Abstract
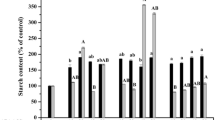
Efficient protocols for somatic embryogenesis of papaya (Carica papaya L.) have great potential for selecting elite hybrid genotypes. Addition of polyethylene glycol (PEG), a nonplasmolyzing osmotic agent, to a maturation medium increases the production of somatic embryos in C. papaya. To study the effects of PEG on somatic embryogenesis of C. papaya, we analyzed somatic embryo development and carbohydrate profile changes during maturation treatments with PEG (6%) or without PEG (control). PEG treatment (6%) increased the number of normal mature somatic embryos followed by somatic plantlet production. In both control and PEG treatments, pro-embryogenic differentiation to the cotyledonary stage was observed and was significantly higher with PEG treatment. Histomorphological analysis of embryonic cultures with PEG revealed meristematic centers containing small isodiametric cells with dense cytoplasm and evident nuclei. Concomitant with the increase in the differentiation of somatic embryos in PEG cultures, there was an increase in the endogenous content of sucrose and starch, which appears to be related to a rising demand for energy, a key point in the conversion of C. papaya somatic embryos. The endogenous carbohydrate profile may be a valuable parameter for developing optimized protocols for the maturation of somatic embryos in papaya.




Similar content being viewed by others
References
Anandan R, Sudhakar D, Balasubramanian P, Gutieirrez-Mora A (2012) In vitro somatic embryogenesis from suspension cultures of Carica papaya L. Sci Hortic 136:43–49. https://doi.org/10.1016/j.scienta.2012.01.003
Aragão VPM, Ribeiro YRS, Reis RS, Macedo AF, Floh EIS, Silveira V, Santa-Catarina C (2015) In vitro organogenesis of Cedrela fissilis Vell. (Meliaceae): the involvement of endogenous polyamines and carbohydrates on shoot development. Plant Cell Tissue Organ Cult 124(3):611–620. https://doi.org/10.1007/s11240-015-0919-8
Arnhold E (2013) Pacote em ambiente R para análise de variância e análises complementares. Braz J Vet Res Anim Sci 50(6):488–492
Blanc G, Lardet L, Martin A, Jacob J, Carron M (2002) Differential carbohydrate metabolism conducts morphogenesis in embryogenic callus of Hevea brasiliensis (Müll. Arg.). J Exp Bot 53(373):1453–1462. https://doi.org/10.1093/jexbot/53.373.1453
Bruijne E, Langhe E, Rijck RV (1974) Actions of hormones and embryoid formation in callus cultures of Carica papaya. International Symposium on Crop Protection, Fytopharmacie en Fytiatrie Rijkslandbouwhoogeskool Medelingen 39:637–645
Businge E, Bygdell J, Wingsle G, Moritz T, Egertsdotter U (2013) The effect of carbohydrates and osmoticum on storage reserve accumulation and germination of Norway spruce somatic embryos. Physiol Plant 149(2):273–285. https://doi.org/10.1111/ppl.12039
Cangahuala-Inocente GC, Steiner N, Santos M, Guerra MP (2004) Morphohistological analysis and histochemistry of Feijoa sellowiana somatic embryogenesis. Protoplasma 224(1–2):33–40. https://doi.org/10.1007/s00709-004-0055-5
Colvin HW Jr, Attebery J, Ivy J (1961) Comparison of the anthrone reagent and a copper-reduction method for determining blood sugar in calves. J Dairy Sci 44(11):2081–2088. https://doi.org/10.3168/jds.S0022-0302(61)90022-4
Correia SM, Canhoto JM (2010) Characterization of somatic embryo attached structures in Feijoa sellowiana Berg. (Myrtaceae). Protoplasma 242(1–4):95–107. https://doi.org/10.1007/s00709-010-0130-z
