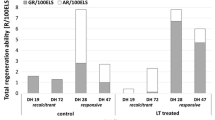
Summary
Microspore-derived embryos fromBrassica napus cv. Topas (low erucic acid) and Reston (high erucic acid) were subjected to treatment with abscisic acid (ABA) during late-stage embryo development and then dried under controlled relative humidities to mature dry seed levels of moisture. Exogenously medium-supplied ABA arrested growth and development, reduced moisture content, increased total fatty acids on a dry weight basis, and stimulated systhesis of proteins in microspore-derived embryos. ABA also resulted in a higher proportion of 22∶1 in cv. Reston (high 22∶1) and increased the level of fatty acid unsaturation in cv. Topas (low 22∶1). The accumulation of two proteins that co-migrated with cruciferin and napin on sodium dodecyl sulfate-polyacrylamide gel electrophoresis and two-dimensional gels were also promoted by exposure to ABA, and the degree of accumulation was dependent on the concentration and time of application of ABA. Controlled desiccation of microspore embryos, used to simulate normal maturation and dehydration of zygotic embryos during seed development, did not seem to cause an increase of either storage proteins, total fatty acids, or 22∶1 (in cv. Reston), suggesting that dehydration is not a prerequisite for these processes, at least in culturedBrassica embryos.
Similar content being viewed by others
References
Ajvioglu, A.; Knox, R. B. Storage of lipid accumulation by zygotic and somatic embryos in culture. Ann. Bot. 63:409–420; 1989.
Black, M. Involvement of ABA in the physiology of developing and mature seeds. In: Davies, W. J.; Jones, H. G., ed. Abscisic acid, physiology and biochemistry. Oxford, U.K.: Bios Scientific Publishers; 1991:99–124.
Brown, D. C. W.; Watson, E. M.; Pechan, P. M. Induction of desiccation tolerance in microspore derived embryos ofBrassica napus. In Vitro Cell. Dev. Biol. 29P:113–118; 1993.
Browse, J.; McCourt, P. J.; Somerville, C. R.: Fatty acid composition of leaf lipids determined after combined digestion and fatty acid methyl ester formation from fresh tissue. Anal. Biochem. 152:141–145; 1986.
Davies, W. J.; Jones, H. C., editors. Abscisic acid, physiology and biochemistry. Oxford, U.K.: Bios Scientific Publishers, 1991.
Finkelstein, R.; Somerville, C. Abscisic acid or osmoticum promote accumulation of long chain fatty acids in developing embryos ofBrassica napus. Plant Sci. 61:213–217; 1989.
Finkelstein, R.; Tenbarge, K. M.; Shumway, J. E., et al. Role of ABA in maturation of rapeseed embryos. Plant Physiol. 78:630–636; 1985.
Gamborg, O. L.; Miller, R. A.; Ojima, L. Nutrient requirements of suspension cultures of soybean root cells. Exp. Cell. Res. 50:151–158; 1968.
Holbrook, L.; Magus, J. R.; Taylor, D. C. Abscisic acid induction of elongase activity, biosynthesis, and accumulation of very long monoun-saturated fatty acids and oil body proteins in microspore derived embryos ofBrassica napus L cv. Reston. Plant Sci 84:99–115; 1992.
Huang, B.; Keller, W. A. Microspore culture technology. J. Tissue Cult. Methods 12:171–178; 1989.
Kim, Y.-H.; Janick, J. Abscisic acid and proline improve desiccation tolerance and increase fatty acid content of celery somatic embryos. Plant Cell Tissue Organ Cult. 24:83–89; 1991.
Laemmli, U. K. Cleavage of structural proteins during assembly of the head of bacteriophage T4. Nature 227:680–685; 1970.
Lichter, R. Anther culture ofBrassica napus in a liquid medium. Z. Pflanzenzüchtg. 103:229–237; 1981.
O'Farrell, P. H. High resolution two-dimensional electrophoresis of proteins. J. Biol. Chem. 250:4007–4021; 1975.
Pomeroy, M. K.; Kramer, J. K. G.; Hunt, D. J., et al. Fatty acid changes during development of zygotic and microspore-derived embryos ofBrassica napus. Physiol. Plant. 81:447–454; 1991a.
Pomeroy, M. K.; Sparace, S. A.; Keller, W. A. Fatty acid and triglycerol biosynthesis in microspore embryos ofBrassica napus. In: McGregor, D. I., ed. Rapeseed in a changing world. GCIRC Eighth Int'l. Rapeseed Congress, Publisher, Saskatoon, Canada. 1991b:154–158.
Saranga, Y.; Kim, Y.-H.; Janick, J. Changes in tolerance to partial desiccation and in metabolite content of celery somatic embryos induced by reduced osmotic potential. J. Am. Soc. Hortic. Sci. 117: 342–345; 1992a.
Saranga, Y.; Rhodes, D.; Janick, J. Changes in amino acid composition associated with tolerance to partial desiccation of celery somatic embryos. J. Am. Soc. Hortic. Sci. 117:337–342; 1992b.
Senaratna, T.; Kott L.; Beversdorf, W. D., et al. Desiccation of microspore derived embryos of oilseed rape (Brassica napus L.). Plant Cell Rep. 10:342–344; 1991.
Senaratna, T.; McKersie, B. D.; Bowley, S. R. Artificial seeds of alfalfa (Medicago sativa L.): induction of dessication tolerance in somatic embryos. In Vitro Cell. Dev. Biol. 26:85–90; 1990.
Takahata, Y.; Wakui, K.; Kaizuma, N., et al. A dry artificial seed system forBrassica crops. Acta Hortic. 319:317–322; 1992.
Takahata, Y.; Brown, D. C. W.; Keller, W. A., et al. Dry artificial seeds and desiccation tolerance induction in microspore-derived embryos of broccoli. Plant Cell Tissue Organ Cult. 35:121–129; 1993.
Taylor, D. C.; Weber, N.; Underhill, E. W., et al. Storage protein regulation and lipid accumulation in microspore embryos ofBrassica napus L. Planta 181:18–26; 1990.
Walker-Simmons, M. ABA levels and sensitivity in developing wheat embryos of sprouting resistant and sensitive cultivars. Plant. Physiol. 84:61–66; 1987.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Pomeroy, K., Brown, D.C.W. & Takahata, Y. Response ofBrassica napus L. Microspore-derived embryos to exogenous abscisic acid and desiccation. In Vitro Cell.Dev.Biol.–Plant 30, 196–203 (1994). https://doi.org/10.1007/BF02823032
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02823032