Abstract
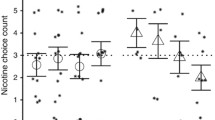
Mecamylamine is an antihypertensive that acts via nicotinic antagonism and has been suggested as an aid in smoking cessation. Nicotine dependent patients may not accept mecamylamine if it precipitates withdrawal, as it does in nicotine dependent rats. This study examined mecamylamine’s effects using procedures designed to measure precipitated withdrawal symptoms in humans. Ten cigarette smokers (mean of 37.5 cigarettes/day) and ten non tobacco-using subjects participated in three 6-h sessions. After a 2-h baseline period in which smokers smoked one cigarette every 30 min, oral mecamylamine (0, 10, or 20 mg randomly ordered across sessions) was administered (double-blind). No smoking was allowed for the remainder of the session. Mecamylamine reduced blood pressure and increased heart rate relative to placebo in both the smokers and the non-tobacco users. No reliable direct subjective effects of mecamylamine were observed. Smokers’ subjective reports of cigarette craving and tobacco withdrawal increased, and DSST performance was disrupted over the last 4 h of each session. Effects were independent of dose (placebo versus active). These results suggest that up to 20 mg mecamylamine will not precipitate nicotine withdrawal and that this medication would be acceptable for use in smoking cessation.
Similar content being viewed by others
References
Clarke PBS, Kumar R (1983a) The effects of nicotine on locomotor activity in non-tolerant and tolerant rats. Br J Pharmacol 78: 329–337
Clarke PBS, Kumar R (1983b) Nicotine does not improve discrimination of brain stimulation reward by rats. Psychopharmacology 79: 271–277
Eissenberg T, Greenwald MK, Johnson RE, Liebson IA, Bigelow GE, Stitzer ML (1996) Buprenorphine’s physical dependence potential: antagonist-precipitated withdrawal in humans. J Pharmacol Exp Ther 276: 449–459
Fagerstrom KO (1978) Measuring degree of physical dependence to tobacco smoking with reference to individualization of treatment. Addict Behav 3: 235–241
Griffiths RR, Bigelow GE, Liebson I (1983) Differential effects of diazepam and pentobarbitol on mood and behavior. Arch Gen Psychiatry 40: 865–873
Hall GH (1972) Changes in body temperature produced by cholinomimetic substances injected into the cerebral ventricles of unanesthetized cats. Br J Pharmacol 44: 634–641
Himmelsbach CK (1941) The morphine abstinence syndrome, its nature and treatment. Annu Int Med 15: 829–839
Hughes JR (1994) Non-nicotine pharmacotherapies for smoking cessation. J Drug Dev 6: 197–203
Hughes JR, Hatsukami DK (1986) Signs and symptoms of tobacco withdrawal. Arch Gen Psychiatry 43: 289–294
Hughes JR, Hatsukami DK, Pickens RW, Krahn D, Malin S, Luknie A (1984) Effect of nicotine on the tobacco withdrawal syndrome. Psychopharmacology 83: 82–87
Malin DH, Lake JR, Carter VA, Cunningham JS, Hebert KM, Conrad DL, Wilson OB (1994) The nicotinic anatagonist mecamylamine precipitates nicotine abstinence syndrome in the rat. Psychopharmacology 115: 180–184
Martin BR, Onaivi ES, Martin TJ (1989) What is the nature of mecamylamine’s antagonism of the central effects of nicotine? Biochem Pharmacol 38: 3391–3397
McLeod D, Griffiths RR, Bigelow GE, Yingling J (1982) An automated version of the digit symbol substitution test (DSST). Behav Res Methods Instrum 14: 463–466
McNair DM, Lorr M, Droppelman LF (1971) Profile of mood states (manual). Education and Industrial Testing Services, San Diego
Nemeth-Coslett R, Henningfield JE, O’Keefe MK, Griffiths RR (1986) Effects of mecamylamine on human cigarette smoking and subjective ratings. Psychopharmacology 88: 420–425
Newhouse PA, Potter A, Corwin J, Lenox R (1992) Acute nicotine blockade produces cognitive impairment in normal humans. Psychopharmacology 108: 480–484
Petrie RXA, Deary IJ (1989) Smoking and human information processing. Psychopharmacology 99: 393–396
Pomerleau CS, Pomerleau OF, Majchrzak MJ (1987) Mecamylamine pretreatment increases subsequent nicotine self-administration as indicated by changes in plasma nicotine level. Psychopharmacology 91: 391–393
Rose JE, Sampson A, Levin ED, Henningfield JE (1989) Mecamylamine increases nicotine preference and attenuates nicotine discrimination. Pharmacol Biochem Behav 32: 933–938
Rose JE, Behm FM, Westman EC, Levin ED, Stein RM, Lane JD, Ripka GV (1994a) Combined effects of nicotine and mecamylamine in attenuating smoking satisfaction. Exp Clin Psychopharm 2: 328–344
Rose JE, Behm FM, Westman EC, Levin ED, Stein RM, Ripka GV (1994b) Mecamylamine combined with nicotine skin patch facilitates smoking cessation beyond nicotine patch treatment alone. Clin Pharmacol Ther 56: 86–99
Schuh KJ, Stitzer ML (1995) Desire to smoke during spaced smoking intervals. Psychopharmacology 120: 289–295
Shiffman SM, Jarvik ME (1976) Smoking withdrawal symptoms in two weeks of abstinence. Psychopharmacology 50: 35–39
Snyder FR, Davis FC, Henningfield JE (1989) The tobacco with-drawal syndrome: performance decrements assessed on a computerized test battery. Drug Alcohol Depend 23: 259–266
Stitzer M, Morrison J, Domino EF (1970) Effects of nicotine on fixed interval behavior and modification by cholinergic antagonists. J Pharmacol Exp Ther 171: 165–177
Stolerman IP (1986) Could nicotine antagonists be used in smoking cessation? Br J Addict 81: 47–53
Stolerman IP, Goldfarb T, Fink R, Jarvik ME (1973) Influencing eigarette smoking with nicotine antagonists. Psychopharmacologia 28: 247–259
Tennant FS Jr, Tarver AL, Rawson RA (1984) Clinical evaluation of mecamylamine for withdrawal from nicotine dependence. NIDA Res Monogr 49: 239–246
Tiffany ST, Drobes DJ (1991) The development and initial validation of a questionnaire on smoking urges. Br J Addict 86: 1467–1476
Wu KM, Martin WR (1983) An analysis of nicotinic and opioid processes in the medulla oblongata and nucleus ambiguus of the dog. J Pharmacol Exp Ther 227: 302–306
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Eissenberg, T., Griffiths, R.R. & Stitzer, M.L. Mecamylamine does not precipitate withdrawal in cigarette smokers. Psychopharmacology 127, 328–336 (1996). https://doi.org/10.1007/BF02806011
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02806011