de Feria M, Jiménez E, Barbón R, Capote A, Chávez M, Quiala E (2003) Effect of dissolved oxygen concentration on differentiation of somatic embryos of Coffea arabica cv. Catimor 9722. Plant Cell, Tissue Organ Cult 72(1):1–6. https://doi.org/10.1023/A:1021202305692
El Dawayati MM, Bar AE, Ola H, Zaid ZE, Zein El Din AF (2012) In vitro morpho-histological studies of newly developed embryos from abnormal malformed embryos of date palm cv. Gundila under desiccation effect of polyethelyne glycol treatments. Ann Agric Sci 57(2):117–128. https://doi.org/10.1016/j.aoas.2012.08.005
Eveland AL, Jackson DP (2012) Sugars, signalling, and plant development. J Exp Bot 63(9):3367–3377. https://doi.org/10.1093/Jxb/Err379
Gibson SI (2005) Control of plant development and gene expression by sugar signaling. Curr Opin Plant Biol 8(1):93–102. https://doi.org/10.1016/j.pbi.2004.11.003
Gupta RB, Khan K, Macritchie F (1993) Biochemical basis of flour properties in bread wheats. I. Effects of variation in the quantity and size distribution of polymeric protein. J Cereal Sci 18(1):23–41. https://doi.org/10.1006/jcrs.1993.1031
He Y, Young TE, Clark KR, Kleppinger-Sparace KF, Bridges WC, Sparace SA (2011) Developmental profile of storage reserve accumulation in soybean somatic embryos. In Vitro Cell Dev Biol Plant 47(6):725–733. https://doi.org/10.1007/s11627-011-9375-2
Heringer AS, Vale EM, Barroso T, Santa-Catarina C, Silveira V (2013) Polyethylene glycol effects in somatic embryogenesis of papaya hybrid UENF/CALIMAN01 seeds. Theor Exp Plant Physiol 25(2):116–124. https://doi.org/10.1590/S2197-00252013000200004
Hudec L, Konrádová H, Hašková A, Lipavská H (2016) Norway spruce embryogenesis: changes in carbohydrate profile, structural development and response to polyethylene glycol. Tree Physiol 36(5):548–561. https://doi.org/10.1093/treephys/tpw016
James MG, Denyer K, Myers AM (2003) Starch synthesis in the cereal endosperm. Curr Opin Plant Biol 6(3):215–222
Kärkönen A (2000) Anatomical study of zygotic and somatic embryos of Tilia cordata. Plant Cell, Tissue Organ Cult 61(3):205–214. https://doi.org/10.1023/A:1006455603528
Kaur D, Ogra RK, Bhattacharya A, Sood A (2012) Changes in sugar levels during slow growth of Dendrocalamus hamiltonii somatic embryos due to liquid paraffin overlay. In Vitro Cell Dev Biol Plant 48(1):120–126. https://doi.org/10.1007/s11627-011-9392-1
Kim T, Anbazhagan VR, Park J (2005) Somatic embryogenesis in Schisandra chinensis (Turcz.) Baill. In Vitro Cell Dev Biol Plant 41(3):253–257. https://doi.org/10.1079/IVP2004630
Kubeš M, Drážná N, Konrádová H, Lipavská H (2014) Robust carbohydrate dynamics based on sucrose resynthesis in developing Norway spruce somatic embryos at variable sugar supply. In Vitro Cell Dev Biol Plant 50(1):45–57
Lai FM, McKersie BD (1994) Regulation of starch and protein accumulation in alfalfa (Medicago sativa L.) somatic embryos. Plant Sci 100(2):211–219. https://doi.org/10.1016/0168-9452(94)90078-7
Langhansova L, Marsik P, Vanek T (2005) Production of saponins from Panax ginseng suspension and adventitious root cultures. Biol Plant 49(3):463–465. https://doi.org/10.1007/s10535-005-0030-9
Li H, Li F-l Du, J-c LuH, Z-q He (2008) Somatic embryogenesis and histological analysis from zygotic embryos in Vitis vinifera L. ‘Moldova’. For Stud China 10(4):253–258. https://doi.org/10.1007/s11632-008-0040-8
Linossier L, Veisseire P, Cailloux F, Coudret A (1997) Effects of abscisic acid and high concentrations of PEG on Hevea brasiliensis somatic embryos development. Plant Sci 124(2):183–191. https://doi.org/10.1016/s0168-9452(97)04597-4
