Summary
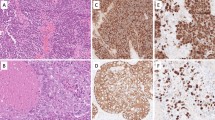
We undertook an immunohistochemical analysis of human bronchopulmonary epithelial neoplasms and pleural mesotheliomas using a monoclonal antibody which recognizes ras oncogene products (p21ras). The monoclonal antibody, RAP-5, recognizes both unaltered and certain mutated p21ras. Formalin fixed and paraffin embedded tissue samples of 187 lung epithelial tumors and 27 pleural mesotheliomas were investigated; normal and bronchiectatic lungs were similarly studied. Normal lung and pleural tissue did not immunostain except for occasional type II pneumocytes. Reactive type II pneumocytes adjacent to carcinomas and bronchiectasis immunostained consistently. Twenty four/34 (71%) squamous carcinomas immunostained. Only 8/50 (16%) adenocarcinomas immunostained focally and weakly whereas 19/24 (79%) bronchioloalveolar carcinomas immunostained. Eleven/18 (61%) large cell carcinomas immunostained with variable intensity. Eleven/13 (85%) carcinoids, 6/7 (85%) well differentiated neuroendocrine carcinomas, and 18/21 (86%) intermediate cell neuroendocrine carcinomas immunostained while none of 20 small cell neuroendocrine carcinomas immunostained. Only a few mesotheliomas were immunostained focally. Two/14 (14%) epithelial type and 1/9 (11%) biphasic type mesotheliomas immunostained weakly; none of 4 spindle cell mesotheliomas immuno stained.
We conclude that while at least occasional cases of most types of pulmonary epithelial neoplasms express p21ras, the frequency and intensity of the expression are distinctly greater in certain tumor types such as squamous, bronchioloalveolar, and neuroendocrine neoplasm except for the small cell type. Contrary to these lung epithelial neoplasms, most mesotheliomas did not immunostain for p21ras. Whether the enhanced p21ras expression may point to a different mechanism of transformation or may merely reflect differentiation features remains undetermined.
Similar content being viewed by others
References
Bishop JM (1983) Cellular oncogenes and retroviruses. Annu Rev Biochem 52:301–354
Blair DG, Oskarsson M, Wood TG, McClements WL, Fischinger PJ (1981) Activation of the transforming potential of a normal cell sequence: a molecular model for oncogenesis. Science 212:941–943
Blobel GA, Gould VE, Moll R, Lee I, Huszar M, Geiger B, Franke WW (1985) Coexpression of neuroendocrine markers and epithelial cytoskeletal proteins in bronchopulmonary neuroendocrine neoplasms. Lab Invest 52:39–51
Blobel GA, Moll R, Franke WW, Kayser KW, Gould VE (1985) The intermediate filament cytoskeleton of malignant mesotheliomas and its diagnostic significance. Am J Pathol 121: 235–247
Blobel GA, Moll R, Franke WW, Vogt-Mykopf I, Gould VE (1987) Cytoskeletal characterization of normal and transformed lung epithelial cells: Improved histodiagnosis of carcinomas of the lung. Klin Wochenschr (in press)
Carter D, Eggleston J (1979) Tumors of the lower respiratory tract. Atlas of tumor pathology, Fascicle 17, Armed Forces Institute of Pathology, Washington DC, USA
DeFeo D, Gonda MA, Young HA, Chang EH, Lowy DR, Scolnick EM, Ellis RW (1981) Analysis of two different rat genomic clones homologous to the transforming gene of Harvey murine sarcoma virus. Proc Natl Acad Sci (USA) 78: 3328–3332
Der CJ, Cooper GM (1983) Altered gene products are associated with activation of cellular ras genes in human lung and colon carcinomas. Cell 32:201–208
Fujita J, Srivastava S, Kraus M, Rhim J, Tronick S, Aaronson S (1984) Frequency of molecular alteration affecting ras proto-oncogenes in human urinary tract tumors. Proc Natl Acad Sci (USA) 82:3849–3853
Gallick GE, Kurzrock R, Kloetzer WS, Arlinghaus RB, Gutterman JU (1985) Expression of p21ras in fresh primary and metastatic human colorectal tumors. Proc Natl Acad Sci (USA) 82:1795–1799
Gould VE, Linnoila RL, Memoli VA, Warren WH (1983) Neuroendocrine components of the bronchopulmonary tract: hyperplasias, dysplasias, and neoplasms. Lab Invest 49:5, 519–537
Gould VE, Linnoila RI, Memoli VA, Warren WH (1983) Neuroendocrine cells and Neuroendocrine neoplasms of the lung. Pathol Annu 18:287–330
Hand PH, Thor A, Wunderlich D, Muraro R, Caruso A, Schlom J (1984) Monoclonal antibodies of predifined specificity detect activated ras gene expression in human mammary and colonic carcinomas. Proc Natl Acad Sci (USA) 81:5227–5231
