Abstract
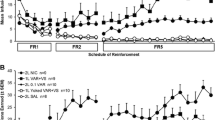
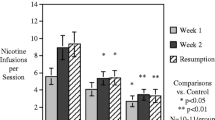
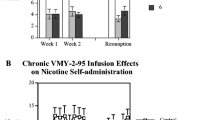
Recently, a rodent model of nicotine abstinence syndrome has been developed based on observing the frequency of spontaneous behavioral signs following termination of continuous subcutaneous infusion of nicotine tartrate. In the present study, the nicotinic antagonist mecamylamine precipitated an abstinence syndrome in nicotine-dependent rats. Twelve rats were each infused for 7 days with 9 mg/kg per day nicotine tartrate in saline via Alzet osmotic minipumps; another 12 rats were shamoperated and remained nicotine-naive. Six rats from each group received 1 mg/kg mecamylamine in saline SC immediately before a 30-min observation, while the remaining six rats from each group received saline alone. Nicotine-infused rats receiving mecamylamine exhibited significantly more (P<0.01), overall abstinence signs than all other groups. In terms of categories of signs, they displayed significantly more gasps/writhes, teeth chatter/chews, shakes/tremors and ptosis. In a second experiment utilizing only nicotine-naive rats, a far higher dose of mecamylamine (5 mg/kg sc) induced a quasi-nicotine abstinence syndrome. The results provide further validation for this rodent model of nicotine abstinence syndrome.
Similar content being viewed by others
References
Banerjee S, Punzi, JS, Kreilick K, Abood LG (1990) [3H] Mecamylamine binding to rat brain membranes. Biochem Pharmacol 40:2105–2110
Carroll ME, Lac ST, Asencio M, Keenan RM (1989) Nicotine dependence in rats. Life Sci 45:1381–1388
Clarke PBS, Kumar R (1983) The effects of nicotine on locomotor activity in non-tolerant and tolerant rats. Br J Pharmacol 78:329–337
Corrigall WA, Herling S, Coen KM (1989) Evidence for a behavioral deficit during withdrawal from chronic nicotine treatment. Pharmacol Biochem Behav 33:559–562
Davenport KE, Houdi AA, VanLoon GR (1990) Nicotine protects against mu-opioid receptor antagonism by beta-funaltrexamine: evidence for nicotine-induced release of endogenous opioid. Neurosci Lett 113:40–46
Gianutsos G, Drawbaugh R, Hynes M, Lal H (1975) The narcotic withdrawal syndrome in the rat. In: Ehrenpreis S, Neidle A (eds) Methods in narcotic research. Dekker, New York, pp 293–309
Gomita Y, Suemaru K, Furuno K, Araki Y (1989) Nicotine-induced tail-tremor and drug effects. Pharmacol Biochem Behav 34:817–821
Hall GH (1972) Changes in body temperature produced by cholinomimetic substances injected into the cerebral ventricles of unanesthetized cats. Br J Pharmacol 44:634–641
Helton D, Tizzano J, Modlin D, Rasmussen K (1991) Nicotine tolerance and dependence: a behavioral assessment using scheduled controlled responding, locomotor activity, and sensorimotor reactivity. Soc Neurosci Abstr 17:331
Herning RI, Jones RT, Bachman J (1983) EEG changes during tobacco withdrawal. Psychophysiology 20:507–512
Hexum TD, Russett LR (1987) Plasma enkephalin-like peptide response to chronic nicotine infusion in guinea pig. Brain Res 406:370–372
Hughes JR, Gust SW, Skoog K, Keenan RM, Fenwick JW (1991) Symptoms of tobacco withdrawal: a replication and extension. Arch Gen Psychiatry 48:52–59
Jensen RA, Gilbert DG, Meliska CJ, Landrum TA (1990) Characterization of a dose-response curve for nicotine-induced conditioned taste aversion in rats: relationship to elevation of plasma β-endorphin concentration. Behav Neural Biol 53:428–440
Levin ED, Castonguay M, Ellison GD (1989) Effects of the nicotinic receptor blocker mecamylamine on radial-arm maze performance in rats. Behav Neural Biol 48:206–212
Levin ED, Morgan MM, Galvez C, Ellison GD (1987b) Chronic nicotine and withdrawal effects on body weight and food and water consumption in female rats. Physiol Behav 39:441–444