Lipavská H, Svobodová H, Albrechtová J, Kumstýřová L, Vágner M, Vondráková Z (2000) Carbohydrate status during somatic embryo maturation in Norway spruce. In Vitro Cell Dev Biol Plant 36(4):260–267. https://doi.org/10.1007/s11627-000-0048-9
Liu C, Xu Z, Chua N-H (1993) Auxin polar transport is essential for the establishment of bilateral symmetry during early plant embryogenesis. Plant Cell 5(6):621–630. https://doi.org/10.1105/tpc.5.6.621
Lygin AV, Abdel-Rahman MM, Ulanov AV, Widholm JM, Lozovaya VV (2012) Polyethylene glycol treatment promotes metabolic events associated with maize callus morphogenic competence. Phytochemistry 82:46–55
Malabadi RB, Kumar SV, Mulgund GS, Nataraja K (2011) Induction of somatic embryogenesis in papaya (Carica papaya). Res Biotechnol 2(5):40–55
Martin AB, Cuadrado Y, Guerra H, Gallego P, Hita O, Martin L, Dorado A, Villalobos N (2000) Differences in the contents of total sugars, reducing sugars, starch and sucrose in embryogenic and non-embryogenic calli from Medicago arborea L. Plant Sci 154(2):143–151. https://doi.org/10.1016/S0168-9452(99)00251-4
McCready R, Guggolz J, Silviera V, Owens H (1950) Determination of starch and amylose in vegetables. Anal Chem 22(9):1156–1158. https://doi.org/10.1021/ac60045a016
Mishra M, Shukla N, Chandra R (2010) Role of polyethylene glycol in maturation and germination of transformed somatic embryos of papaya (Carica papaya L.). In: Kumar N, Soorianathasundaram K, Jeyakumar P (eds) Ii International symposium on papaya. Acta Horticulturae, vol 851, Madurai, India
Mishra S, Sanyal I, Amla DV (2012) Changes in protein pattern during different developmental stages of somatic embryos in chickpea. Biol Plant 56(4):613–619. https://doi.org/10.1007/s10535-012-0124-0
Misra S, Attree SM, Leal I, Fowke LC (1993) Effect of abscisic-acid, osmoticum, and desiccation on synthesis of storage proteins during the development of white spruce somatic embryos. Ann Bot 71(1):11–22. https://doi.org/10.1006/anbo.1993.1002
Moussian B, Schoof H, Haecker A, Jürgens G, Laux T (1998) Role of the ZWILLE gene in the regulation of central shoot meristem cell fate during Arabidopsis embryogenesis. EMBO J 17(6):1799–1809
Murashige T, Skoog F (1962) A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol Plant 15(3):473–497. https://doi.org/10.1111/j.1399-3054.1962.tb08052.x
Nieves N, Segura-Nieto M, Blanco MA, Sanchez M, Gonzalez A, Gonzalez JL, Castillo R (2003) Biochemical characterization of embryogenic and non-embryogenic calluses of sugarcane. In Vitro Cell Dev Biol Plant 39(3):343–345. https://doi.org/10.1079/ivp2002391
Norton G, Harris J (1975) Compositional changes in developing rape seed (Brassica napus L.). Planta 123(2):163–174. https://doi.org/10.1007/BF00383865
Padilla-Chacón D, Cordoba E, Olivera T, Sánchez S, Coello P, León P, Tiessen A, Martínez-Barajas E (2010) Heterologous expression of yeast Hxt2 in Arabidopsis thaliana alters sugar uptake, carbon metabolism and gene expression leading to glucose tolerance of germinating seedlings. Plant Mol Biol 72(6):631–641. https://doi.org/10.1007/s11103-010-9602-y
Pescador R, Kerbauy GB, Kraus JE, de Melo Ferreira W, Guerra MP, Rita de Cássia L (2008) Changes in soluble carbohydrates and starch amounts during somatic and zygotic embryogenesis of Acca sellowiana (Myrtaceae). In Vitro Cell Dev Biol Plant 44(4):289–299
R Core Team (2014) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Pinto DLP, de Almeida AMR, Rêgo MM, da Silva ML, de Oliveira EJ, Otoni WC (2011) Somatic embryogenesis from mature zygotic embryos of commercial passionfruit (Passiflora edulis Sims) genotypes. Plant Cell, Tissue Organ Cult 107(3):521–530. https://doi.org/10.1007/s11240-011-0003-y