Hsu SM, Raine L, Fanger H (1981) The use of avidin-biotin peroxidase complex (ABC) in immunoperoxidase techniques: A comparison between ABC and unlabeled antibody (PAP) procedures. J Histochem Cytochem 29:577–581
Kurzrock R, Gallick GE, Gutterman JU (1986) Differential expression of p21ras gene products among histological subtypes of fresh primary human lung tumors. Cancer Res 46: 1530–1534
Lacal JC, Srivastava SK, Anderson PS, Aaronson SA (1986) Ras p21 proteins with high or low GTPase activity can efficiently transform NIH/3T3 cells. Cell 44:609–617
Land H, Parada LF, Weingerg RA (1983) Cellular oncogenes and multistep carcinogenesis. Science 22:771–778
Lee I, Radosevich JA, Ma Y, Combs SG, Rosen ST, Gould VE (1985) Immunohistochemical analysis of pulmonary carcinomas using monoclonal antibody 44-3A6. Cancer Res 45: 5808–5812
Lee I, Radosevich JA, Chejfec G, May Y, Warren WH, Gould VE (1986) Mesotheliomas: Improved differential diagnosis from metastatic lung carcinomas using monoclonal antibodies 44-3A6 and 624A12. Am J Pathol 123:497–507
Lee I, Radosevich JA, Ma Y, Warren WH, Rosen ST, Gould VE (1987) Immunohistochemical demonstration of lacto-N-fucopentose III in lung carcinomas with monoclonal antibody 624A12. Pathol Res Pract 182:40–47
Lee I, Gould KA, Radosevich JA, Schlom J, Banner BF, Gould VE (1987) Differential expression of ras oncogene products in liver diseases. Lab Invest 56:42(A)
McCoy MS, Toole JJ, Cunningham JM, Chang EH, Lowy DR, Weinberg RA (1983) Characterization of a human colon/lung carcinoma oncogene. Nature (Lond) 302:79–81
Piehl MR, Lee I, Ma Y, Warren WH, Radosevich JA, Rosen ST, Gould VE (1987) Subsets of pulmonary large cell undifferentiated carcinomas defined immunohistochemically. Lab Invest 56:60 (A)
Pulciani S, Santos E, Lauver AV, Long LK, Aaronson SA, Barbacid (1982) Oncogenes in solid human tumors. Nature (Lond) 300:539–542
Reddy EP, Reynolds RK, Santos E, Barbacid M (1982) A point mutation is responsible for the acquisition of transforming proterties by T24 human bladder carcinoma oncogene. Nature (Lond) 300:149–152
Rosen ST, Mulshine JL, Cuttitta F, Fedorke J, Carnewy DN, Gazdar AF, Minna JD (1984) Analysis of human small cell lung cancer differentiation antigens using a panel of rat monoclonal antibodies. Cancer Res 44:2052–2061
Santos E, Martin-Zanca D, Reddy EP, Pierotti MA, Porta GD, Barbacid M (1984) Malignant activation of a K-ras oncogene in lung carcinoma but not in normal tissue of the same patient. Science 223:661–664
Slamon DJ, DeKernion JB, Verma IM, Cline MJ (1984) Expression of cellular oncogenes in human malignancies. Science 24:256–262
Smith MR, DeGudicibus SJ, Stacey DW (1986) Requirement of c-ras proteins during viral oncogene transformation. Nature (Lond) 320:540–543
Spandidos DA, Wilkie NM (1984) Malignant transformation of early passage rodent cells by a single mutated human oncogene. Nature (Lond) 310:469–475
Taparowsky E, Suard Y, Fasano O, Shimizu K, Goldfarb M, Wigler M (1982) Activation of the T24 bladder carcinoma transforming gene is linked to a single amino acid change. Nature (Lond) 300:762–764
Thor A, Hand W, Wunderlich D, Caruso A, Muraro R, Schlom J (1984) Monoclonal antibodies define differential ras gene expression in malignant and benign colonie diseases. Nature (Lond) 311:562–565
Thor A, Ohuchi N, Hand PH, Callahan R, Weeks MO, Theillet C, Lidereau R, Escot C, Page DL, Vilasi V, Schlom J (1986) Ras oncogene alterations and enhanced levels of ras p21 expression in a spectrum of benign and malignant human mammary tissue. Lab Invest 55:603–615
Viola MV, Fromowitz, Oravez S, Deb S, Finkel G, Lundy J, Hand P, Thor A, Schlom J (1986) Expression of ras oncogene p21 in prostate cancer. N Engl J Med 314:133–137
Warren WH, Memoli VA, Kittle CF, Jensik RJ, Faber LP, Gould VE (1984) The biological implications of bronchial tumors. J Thorac Cardiov Surg 87:274–282
World Health Organization Histological typing of Lung Tumors (1982) Second Edition. Am J Clin Pathol 77:123–136
Author information
Authors and Affiliations
Additional information
This work was partly supported by a Veterans Administration Merit Review Grant and the Otho S. Sprague Memorial Fund
Rights and permissions
About this article
Cite this article
Lee, I., Gould, V.E., Radosevich, J.A. et al. Immunohistochemical evaluation of ras oncogene expression in pulmonary and pleural neoplasms. Virchows Archiv B Cell Pathol 53, 146–152 (1987). https://doi.org/10.1007/BF02890237
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02890237