Summary
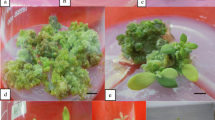
An efficient and reproduciblein vitro culture system has been developed for regeneration of multiple shoot clumps from intact seedlings of both lowland and upland cultivars of switchgrass (Panicum virgatum L.). The multiple shoots were induced on Murashige and Skoog medium supplemented with various combinations of 2,4-dichlorophenoxyacetic acid (2,4-D) and 1-phenyl-3-(1,2,3-thiadiazol-5YL)-urea (thidiazuron or TDZ). Maximum response was obtained with 4.5 μM 2,4-D and 18.2 μM TDZ. These shoots proliferated and rooted efficiently on MS medium without growth regulators. The developmental pattern of the multiple shoots indicated their origin from the enlarged shoot apex via proliferation of axillary buds and subsequent reprogramming of shoot meristems followed by secondary differentiation of adventitious shoots The simplicity of the protocol and direct production of multiple shoots make this a potential system that is highly attractive and amenable for microprojectile-mediated gene transfer.
Similar content being viewed by others
References
Alexandrova, K. S.; Denchev, P. D.; Conger, B. V. In vitro development of inflorescences from switchgrass nodal segments. Crop Sci. 36:175–178; 1996.
Asano, Y.; Katsumoto, H.; Inokuma, S., et al. Cytokinin and thiamine requirements and stimulative effects of riboflavin and α-ketoglutaric acid on embryogenic callus induction from the seeds ofZoysia japonica Steud. J. Plant Physiol. 149:413–417; 1996.
Bilang, R.; Zhang, S.; Leduce, N., et al. Transient gene expression in vegetative shoot apical meristems of wheat after ballistic microtargeting. Plant J. 4:735–744; 1993.
Capelle, S. C.; Mok, D. W. S.; Kirchner, S. C., et al. Effects of thidiazuron on cytokinin automony and the metabolism of N6-(2-isopentenyl)[814C] adenosine in callus tissue ofPhaseolus lunatus L. Plant Physiol. 73:796–802; 1983.
Denchev, P. D.; Conger, B. V. Plant regeneration from callus cultures of switchgrass. Crop Sci. 34:1623–1627; 1994.
Denchev, P. D.; Conger, B. V. In vitro culture of switchgrass: influence of 2,4-D and picloram in combination with benzyladenine on callus initiation and regeneration. Plant Cell Tissue Organ Cult. 40:43–48; 1995.
Gambley, R. L.; Fond, R.; Smith, G. R. Microprojectile transformation of sugarcane meristems and regeneration of shoots expressing β-glucuronidase. Plant Cell Rep. 12:343–346; 1993.
Graham, E. T.; Joshi, P. A. Novel fixation of plant tissue, staining through paraffin with Alcian blue and Hematoxylin and improved slide preparation. Biotech. Histochem. 70:263–266; 1995.
Iglesias, V. A.; Gisel, A.; Bilang, R., et al. Transient expression of visible marker genes in meristem cells of wheat embryos after ballistic microtargeting. Planta 192:84–91; 1994.
Irish, E. E.; Nelson, T. M. Development of maize plants from cultured apices. Planta 175:9–12; 1988.
Lowe, K.; Bowen, B.; Hoerster, G., et al. Germline transformation of maize following manipulation of chimeric shoot meristems. Biotechnology 13:677–682; 1995.
Lu, C. The use of thidiazuron in tissue culture. In Vitro Cell. Dev. Biol. 29P:92–96; 1993.
Mok, M. C.; Mok, D. W. S.; Armstrong, D. J., et al. Cytokinin activity of N-phenyl-N′-1,2,3-thidiazol-5-ylurea (thidiazuron). Phytochemistry 21:1509–1511; 1982.
Moser, L. E.; Vogel, K. P. Switchgrass, big bluestem, and indiangrass. In: Barnes, R. F., et al., ed. Forages, Vol. 1. 5th ed. An introduction to grassland agriculture. Ames: Iowa State Univ. Press; 1995:409–420.
Murashige, T.; Skoog, F. A revised medium for rapid growth and bioassay with tobacco tissue cultures. Physiol. Plant. 15:473–497; 1962.
Sanderson, M. A.; Reed, R. A.; McLaughlin, S. B., et al. Switchgrass as a sustainable bioenergy source. Bioresour. Technol. 56:83–93; 1996.
Thomas, J. C.; Katterman, F. R. Cytokinin activity induced by thidiazuron. Plant Physiol. 81:681–683; 1986.
tomes, D. Transformation in corn: non-sexual gene transfer. In: Twenty-sixth annual Illinois corn breeders school symposium at the University of Illinois, Urbana-Champaign; 1990:1–13.
Zhang, S.; Zhong, H.; Sticklen, M. B. Production of multiple shoots from apical meristems of oat (Avena sativa L.). J. Plant Physiol. 148:667–671; 1996.
Zhong, H.; Srinivasan, C.; Sticklen, M. B., In vitro morphogenesis of corn (Zea mays L.). I. Differentiation of multiple shoot clumps and somatic embryos from shoot tips. Planta 187:483–489; 1992.
Zhong, H.; Sun, B.; Warkentin, D., et al. The competence of maize shoot meristems for integrative transformation and inherited expression of transgenes. Plant Physiol. 110:1097–1107; 1996.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Gupta, S.D., Conger, B.V. In vitro differentiation of multiple shoot clumps from intact seedlings of switchgrass. In Vitro Cell.Dev.Biol.-Plant 34, 196–202 (1998). https://doi.org/10.1007/BF02822708
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02822708