Saranga Y, Kim Y-H, Janick J (1992) Changes in tolerance to partial desiccation and in metabolite content of celery somatic embryos induced by reduced osmotic potential. J Am Soc Hortic Sci 117(2):342–345. http://journal.ashspublications.org/content/117/2/342
Smulders MJM, Klerk GJ (2011) Epigenetics in plant tissue culture. Plant Growth Regul 63(2):137–146. https://doi.org/10.1007/s10725-010-9531-4
Stasolla C, Yeung EC (2003) Recent advances in conifer somatic embryogenesis: improving somatic embryo quality. Plant Cell, Tissue Organ Cult 74(1):15–35. https://doi.org/10.1023/a:1023345803336
Stasolla C, van Zyl L, Egertsdotter U, Craig D, Liu W, Sederoff RR (2003a) The effects of polyethylene glycol on gene expression of developing white spruce somatic embryos. Plant Physiol 131(1):49–60. https://doi.org/10.1104/pp.015214
Stasolla C, van Zyl L, Egertsdotter U, Craig D, Liu W, Sederoff RR (2003b) Transcript profiles of stress-related genes in developing white spruce (Picea glauca) somatic embryos cultured with polyethylene glycol. Plant Sci 165(4):719–729. https://doi.org/10.1016/S0168-9452(03)00228-0
Sturm A, Tang G-Q (1999) The sucrose-cleaving enzymes of plants are crucial for development, growth and carbon partitioning. Trends Plant Sci 4(10):401–407. https://doi.org/10.1016/S1360-1385(99)01470-3
Svobodová H, Albrechtová J, Kumstırová L, Lipavská H, Vágner M, Vondráková Z (1999) Somatic embryogenesis in Norway spruce: anatomical study of embryo development and influence of polyethylene glycol on maturation process. Plant Physiol Biochem 37(3):209–221. https://doi.org/10.1016/S0981-9428(99)80036-9
Tautorus TE, Fowke LC, Dunstan DI (1991) Somatic embryogenesis in conifers. Can J Bot 69(9):1873–1899. https://doi.org/10.1139/b91-237
Troch V, Werbrouck S, Geelen D, Van Labeke M-C (2009) Optimization of horse chestnut (Aesculus hippocastanum L.) somatic embryo conversion. Plant Cell, Tissue Organ Cult 98(1):115–123. https://doi.org/10.1007/s11240-009-9544-8
Vale EM, Heringer AS, Barroso T, da Silva Ferreira AT, da Costa MN, Perales JE, Santa-Catarina C, Silveira V (2014) Comparative proteomic analysis of somatic embryo maturation in Carica papaya L. Proteome Sci 12(1):37. https://doi.org/10.1186/1477-5956-12-37
Walker DR, Parrott WA (2001) Effect of polyethylene glycol and sugar alcohols on soybean somatic embryo germination and conversion. Plant Cell, Tissue Organ Cult 64(1):55–62. https://doi.org/10.1023/a:1010601402098
Yie S-T, Liaw S (1977) Plant regeneration from shoot tips and callus of papaya. In vitro 13(9):564–568. https://doi.org/10.1007/BF02627852
Acknowledgements
This research was funded by the Carlos Chagas Filho Foundation for Research Support in the State of Rio de Janeiro (FAPERJ) (Proc. E26/201.574/2014 and E26/110.058/2014) and the National Council for Scientific and Technological Development-CNPq (Proc. 454451/2014-8 and 305415/2016-6). Scholarships were provided by the Coordination for the Improvement of Higher Education Personnel (CAPES) to EMV and LZP and by the FAPERJ to RSR. We kindly thank Caliman Agricola S/A for supplying seeds.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors of the manuscript have no conflict of interest to declare.
Rights and permissions
About this article
Cite this article
Vale, E.M., Reis, R.S., Passamani, L.Z. et al. Morphological analyses and variation in carbohydrate content during the maturation of somatic embryos of Carica papaya. Physiol Mol Biol Plants 24, 295–305 (2018). https://doi.org/10.1007/s12298-017-0501-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12298-017-0501-4