Malin DH, Murray JB, Crucian GP, Schweitzer FC, Cook RE, Skolnick MH (1988) Auricular micro-electrostimulation: naloxone reversible attenuation of opiate abstinence syndrome. Biol Psychiatry 24:886–890
Malin DH, Lake JR, Newlin-Maultsby P, Roberts LK, Lanier JG, Carter VA, Cunningham JS, Wilson OB (1992) A rodent model of nicotine abstinence syndrome. Pharmacol Biochem Behav 43:779–784
Malin DH, Lake JR, Carter VA, Cunningham JS, Wilson OB (1993) Naloxone precipitates nicotine abstinence syndrome in the rat, Psychopharmacology 112:339–342
Mansner R, Mattila MJ (1975) Nicotine induced tremor and antidiuresis and brain nicotine levels in the rat. Med Biol 53:169–176
Marks MJ, Collins AC (1982) Characterization of nicotine binding in mouse brain and comparison with the binding of α-bungarotoxin and quinuclidinyl benzilate. Mol Pharmacol 22:554–564
Martin BR, Onaivi ES, Martin TJ (1989) What is the nature of mecamylamine's antagonism of the central effects of nicotine? Biochem Pharmacol 38:3391–3397
Martin TJ, Suchocki J, May EL, Martin BR (1990) Pharmacological evaluation of the antagonism of nicotine's central effects by mecamylamine and pempidine. J Pharmacol Exp Ther 254:45–51
Meltzer LT, Rosecrans JA (1981) Investigations on the CNS sites of action of the discriminative stimulus effects of arecoline and nicotine. Pharmacol Biochem Behav 15:21–26
Pierzchala K, Houdi AA, VanLoon GR (1987) Nicotine-induced alteractions in brain regional concentrations of native and cryptic met- and leu-enkephalin. Peptides 8:1035–1043
Pomerleau OF (1992) Nicotine and the central nervous system: biobehavioral effects of cigarette smoking. Am J Med 93 [1A]:2–8
Pomerleau OF, Rosecrans J (1989) Neuroregulatory effects of nicotine. Psychoneuroendocrinology 14:407–423
Pomerleau OF, Fertig JB, Seyler E, Jaffe J (1983) Neuroendocrine reactivity to nicotine in smokers. Psychopharmacology 81:61–67
Rose JE, Sampson A, Levin ED, Henningfield JE (1989) Mecamylamine increases nicotine preference and attenuates nicotine discrimination. Pharmacol Biochem Behav 32:933–938
Rosecrans JA, Hendry JS, Hong JS (1985) Biphasic effects of chronic nicotine treatment on hypothalamic immunoreactive beta-endorphin in the mouse. Pharmacol Biochem Behav 23:141–143
Shiffman SM, Jarvik ME (1976) Smoking withdrawal symptoms in two weeks of abstinence. Psychopharmacology 50:35–39
Stitzer M, Morrison J, Domino E (1970) Effects of nicotine on fixed interval behavior and modification by cholinergic antagonists. J Pharmacol Exp Ther 171:165–177
Stolerman IP, Pratt JA, Garcha HS, Giardini B, Kumar R (1983) Nicotine cue in rats analyzed with drugs acting on cholinergic and 5-hydroxytryptamine mechanisms. Neuropharmacology 22:1029–1037
Tripathi HL, Martin BR, Aceto MD (1982) Nicotine-induced antinociception in rats and mice: correlation with nicotine brain levels. J Pharmacol Exp Ther 221:91–96
US Department of Health and Human Services (1988) The health consequences of smoking; nicotine addiction. A report of the Surgeon General. DHHS Publication (CDC) 88–8406
Wei E, Loh HH, Way EL (1973) Quantitative aspects of precipitated abstinence in morphine-dependent rats. J Pharmacol Exp Ther 184:398–403
Wu KM, Martin WR (1983) An analysis of nicotinic and opioid processes in the medulla oblongata and nucleus ambiguous of the dog. J Pharmacol Exp Ther 227:302–307
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Malin, D.H., Ronald Lake, J., Carter, V.A. et al. The nicotinic antagonist mecamylamine precipitates nicotine abstinence syndrome in the rat. Psychopharmacology 115, 180–184 (1994). https://doi.org/10.1007/BF02244770
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF02244770