarian tumors: an immunohistochemical study with the use of monoclonal antibodies. Int J Gynecol Pathol 1:231–245
Coudrier E, Reggio H, Louvard D (1983) Characterization of an integral membrane glycoprotein associated with the microfilaments of pig intestinal microvilli. EMBO J 2:469–475
Denk H, Tappeiner G, Eckerstorfer R, Holzner JH (1972) Carcinoembryonic antigen (CEA) in gastrointestinal and extragastrointestinal tumors and its relationship to tumor-cell differentiation. Int J Cancer 10:262–272
Dietel M, Arps H, Klapdor (1985) Morphogisch-klinische Korrelation der Tumormarker CA 19-9, CA 125 und CEA bei Pankreaskarzinomen. In: Greten H, Klapdor R (eds) Neue tumorassoziierte Antigene. Georg Thieme, Stuttgart, New York, pp 78–85
Drenckhahn D, Mannerz HG (1983) Distribution of actin and the actin-associated proteins myosin, tropomyosin, alphaactinin, vinculin, and villin in rat and bovine exocrine glands. Eur J Cell Biol 30:167–176
Dudouet B, Robine S, Huet C, Sahuquillo-Merino C, Blair L, Coudrier E, Louvard D (1987) Changes in villin synthesis and subcellular distribution during intestinal differentiation of HT29-18 clones. J Cell Biol 105:359–369
Franke WW, Appelhans B, Schmid E, Freudenstein C, Osborn M, Weber K (1979) Identification and characterization of epithelial cells in mammalian tissues by immunofluorescence microscopy using antibodies to prekeratin. Differentiation 15:7–25
Gatter KC, Abdulaziz Z, Beverley P, Corvalan JRF, Ford C, Lane EB, Mota M, Nash JRG, Pulford K, Stein H, TaylorPapadimitriou J, Woodhouse C, Mason DY (1982) Use of monoclonal antibodies for the histopathological diagnosis of human malignancy. J Clin Pathol 35:1253–1267
Goldenberg DM, Sharkey RM, Primus FJ (1978) Immunocytochemical detection of carcinoembryonic antigen (CEA) in conventional histopathologic specimens. Cancer 42:1546–1553
Gröne H-J, Weber K, Helmchen U, Osborn M (1986) Villin a marker of brush border differentiation and cellular origin in human renal cell carcinomas. Am J Pathol 124:294–302
Henderson DW, Papadimitriou JM, Coleman M (1986) Ultrastructural appearances of tumours. Diagnosis and classification of human neoplasia by electron microscopy. Churchill Livingstone, Edinburgh, 2nd edition, pp 88–112 and 133–138
Iida F, Kusama J (1982) Gastric carcinoma and intestinal metaplasia. Significance of types of intestinal metaplasia upon development of gastric carcinoma. Cancer 50:2854–2858
Kabawat SE, Bast RC Jr, Welch WR, Knapp RC, Colvin RB (1983) Immunopathologic characterization of a monoclonal antibody that recognizes common surface antigens of human ovarian tumors of serous, endometrioid, and clear cell type. Am J Clin Pathol 79:98–104
Koprowski H, Steplewski Z, Mitchell K, Herlyn M, Herlyn D, Fuhrer JP (1979) Colorectal carcinoma antigens detected by hybridoma antibodies. Somatic Cell Genet 5:957–972
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature (London) 227:680–685
Marcus PB (1981) Glycocalyceal bodies and their role in tumor typing. J Submicroscop Cytol 13:483–500
McNutt AM, Bolen JW, Gown AM, Hamar SP, Vogel AM (1985) Coexpression of intermediate filaments in human epithelial neoplasms. Ultrastruct Pathol 9:31–43
Moll R, Franke WW, Schiller DL, Geiger B, Krepier R (1982) The catalog of human cytokeratin polypeptides: Patterns of expression of specific cytokeratins in normal epithelia, tumors, and cultured cells. Cell 31:11–24
Moll R, Franke WW (1986) Cytochemical cell typing of metastatic tumors according to their cytoskeletal proteins. In: Lapis K, Liotta LA, Rabson AS (eds) Biochemistry and molecular genetics of cancer metastasis. Martinus Nijhoff Publ, Boston, pp 101–114
Osborn M, van Lessen G, Weber K, Klöppel G, Altmannsberger M (1986) Differential diagnosis of gastrointestinal carcinomas by using monoclonal antibodies specific for individual keratin polypeptides. Lab Invest 55:497–504
Pitz S, Moll R, Störkel S, Thoenes W (1987) Expression of intermediate filament proteins in subtypes of renal cell carcinomas and in renal oncocytomas: Distinction of two classes of renal cell tumors. Lab Invest 56:642–653
Robine S, Huet C, Moll R, Sahuquillo-Merino C, Coudrier E, Zweibaum A, Louvard D (1985) Can villin be used to identify malignant and undifferentiated normal digestive epithelial cells? Proc Natl Acad Sci USA 82:8488–8492
Shibayama T, Carboni JM, Mooseker MS (1987) Assembly of the intestinal brush border: Appearance and redistribution of microvillar core proteins in developing chick enterocytes. J Cell Biol 105:335–344
Störkel S, Rumpelt HJ, Jacobi G, Lippold R, Thoenes W (1986) Prognosekriterien beim Nierenzellkarzinom. Verh Dtsch Ges Pathol 70:635
Thoenes W, Störkel S, Rumpelt JH (1985) Human chromophobe cell renal carcinoma. Virchows Arch [Cell Pathol] 48:207–217
Thoenes W, Störkel S, Rumpelt HJ (1986 a) Histopathology and classification of renal cell tumors (adenomas, oncocytomas and carcinomas): the basic cytological and histopathological elements and their use for diagnosis. Pathol Res Pract 181:125–143
Thoenes W, Störkel S, Rumpelt HJ (1986b) Das Nierenzellkarzinom. Eine Systematik aufgrund zytomorphologischer Merkmale. Zentralbl Allg Pathol 132:503–513
Thompson DMP, Krupey J, Freedman SO, Gold P (1969) The radioimmunoassay of circulating carcinoembryonic antigen of the human digestive system. Proc Natl Acad Sci USA 64:161–167
Weber K, Glenney Jr, JR (1982) Calcium-modulated multifunctional proteins regulating F-actin organization. Cold Spring Harbor Symp Quant Biol 46:541–552
West AB, Isaac CA, Carboni JM, Morrow JS, Mooseker MS, Barwick KW (1987) Localization of villin, a cytoskeletal protein specific to microvilli, in human ileum and colon and in colonie neoplasms. Gastroenterology (in press)
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Moll, R., Robine, S., Dudouet, B. et al. Villin: a cytoskeletal protein and a differentiation marker expressed in some human adenocarcinomas. Virchows Archiv B Cell Pathol 54, 155–169 (1987). https://doi.org/10.1007/BF02899208
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02899208